In the United States, over 300 billion beer, beverage and food cans are coated with half a million metric tons of Bisphenol-A (BPA)-containing epoxy resins each year, and the global market is more than twice that large. Although there are currently no U.S. Food and Drug Administration (FDA) or other U.S. regulatory restrictions on the use of BPA-based resins in most food containers, BPA-related health hazards have been recognized by regulators, policymakers and consumers. BPA is banned from use in applications such as infant feeding plastic bottles, and California recently listed BPA as a hazardous chemical.
Major brand owners such as Coca Cola, Heinz, Con Agra and Nestle have been actively working with coating suppliers to develop cost-effective and functional replacements for BPA-based epoxy resins in can coatings that have direct contact with food. The challenges are numerous for the alternative product, as the can coatings must achieve criteria such as: maintain adhesion under sterilization conditions; exhibit no chipping, flaking or peeling during handling and storage; not alter food taste; pass FDA guidelines on direct food contact use; be economically viable; and work across the well-established, various high-speed manufacturing processes.
Resin and coating manufacturers have made several attempts to develop a viable solution to this challenge. Chemistries such as vinyl, acrylic, polyester, polyolefin and a variety of crosslinkers have been tried but have not been successful. They fail in one or more key functional requirements, such as flexibility, adhesion, application method, cure speed, corrosion resistance or hydrolysis under low pH. In addition, the cost of the alternate solutions tends to be significantly higher than the incumbent, namely, BPA-containing epoxy resins.1,2
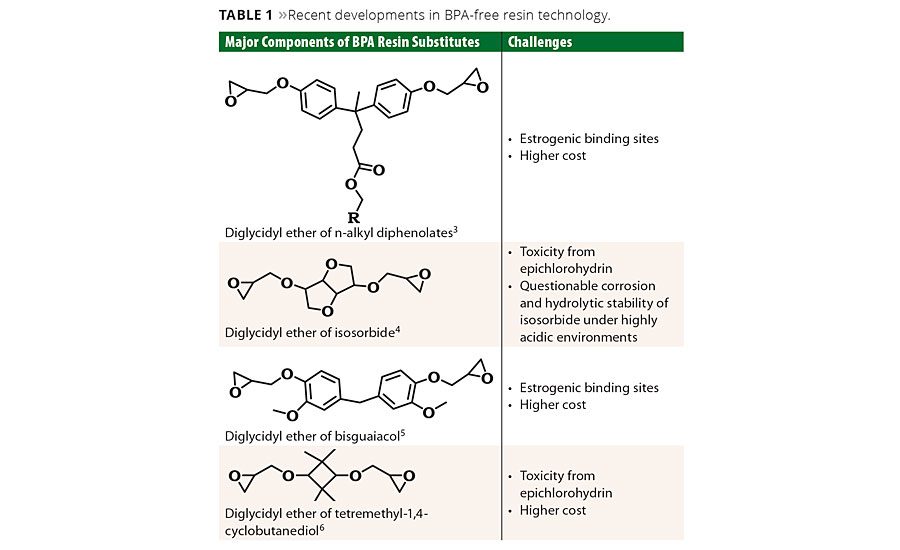
Recent developments introduced as BPA substitutes are still epoxy-based resins; they are derived from a BPA-like structure and use the oxirane ring for the resin cure (Table 1).
Oleoresins (derived from oxidative drying of natural oils) were used as can coatings before the emergence of BPA resin. The latter’s dominance is due to its faster cure speed, excellent corrosion performance and lower cost. The recognized health hazards with BPA resin have led many can coaters to revisit oleoresins; however, slower kinetics of the oxidative drying do not work with current high-speed can coating operations. Metallic driers cannot be used to accelerate the cure speed, as they are not permitted for food-contact use. The resins derived from epoxy, hydroxyl or carboxyl derivatives of natural oil have been studied as alternatives to BPA resin, but they do not have adequate corrosion and acid resistance, mainly because of the presence of an open micelle structure in the cured coating.7
Earlier research suggests resins based on the acetoacetyl functional group have superior corrosion resistance compared to their hydroxyl or carboxyl counterparts.8 Acetoacetylation of vegetable oil-modified resins (alkyds) has been reported. In such cases, acetoacetylation is carried out to reduce the viscosity of the resin. The concentration of acetoacetyl functionality in the final alkyd resin is too low to form a tight, crosslinked network typically required for corrosion protection. Recently, polyols derived from vegetable oil have been acetoacetylated9 and used as toughening agents in adhesive formulations. Acetoacetylated vegetable oil is not commercially available, and their potential in coating formulations has not been studied. In our research, soybean oil is modified with acetoacetyl functionality. The resultant acetoacetylated soy derivative (Soy-PK™) is formulated with Cymel®303, an FDA-approved -food-safe crosslinker.
Results and Discussion
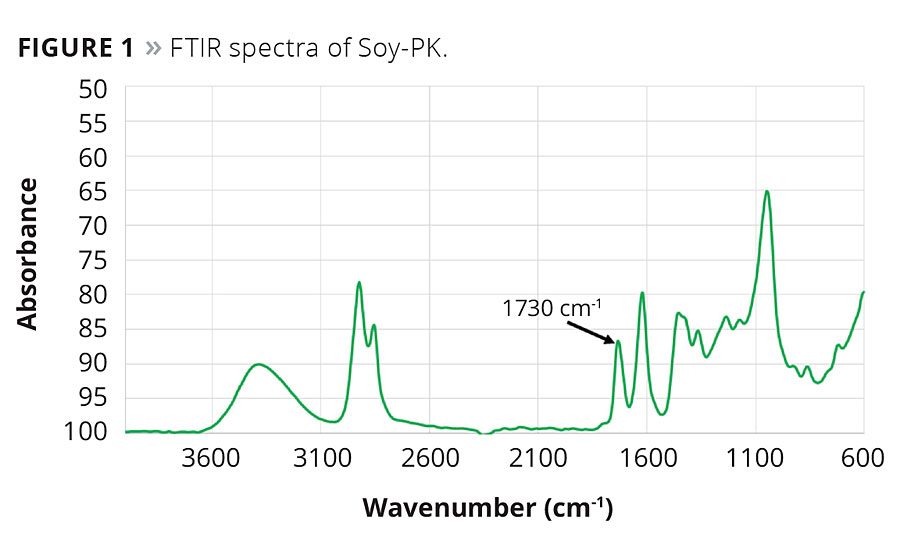
Soy-PK was obtained using a solvent-free acetoacetylation process. Fourier Transform Infrared (FTIR) spectra of the product is given in Figure 1, and the peak at 1730 cm-1 is characteristic of the acetoacetoate functional group.
This new resin is 100% solids and can be diluted with methyl ethyl ketone (MEK). Its physical properties are provided in Table 2.
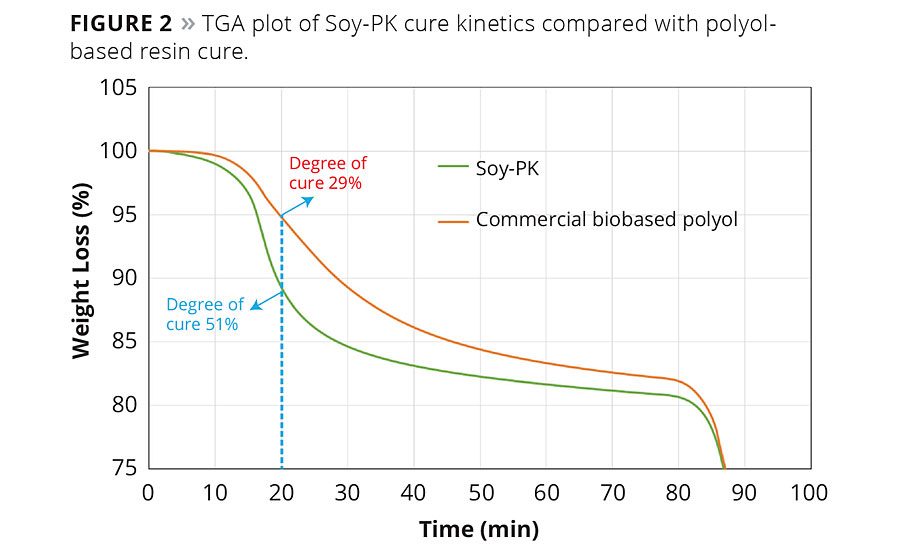
The acetoacetyl functional group present in the Soy-PK reacts with wide varieties of functional groups such as amines, hexamethylol melamine, isocyanates and acrylics. Therefore, it can be cured with wide varieties of commercially available crosslinkers such as Anacamine®2049, Cymel 303 and polymeric isocyanates to obtain the desired coating performance. It was also observed from Thermogravimetric Analysis (TGA) that Soy-PK cures faster than commercial biobased polyol. The TGA plot of Soy-PK curing with Cymel 303 is compared with biobased polyol and shown in Figure 2.
The degree of cure, a, can be calculated from the TGA data using the following equation:10
|
∆mt,T a = ———— ∆y |
(1)
|
Where Dmt,T is the difference in mass at time t and temperature T; and Dy is the derivative at the given cure temperature T. The derivative at 200 °C for Soy-PK and polyol-based resin is respectively 19.2 and 17.9%. Therefore from Equation 1, the degree of cure at 20 min for Soy-PK is 51% and for commercial biobased polyol is 29%.
Three parts of Soy-PK were mixed with one part of Cymel 303 and applied as a 2-mil wet film thickness on an aluminum panel. The coating was cured at 190 °C for 20 min. The performance of coatings obtained from Soy-PK resin is compared with that of commercial BPA resin coatings in Table 3.
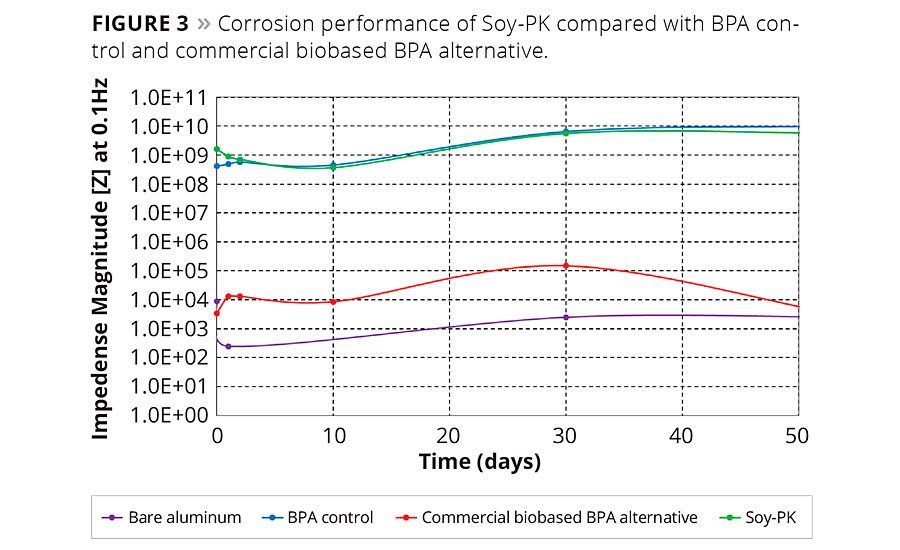
The corrosion performance of Soy-PK is evaluated using Electrochemical Impedance Spectroscopy (EIS). For this purpose, the coating is exposed to 3.5 wt% NaCl and the impedance is measured using a PAR potentiostat/galvanostat and Solartron equipment between the frequency range of 0.01 Hz to 65 kHz. The total coating impedance at a frequency of 0.1Hz is used as a guide to predict the corrosion performance of a coating.11 The performance for the Soy-PK coating over a period of 50 days is shown compared to traditional coatings (Figure 3). It is evident that Soy-PK is on par with the corrosion performance of BPA-based resin and outperforms commercial biobased BPA-free alternative coatings.
The toxicity of Soy-PK was assessed using BG1LUC assay as per the previously reported procedure.12,13 It has no detectable estrogenic or anti-estrogenic activity (Figure 4).
Conclusions
We have developed a BPA-free coating based on acetoacetyl-modified soybean oil (Soy-PK) and demonstrated its potential application for coating food and beverage containers. Soy-PK showed comparable performance to that of epoxy coatings derived from BPA. Toxicity studies suggest it does not have adverse health and environmental effects, as illustrated by no detectable anti-estrogenic and estrogenic activity. This study demonstrated this new resin is a drop-in replacement for BPA-containing epoxy resins in beer, beverage and food can coating applications. Its ability to crosslink with multifunctional isocyanates, amines and acrylate oligomers will further provide a myriad of opportunities to formulators and coating companies to develop novel and differentiated products based on renewable resources.
Acknowledgements
The authors RSL, RJC and MJP are grateful to Ohio Soybean Council for the financial support.
References
1 Bomgardner, M.M. Chemical and Engineering News2013, 91 24-25.
2 Lakind, J.S. Int. J. Technol. Policy Management2013, 13 80-95.
3 Maiorana, A.; Spinella, S.; Gross, R.A. Biomacromolecules 2015, 16 1021-1031.
4 Hammond, W.; East, A.; Jaffe, M.; Feng, X. US Patent 20150307650, 2015.
5 Reno, K.; Havery, J.; Stanzione, F.; Iii, R.; Wool, P.; Sadler, J.; J. Lascala, J.; Hernandez, E.; PCT WO/2015/183892, 2015.
6 Schmidt, D. US Patent 9139690, 2015.
7 Robertson, G.L. Food Packaging: Principles and Practice. 2nd ed., CRC Press, 2006.
8 Del Rector, F.; Blount, W.W.; Leonard, D.R. J. Coat. Technol.1989, 61 31-37.
9 C. J. Campbell, K. M. Lewandowski and K. Owusu-Adom US Patent 20130217804, 2013.
10Gotro, J. Thermoset Cure Kinetics Part 7: TGA Kinetics. http://polymerinnovationblog.com/thermoset-cure-kinetics-part-7-tga-kinetics/ (accessed December, 2015).
11Buchheit, R.G.; Cunningham, M.; Jensen, H.; Kendig, M.W.; Martinez, M.A. Conference Proceedings of the Meeting of the Electrochemical Society, October 1996, San Antonio, TX.
12OECD Guideline for the Testing of Chemicals: BG1Luc Estrogen Receptor Transactivation Test Method for Identifying Estrogen Receptor Agonists and Antagonists. http://ntp.niehs.nih.gov/ntp/about_ntp/sacatm/2012/september/d_draft_oecdtg457bg1luc_508.pdf (accessed Septemper 2015)
13Bittner, G.; Denison, M.; Yang, C.; Stoner, M.; He, G. Environmental Health 2014, 13 103.
For more information, email lalgudir@battelle.org or call 614/424.7679.
This paper was originally presented at the 2016 Waterborne Symposium in New Orleans.









Report Abusive Comment