New Developments in Pigment Dispersing Agents Improve Performance and Reduce Energy Consumption
The Dispersion Process
The dispersion process is generally considered to consist of three distinct stages that take place either sequentially or simultaneously. These stages are wetting, reduction of agglomerate size or separation, and stabilization. The dispersion system will lose some of its optimum properties should any of these factors be compromised. This decline in properties can manifest itself in such things as a loss of color development or dispersion stability.
Wetting
The wetting stage can be described as the process of bringing particle agglomerates into intimate contact with the grinding medium. It involves preferential displacement of gases, liquids and other contaminants from the surface of the pigment with the grinding medium.(2) The dispersing agent can assist greatly in this process by acting to form the direct grinding medium/pigment interface needed for creating a stable dispersion. In most cases, a substantial amount of the wetting process can occur without the use of mechanical energy, particularly when a proper, efficient dispersing agent is used.
Particle Size Reduction
This stage is also referred to as “dispersion” or deflocculation. It generally includes mechanical action, which leads to agglomeration size reduction. The eventual target is a particle size distribution that exhibits the optimum set of properties for the pigment system.
Stabilization
The stabilization process can also be known as controlled flocculation. There will be some minor flocculation in most dispersions as dispersant molecules are in motion.However, the best dispersants will keep the pigment particles separated to prevent interactions that would reverse the properties achieved by proper wetting and agglomeration size reduction. The polymeric dispersant molecule acts by adsorbing onto the pigment particle and providing a barrier against particle agglomeration.
To make the dispersion complete, it is important for the wetting stage to continue as the optimum particle size distribution is reached. As the agglomerates continue to be broken, the new particle surface needs to be wetted to avoid the reformation of the agglomerate. A normal dispersion process will have some reversibility, and an amount of flocculation will occur.

|
| Figure 1 Click to enlarge |
Dispersion Mechanisms
The literature generally sites two types of stabilizing mechanisms: charge stabilization and steric or entropic stabilization.(2) However, the polymeric dispersants introduced in this article also use what can be called “electrosteric” stabilization, a designed combination of the two well-known mechanisms.
Charge, or electrostatic stabilization, is a result of preferential adsorption of the charged dispersant molecules onto the surface of the pigment particle and the thickening of the charge layer within the surrounding medium.(3) The choice of dispersant can influence the magnitude of the repulsive energy, as can the ionic character of the medium. As a result, electrostatic stabilization is susceptible to and less stable in high electrolyte conditions.
Steric, or entropic stabilization, is a result of dispersant structure in which one functional moiety or anchoring group has a strong affinity for the pigment surface, and extending into the medium are solvated polymeric stabilizing chains.(4) The solvated chains lose their degree of freedom as particles approach each other, and this leads to repulsion to overcome the change in entropy. This method of stabilization is not affected by high electrolyte conditions.
|
| Figure 2 Click to enlarge |
Electrosteric stabilization is the combination of the two traditional types. It could be referred to as the best of both types.
Program Objective
The program objective was to evaluate three distinctly different types of polymeric dispersants to determine their performance in three separate case studies. In all three studies, these polymeric materials will be compared to competitive samples that are commercially used in the dispersion industry today.
Dispersant Candidate Types
The candidates evaluated in this program can be divided into three types. They are as follows.
Type 1
|
| Figure 3 Click to enlarge |
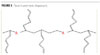
Type 1 of the polymeric dispersants tested can be described as a non-emulsifying dispersant in that it does not have the typical hydrophobe/hydrophile structure of a surfactant. These materials are specifically designed as dispersants. They have an anchoring group that has a strong, specific interaction with the pigment surface, and a polymeric stabilizing group that is compatible with the medium and extends out, providing steric stabilization. Figure 1 shows an example of such a structure with the anchoring group on one end and the branched stabilizing chain extending from it.
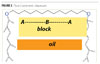
Type 2
The Type 2 polymeric dispersant evaluated is an ABA triblock copolymer. In the B chain it has multiple points of anchoring for excellent adsorption. The two stabilizing tails (A chains) extend into the medium for added density of the steric barrier. An example of the triblock material is shown in Figure 2.

|
| Figure 4 Click to enlarge |
Results and Discussion
Study 1: Organic Pigment Dispersed in a Nonaqueous Binder
A study was conducted that examined two different types of dispersants in a nonaqueous binder with three different types of organic pigments. Three separate paints were made with the nonaqueous binder and the pigments. All of the paints used a competitive dispersant that had been causing issues with dispersing the pigments. This study used a Type 1 anionic polymeric dispersant, with the nonaqueous binder and the three pigments; it compared paints with the competitive dispersant to paints with the Type 1 anionic polymeric dispersant.
Yellow Pigment Dispersed in Nonaqueous Binder
The first part of the study presents the data on a yellow organic pigment dispersed in a nonaqueous binder. The dispersant was added to the sample and the yellow pigment added at a 17% by weight loading level, indicating a low solids-type paint. The sample was then dispersated at 5000 rpm for 5 min.
Figure 4 shows the difference between the viscosities of the two paints based on the pigment dispersant used. Temperature point 1 was the viscosity taken at 20 ºC, whereas Temperature point 2 was the viscosity after the samples were exposed for 67 h at 50 ºC, and then allowed to cool down.

|
| Fiugre 5 Click to enlarge |
While the competitive dispersant showed reduced viscosity after exposure to 50 ºC for 67 h, the sample also had a large amount of agglomeration on the sides and bottom of the container. The Type 1 anionic polymeric dispersant showed lower viscosity in both tests, and did not have agglomeration in the container after testing at both 20 ºC and 50 ºC.
Orange Pigment 1 Dispersed in Nonaqueous Binder
The second part of the study involved dispersating an organic orange pigment (hereafter referred to as orange pigment 1) in the same nonaqueous binder. The dispersant was added to the sample, and the orange pigment 1 was added at a 14% weight loading level. After the additions, the paint was dispersated at 5000 rpm for 5 min.

|
| Figure 6 Click to enlarge |
Figure 6 shows the results of the viscosity testing on the paints made with the orange pigment 1 in the nonaqueous binder. Temperature point 1 shows the viscosity results taken at 20 ºC before exposure to higher temperatures. Temperature point 2 shows the viscosity results taken after the samples were exposed for 50 h at 50 ºC. While both samples showed increased viscosity after exposure to 50 ºC for 50 h, the competitive dispersant showed outgassing often associated with a reaction occurring between the binder and pigment, whereas the Type 1 anionic polymeric dispersant remained fluid, with no signs of reactivity between the pigment and binder.
Orange Pigment 2 Dispersed in Nonaqueous Binder

|
| Figure 7 Click to enlarge |
The third part of the study involved dispersing another organic orange pigment (hereafter referred to as orange pigment 2) in the same nonaqueous binder as the previous pigments. The dispersant was added to the sample, and the orange pigment added at a 14% weight loading level. After the additions were made, the paint samples were dispersed at 5000 rpm for 5 min.
Figure 7 shows the results of the paints after exposure to 50 ºC for 20 h. The sample on the left (JEH-1-66A) shows the reactivity that is occurring in the paint that was dispersed with the competitor dispersant, whereas the sample on the right (JEH-1-66B) shows no reactivity when the paint was dispersed with the Type 1 anionic polymeric dispersant.

|
| Figure 8 Click to enlarge |
The stability of the paints after 96 h is shown in Figure 8. The sample on the left is the paint with the orange pigment 2 dispersed in a nonaqueous binder with the competitive dispersant, whereas the sample on the right has the orange pigment 2 dispersed with the Type 1 anionic polymeric dispersant. The separation between the pigment and the binder is quite evident for the sample on the left. Separation of the binder and pigment is not seen with the sample on the right. Both samples retained their original pigmented color.
Study 2: Inorganic Pigment Dispersed in an Aqueous Binder
A study was done that examined the performance of two different pigment dispersants in an inorganic pigmented aqueous binder that had a pigment loading of greater than 70% by weight. The current dispersant, competitive dispersant 2, was unable to disperse the pigment fully, resulting in hard agglomerations on the bottom of the containers; or separation of the pigment from the binder. A Type 1 polymeric dispersant was used to overcome these issues in high pigment loading aqueous systems.

|
| Table 1 Click to enlarge |
While the pigment was added in at 70+% by weight loading level, each dispersant was used at 2% by weight on pigment. After the addition of the pigment, the paint was dispersed at a range between 1500 and 4000 rpm depending on the difficulty of dispersing the pigment. All samples were dispersed for 5 min.
The results shown in Table 1 indicate that the colorant that used the competitive dispersant 2 was unable to fully disperse the inorganic red iron oxide pigment when used at that high loading level.

|
| Figure 9 Click to enlarge |
The sample showed hard packing and displacement of the pigment from the binder. Type 1 polymeric dispersant was able to disperse the red iron oxide pigment in the aqueous binder and keep it dispersed with no displacement from the binder.
Figure 9 shows the viscosity measurements taken at different temperature points on the two colorants. Temperature point 1 was the initial viscosity taken after each sample was dispersed and cooled down. Temperature point 2 was viscosity measurements taken after exposure for 5 days at 70 ºC, then cooled down. Temperature point 3 was after 5 days at room temperature (20 ºC).

|
| Figure 10-11 Click to enlarge |
The competitive dispersant 2 samples had a greater viscosity at the start of testing and continued upwards, increasing in viscosity the longer the sample remained. The Type 1 dispersed samples showed decreased viscosity of the colorant over time, without increasing agglomerations or precipitating out the pigment from the binder.
The photo presented in Figure 10 shows the separation that occurred in the competitive dispersant 2 colorant after exposure at 70 ºC for 5 days.

|
| Figure 12 Click to enlarge |
Figure 11 shows that after 5 days of constant exposure to 70 ºC, no separation occurred between the pigment and the binder.
Study 3. Color Development and Viscosity Studies
Viscosity and color development studies were completed in the laboratory using 4-ounce paint cans. The cans were filled with the grinding medium and the appropriate type of polymeric dispersant. The pigment or metal oxide was slowly added while mixing to assure all the solids were wetted out. At this time, ceramic beads were added for grinding.
The mixtures were then put on a Red Devil paint shaker for 1 h with samples being removed every 15 min. Viscosity tests were performed only on the 1 h samples, while color development evaluation was done on the samples that were removed every 15 min.

|
| Figure 13 Click to enlarge |
The results of the viscosity studies completed showed significant advantage in using one of the polymeric dispersants. In Figure 12, the viscosity advantage using a Type 1 polymeric dispersant is readily evident. The formulator would therefore be able to significantly increase the solids loading and still have a usable final viscosity.
In both Figures 13 and 14, the 100% color development value was set by the 60 min grind for the control or competitive sample. In both cases, color development of greater than 100% was reached very quickly using the Type 3 polymeric dispersants. It is therefore readily apparent that, in addition to higher color development capability, the Type 3 polymeric dispersants in these evaluations offer the cost savings potential of grinding for less time to reach equal or greater color development. Thus, the producer will have two potential routes to cost savings.

|
| Figure 14 |
Conclusion
Three types of polymeric dispersants have been evaluated in the study presented. Each of the types has shown utility in application testing. It has been illustrated that these materials provide improved wetting and dispersion that results in efficient milling that can provide greater color development or higher productivity and energy savings.
Lower viscosity after grinding is also a feature of these polymeric materials. This viscosity advantage results in the ability to have higher solids loadings, resulting in higher productivity and lower inventory requirements.
Additionally, improved stability of the dispersions produced with the polymeric materials provides longer shelf life and therefore lower storage loss.
This paper was presented at the 38th Annual Waterborne Symposium in New Orleans.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






