Advanced Silica Gel Products for Gloss Reduction and Corrosion Protection
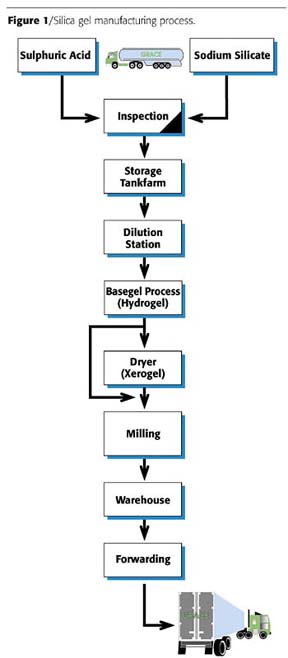
Mixing sulphuric acid and sodium silicate under controlled conditions produces the base gel from which all types of silica gels are made. This glass-like, solid gel material is broken down into granules, then washed and dried to produce the highly porous material needed for matting agents (Figure 1). By control of the washing and drying conditions, the important physical parameters such as porosity, pore size and surface area can be manipulated to produce a range of different silica gel types .1
The physical properties of silica gel differ from other specialty silicas. The internal structure of silica gel is composed of a large network of interconnected microscopic pores that attract and hold water, hydrocarbons and other chemicals by the mechanism of physical adsorption and capillary condensation. This huge pore volume and extensive surface area gives the silica gel many of its unique properties.
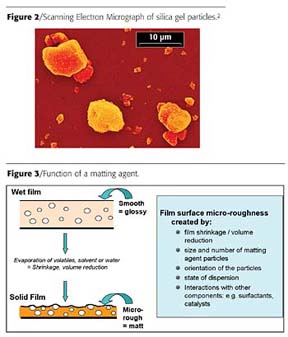
Silica gels are available in a wide range of particle sizes, each type having a well-defined particle size distribution. A typical example is depicted in Figure 2.
Important commercial properties of silica gels with respect to incorporation into coatings are:
- good incorporation and dispersion properties;
- outstanding rigidity;
- high purity;
- controlled particle size; and
exceptional chemical inertness.
Matting Agents
Silica gels are widely used as matting agents in coatings. The rigidity of the silica gel particle is higher than that for precipitated silica and thus better resists the shear forces arising during the manufacture and application of the paint. Silica gels can influence rheological properties, although other agents are typically preferred for rheological control purposes.The matting effect is based on solvent evaporation accompanied by development of surface micro-roughness due to the presence of the particles provided by the matting agent, as indicated in Figure 3.
In this process, the efficiency of the matting agent depends on the type of silica, particle size distribution and porosity of the particle. A high porosity is a key feature of a modern matting agent as it enables the formulator to limit the addition rate of a matting agent in a formulation. Unfavourable viscosity effects as well as limitations in surface properties of the coating can be avoided. A new milestone in micronized silica gel technology was reached by achieving a pore volume of more than 2 ml/g for a micronized silica. Today, matting agents for industrial applications are available in a variety of particle sizes.3
Matting agents are used to reduce the gloss in a variety of coatings applications. The application segments for silica gel matting agents include coil coatings, industrial wood coatings, general industrial and automotive coatings as well as printing inks, leather/textile coatings and decorative coatings. Coil, wood and furniture coatings are the applications with the largest volumes, but radiation-cured coatings and other coating systems as well as printing inks also employ silica gels. The solventborne paint sector has been more important for silica gel than waterborne coatings. However, the pressure to reduce VOC emissions and formulate solvent-free paints has led to the development of grades of silica gel that are suitable for formulation in powder, ultraviolet and waterborne coatings.
Suppliers also offer organically treated silica gels for use as matting agents. These products are recommended for use in clear varnishes because they have somewhat better anti-settling, dispersion and mar-resistance properties in comparison with untreated grades.

Applications
Coil Coatings
Coil coating is the continuous process in which coils of metal strip are cleaned, pre-treated and finished with organic coatings. The stoving finishes applied, referred to as "coil coatings", are based on a variety of resin types and applied to substrates such as steel, galvanized steel and aluminium. The end applications of the resulting pre-painted metal strip determine the type of substrate, pre-treatment and specification of the organic coating applied. For many applications, matt or semi-matt finishes are desirable and specified.High-quality, high-performance stoving finishes such as coil coatings require high-quality, highly efficient matting agents. High-porosity (1.8-2.0 ml/g) silica gel grades give uniform, reproducible gloss reduction at low concentrations. Any potential influence on the excellent flexibility, durability and weathering resistance demanded of these coating systems can be minimized. The trend towards faster line speeds for economic reasons has brought the need for faster curing and more reactive coating systems. The latest silica gel grades have been developed with these applications in mind, and their low pH values prevent interference with acid catalysts used.
In building and construction applications (i.e. cladding, ceilings, partitioning) where large areas are involved, matt surfaces are more desirable than glossy surfaces for reasons of reduced glare. In addition, a low sheen-to-gloss (85°/60°) ratio is desired to avoid the impression of varying appearance when viewing from different angles. A matt finish with a low sheen-to-gloss ratio is best achieved by using coarse-sized silica gels with average particle sizes in the range 9-11 µm.
In the case of domestic appliances or automotive parts where gloss level is largely determined by fashion, the market prefers low gloss because matt finishes make surface damage (e.g. scuffing, scratching or marking) during manufacture or daily use less visually obvious. The coarser (APS 9 µm), untreated grades are often preferred for their higher matting efficiency. On the other hand, finer, surface-treated grades (APS 6-7 µm) give the advantage of greater smoothness and a more pleasant feel, together with a higher mar resistance. High-wax-content grades are also available, where superior mar resistance is required.
Thin-Film Coatings
Thin-film systems can be defined as coatings with an application dry film thickness of less than 10 µm. Typically these systems are used in applications like natural and artificial leather coatings as well as in foil and plastic sheet coatings.In thin-film applications there is a natural limitation on the particle size that can be tolerated. Depending on the solid content of the system and the dry film thickness targeted, wet film thickness may range from 5-30 µm. For obvious reasons, particles larger than the wet film will create serious application problems. Furthermore, particles that are large relative to the dry-film thickness lead to coarse surfaces and provide no advantage in matting effect.
Fine-sized, high-porosity (1.6-2.0 ml/g) silica gel matting agents with an average particle size of 3 µm, specially designed for such applications, are available. The narrow particle size distribution ensures a dispersion fineness below 10 µm, giving the optimum matting effect and a superior surface quality.
Radiation-Cured Coatings
The trend in the coatings market towards cost saving as well as environmental protection has led to increased application line speed and new technologies like UV, waterborne and powder coatings. UV-cured lacquers and thermosetting powder coatings undergo minimal or no shrinkage during film formation. Thus, matting in these cases is determined by other factors in comparison to conventional coatings.In Radcure systems, the curing characteristics have a large impact on the matting properties of the coating. For example, in UV-curing coatings, the speed of cure, in particular of the film surface, directly affects how easily a low-gloss coating can be achieved. Generally, slow-curing systems are relatively easy to matt, whereas the difficulty in reaching low gloss levels increases with increasing reactivity and cure speed.
In spite of these difficulties, good matting results have been obtained in UV-curing coatings using silica gel matting agents and careful formulation. Clear advantages were seen for surface-treated silica gel products of relatively low particle size. Consequently, new tailor-made grades have been introduced with fine particle sizes (4-5 µm), a higher content of specially selected wax treatments and reduced oil adsorption. This optimized combination of features reduces existing problems relating to application quality and enables the formulator to focus on previously unrealistic low-gloss targets. The benefits are already being appreciated by the industrial wood and foil finishing sectors.
Waterborne Coatings
Waterborne products have already acquired a significant share of the market in the industrial finishing of wood, plastic and metal substrates. The main criticism of waterborne technology was its failure to match the high-quality appearance and resistance properties provided by the solventborne systems they replaced. In this respect, advances in resin technology have opened up new opportunities in the industrial segment as well as in architectural applications.Shrinkage also occurs in waterborne systems and, in most cases, matting agents are similar to those used in solventborne coatings. The latest treated and untreated, high-porosity (2.0 ml/g) silica gel grades exhibit high compatibility with the available binder systems, such as polyurethane or acrylic dispersions and alkyd emulsions. The untreated grades tend to be preferred and provide highly effective matting at minimal addition levels, thus enabling the formulation of clear, low-gloss finishes with high transparency and chemical resistance properties. A range of particle sizes is available (APS 3-11 µm) to satisfy the demand of a wide range of application types and film thickness.
Incorporation and handling of pigments as well as the prevention of dust become more and more important in the coatings industry. Cost concerns, together with a more rigid EHS legislation, are driving raw materials suppliers to develop products addressing these needs (Figure 4). For this purpose, specially modified silica products are used. The porous structure of these so-called Hydrogel products is pre-wetted or filled with water.
The advantages of such pre-wetted products are:
- · low dust development;
· fast sink-in and reduced incorporation time;
· avoids coagulation of shear-sensitive binders; and
· reduced micro-foam generation and lower demand for anti-foam or defoamer additives.
Non-Toxic Anti-Corrosion Pigments Based on Modified Silica Gel
Environmental concern about the use of Cr(VI) compounds in anti-corrosion coating technology has resulted in the introduction and use of EHS-compliant products such as calcium-modified silica gel anti-corrosive pigments, especially in the general industrial and coil coatings segment. This has been a continuous and slow process but has been gaining great momentum in the last few years.The pressure on the European coatings industry towards implementation of VOC-free and EHS-compliant technologies significantly increased with the announcement of the EU VOC legislation that comes into effect in 2007 and the new EU-chemical legislation that is currently planned to become effective around 2008 to 2010. The target of the latter legislation is to ban all chemicals that may be considered as hazardous (approximately 20% of all chemicals used in the coatings industry). In response to this ever-increasing environmental pressure, producers of anti-corrosion primers are currently actively testing alternatives to anti-corrosive materials based on Cr(VI) and Zn compounds.
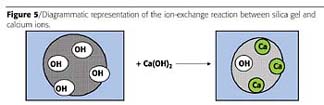
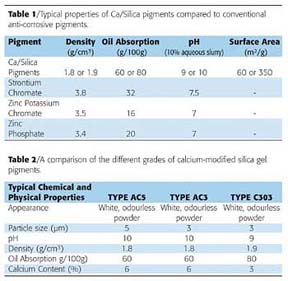
Calcium-modified silica gel anti-corrosive pigments (Ca/Silica pigments) offer a suitable, environmentally compliant alternative when considering future legislation changes. These pigments are non-toxic, heavy-metal-free, micronized, amorphous particles of controlled particle size distribution. The density of these pigments is about 1.8 g/cm3, which is quite low compared to conventional pigments. They also have a moderately alkaline reaction with water as indicated by the pH of an aqueous slurry, which lies in the region of 9-10. The manufacturing process involves an ion-exchange reaction at the surface of silica gel between weakly acidic surface silanol groups and calcium hydroxide, as indicated in Figure 5.
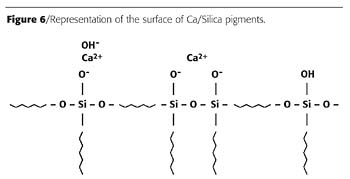
During the manufacturing process, the surface area and porosity of the original silica gel are reduced, which helps to avoid excessively high oil absorption and pigment activity. For reasons of efficiency in use, the surface area is still, however, maintained at a comparatively high level compared to other types of inhibitive pigments. Typical surface areas for the standard Ca/Silica pigments are around 60 m2/g, for example. Calcium ions distribute themselves in the surface layers of the silica particles and the surface of particles can be represented as in Figure 6, which shows how the calcium ions may be attached to the surface sites.

In general, when formulating with Ca/Silica pigments or alternatively, when replacing existing anti-corrosive pigments with Ca/Silica pigments, it is recommended that addition levels be worked out on a volume basis. However, addition levels expressed in weight on the total formulation for different paint systems often fall into the following ranges, and these can be seen as representing guideline addition levels:
- · 1-4 wt.% for waterborne paints;
· 3-6 wt.% for solventborne paints; and
· 3-9 wt.% for reactive primers (e.g. coil coatings).
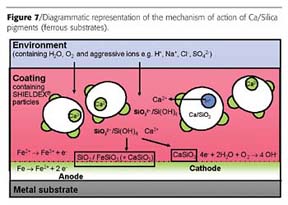
Briefly, water and aggressive ions permeating the coating lead to a release of calcium ions and soluble silica-inhibitive species, which diffuse and migrate to the interface where anode and cathode sites are beginning to develop on the metal surface as a result of ingress of the corrosive environment. At these sites, the inhibitive species form protective films of calcium silicate and other metal silicate compounds according to the nature of the metal being protected. These processes of film and compound formation result in effective suppression of the corrosion reaction. The alkaline pH of the pigments and their buffering properties may also contribute to the protective action by preventing the pH of the corrosive environment within the coating and in the vicinity of the interface from becoming too acidic, an aspect that is dependent on both the nature of the coating and the external environment.
Combinations with other inhibitors and anti-corrosive pigments are also possible, and synergic improvements in anti-corrosive performance have been seen when Ca/Silica pigments are combined with, for example, organic corrosion inhibitors or phosphate pigments. Benefits in adhesion and anti-corrosion performance of the coating system are also observed in specific instances when Ca/Silica pigments are combined with adhesion-promoting substances such as organofunctional silane, titanate and phosphate esters. A number of examples of these combinatorial possibilities are to be seen in the published and technical literature.4-7 Currently, three grades are offered, differing in either particle size or calcium content, as shown in Table 2.
The standard grade has an average particle size of about 5 µm and a calcium content of 6% by weight expressed as a percentage of the total pigment. It is used in all types of solventborne and waterborne systems where the dry film thickness of the coating is about 20 µm or more. For thinner films, Type AC3 is available. It also has a calcium content of 6% by weight, but an average particle size of 3 µm. It is used in wash primers, two-component epoxy and polyurethane coatings and is also employed in coil coating primers.

The lower half of the panels shows the primer result only; the top half shows the result for the primer/topcoat system, the topcoat being a polyurethane or medium oil alkyd respectively. Corrosion inhibition at the scribe and adhesion properties are equivalent to the chromate references. Both primer and topcoat were applied to a dry film thickness of 50 µm.
Figure 9 shows the excellent wet adhesion obtainable in a typical wash primer formulation based on polyvinylbutyral, containing a small amount of phosphoric acid when Type AC3 is combined with the epoxy-capped silane A187.
Exposure conditions were two weeks at 40 °C in distilled water, and the top half of the panels were overcoated with an acrylic/isocyanate topcoat at a dry film thickness of 40 µm. The panels were dabbed dry upon removal from the water bath and crosshatch and sticky tape removal adhesion tests were carried out immediately thereafter.
No signs of adhesion loss or blistering were evident in the case of the primer containing both Type AC3 and silane A187, whether tested as primer only or as a full system with topcoat.
In view of VOC issues, waterborne coatings are currently of course of great interest. Figure 10 provides an example of Type AC5 in combination with a second corrosion inhibitor in a waterborne alkyd applied over cold rolled steel. After salt spray testing, significant improvements in scribe adhesion were seen for the combination compared to the individual inhibitors. Underfilm corrosion was evident in the case of the uninhibited coating.
For the purpose of these tests, primers were applied at 40 µm dry film thickness to degreased cold rolled steel panels and after 7 days air-drying, submitted to hot salt spray for a period of 7 days.
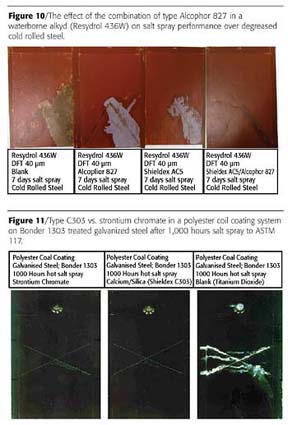
Typical results for Type C303 against strontium chromate and a blank based on titanium dioxide are shown in Figure 11, which refers to a polyester coil coating system applied over Bonder 1303 treated galvanized steel and tested for 1,000 hours in hot salt spray.
Corrosion inhibition and adhesion retention at the cut-edge and scribe together with resistance to white rust formation at the impact region for the system containing Type C303 is equivalent to the strontium chromate reference. Significant corrosion and adhesion loss is seen in the case of the uninhibited coating.
Long-term outdoor exposure testing of coil coating systems containing Type C303 at sites such as Bohus Malmön in Sweden confirm these laboratory test results.
Summary
The development of more environmental friendly coating systems presents new challenges to the manufacturers of speciality chemicals and additives for coatings.New matting agents designed to meet the specific challenges provided by waterborne, high-solids and, to some extent, UV-curing systems have been commercialized in recent years. However, there is a continuing demand for new, improved products, and development programs are ongoing.
It is expected that calcium-modified silica gel pigment technology will be introduced more and more as the preferred non-toxic method of corrosion control in all types of coatings, as the desire to couple high anti-corrosive performance with environmental awareness intensifies.
References
1 US Patent 5,110,769.2 Internal Source.
3 SYLOID" Technical Literature: Grace GmbH & Co. KG.
4 Fletcher, T. Ion Exchange Pigments for Anti-Corrosive Coatings. European Coatings Journal 1991,Vol 9, 553-560.
5 Fletcher, T. Ion Exchange Silicas as Alternatives to Strontium Chromate in Coil Coating Primers. Polymer, Paint, Colour Journal March 18th, 1992.
6 Zin, I.M.; Lyon, S.B.; Badger, S.J.; Scantlebury, J.D.; Pokhmurski, V.I. The effectiveness of chromate-free inhibiting pigments in coil coated galvanised steel. JCSE 1999, Volume 2, Paper 21.
7 SHIELDEX" Technical Literature: GRACE GmbH & Co. KG.
For further information, contact GRACE GmbH & Co. KG, phone +49-6241-403-0, fax +49-6241-403-0.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!








