EU Biocidal Product Regulations – One Year Later
Treated Article Provisions and What They Mean For You
Credit: The REACH Centre
The Biocidal Products Regulation No. 528/2012 (BPR) has been in force in the EU since September 1, 2013. It concerns placing on the market biocidal products, the active substances used in biocidal products and treated articles incorporating active substances. The BPR repealed and replaced the Biocidal Products Directive 98/8/EC (BPD) and, amongst other features, includes extended provisions for treated articles marketed in the EU.
This revision of scope has resulted in the BPR impacting industries that previously had no biocide-related obligations, and who may not have been prepared for a role under the BPR, including suppliers and manufacturers of finished goods containing preservatives. The use of preservatives in finished goods from textiles to wooden furniture, and from coatings to food-contact materials is widespread and well established. Companies in such industries need to be aware of these new requirements and any new obligations, particularly when importing products containing preservatives from outside the EU.
Defining Treated Articles
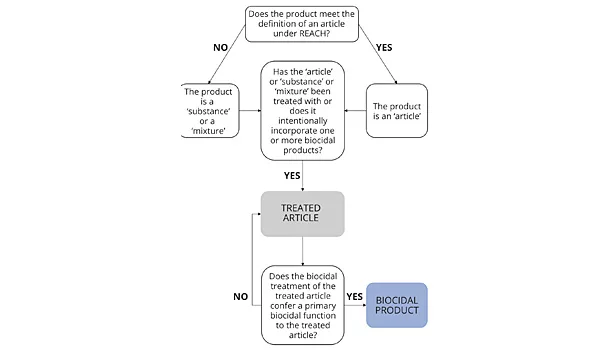
It is crucial to define what is meant by the term ‘treated article’. Those familiar with the REACH Regulation (Registration, Evaluation, Authorisation and Restriction of Chemicals) (Regulation (EC) No 1907/2006) will be aware that an article is defined as ‘an object, which during production is given a special shape, surface or design that determines its function to a greater degree than does its chemical composition’. The meaning of a treated article within the BPR takes things a step further in that it includes articles, as per the meaning of REACH, along with substances and mixtures that are treated with or intentionally incorporate one or more biocidal product. The biocidal product may be present either to protect the treated article itself e.g., in the case of a preservative added to a water-based paint or adhesive, or to contribute to the function of the treated article, e.g., in the case of a disinfectant added to an anti-microbial paint for use in hospital environments. It is this specific function of the biocidal product that determines the obligations placed on suppliers of the treated article. In the case of the latter scenario, treated articles with a biocidal function could fall under the scope of the definition of a biocidal product, which brings with it more extensive obligations on the supplier.
To add further complexity to the situation, since treated articles were not included in the scope of the BPD, there are a series of transitional measures in place with regard to the marketing of treated articles. Treated articles will eventually only be allowed on the EU market if the active substances used to treat them have been included in the EU positive list – known as the Union List. However, the transitional measures allow treated articles to be placed on the EU market during the time the active substances it has been treated with are being evaluated for inclusion in the positive list. In certain cases, this may require submission of a dossier of information on the active substance(s) to the European Chemicals Agency (ECHA).
This requirement to submit information on the active substances results from the need for all active substances on the market in the EU to be approved in accordance with EU standards, and it is this level of robustness that industry, both EU and non-EU, has indicated may be problematic when applied to all treated articles. There is concern from industry regarding the import of complex articles into the EU, and the compliance requirements for their component articles, as well as concern across the board from industries using preservatives in manufacturing, which may or may not be present in the finished goods.
On a positive note, there is already evidence that the ECHA and the EU Commission are listening to industry with regard to treated articles, as demonstrated in the amendment to the BPR, published in April 2014 (Regulation (EU) No 334/2014). This amendment saw the removal of an unforeseen market freeze affecting new treated articles (those not on the market on or before September 1, 2013), which would have been forced to remain off the market until all relevant active substance evaluations had been completed and approved.
Obligations
With reference to the example of a water-based paint containing a preservative, the obligations under the BPR are reasonably straightforward, especially where no biocidal claims are made on the finished label. In such a case, the treated article should be labelled with any relevant instructions for use, including any precautions to be taken, if this is necessary to protect humans, animals and the environment. However, it can reasonably be expected that such information is likely to be already included on the standard product label.
In addition to the label consideration, however, suppliers of treated articles should be aware of the need to respond within 45 days to consumer requests for information on the biocidal treatment of the treated article, free of charge. This area of compliance would appear to be based on a comparable requirement to respond to consumer requests for information on the SVHC content of articles as outlined under the REACH regulation. As such, suppliers may already have a mechanism in place to cover the response to consumer requests under the REACH regulation, and may be in a position to adapt this mechanism to include the treated article information request.
In cases where a biocidal claim is made on the label of a treated article, e.g., if the label includes a statement such as ‘Contains a preservative to control microbial deterioration’, then the label must also contain a statement that the treated article incorporates a biocidal product; the biocidal property; the name of all active substances contained in the biocidal product; the name of all nanomaterials contained in the biocidal product, followed by the word ‘nano’ in brackets; and any relevant instructions for use, including any precautions to be taken due to the presence of the biocidal product. This labelling requirement can also be imposed upon treated articles containing certain active substances based on the evaluation of the active substance(s).
Primary Biocidal Functions
A far more complex procedure is in place for the formal authorization of treated articles deemed to have a primary biocidal function, e.g., an antimicrobial surface paint, where the active substance is present to protect external materials such as the wall, rather than just the paint itself. Such treated articles are defined under the BPR as biocidal products, and consequently must be authorized in accordance with the requirements for biocidal products. The authorization procedure involves the submission of a dossier of information on the product, for subsequent evaluation by the ECHA.
The term ‘primary biocidal function’ is not defined in the BPR but is covered in treated article guidance documents issued by the EU Commission. A treated article will generally be considered to have a primary biocidal function if the treated article has one or more function and the biocidal function is of first rank, importance or value, compared to the other functions. Whether the biocidal function is considered to be primary will depend on a number of considerations such as the prominence of the claim in relation to other property claims; or whether the claim is one related to public health effects, e.g., a claim of effectiveness against MRSA bacteria. The decision as to the primary function of a treated article will in the end be made on a case-by-case basis, with input from the Member States.
Strategies
Clearly the obligations placed on suppliers and importers of treated articles have increased significantly, and now industries with no previous biocide concerns need to consider what steps are needed to ensure compliance. Industry is advised to adopt a best-practice approach to allow identification of what actions they may need to take, including:
- Being aware of the products in your portfolio that contain preservatives at any quantity (note there is no tonnage consideration for biocides obligations). This may require communication along the supply chain, particularly where products are manufactured outside the EU.
- Where preservatives and other active substances are present in your products, reviewing the status of the active substances in relation to the stages of evaluation under the BPR.
- Identifying any treated articles that are marketed for their biocidal function, based on intended uses and labelling claims.
- Considering any additional labelling requirements for treated articles, including those with biocidal claims.
- Generating a mechanism to allow consumer requests to be managed within the defined timescale. This will involve ensuring that as much information as possible is known on each of the treated articles, including those imported into the EU.
- Keeping up to date with new developments regarding treated article regulation, based on discussions between industry and the regulators.
For more information, visit www.thereachcentre.com.
By Siobhan Murphy, Senior Regulatory Scientist, The REACH Centre, Lancashire, UK
Useful Definitions
Active Substance– A substance or micro-organism that has an action on or against harmful organisms, including pathogenic agents, which have an unwanted presence or a detrimental effect on humans, their activities or the products they use or produce, on animals or the environment.
Biocidal Product– Active substances and preparations consisting of, containing or generating one or more active substances, put up in the form in which they are supplied to the user, intended to destroy, deter, render harmless, prevent the action of, or otherwise exert a controlling effect on any harmful organism by chemical or biological means.
Treated Article– Any substance, mixture or article that has been treated with, or intentionally incorporates, one or more biocidal products.
Union List– List of active substances that have been assessed as having hazards that can be sufficiently controlled to allow their effective use in certain biocidal product types. The ‘Union list’ is the replacement for Annex I of BPD.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




