Advances in Low-VOC Coalescent Technology




In the graphic arts industry, coalescents assist in film formation in waterborne overprint varnish,1,2 also known as OPV. To date, the coalescents used in the graphic arts industry have primarily been the more volatile types. While they function well, VOC content is an issue. Phthalates, such as di-n-butyl phthalate (DBP) or butyl benzyl phthalate (BBP), have also been used. Although they are both relatively low in VOCs, their use has been recently restricted as a result of health and safety concerns. Thus, a need exists for low-VOC, non-phthalate plasticizers and coalescent.
New-generation emulsions used in OPVs seem to be following the same trends that have been documented in other coatings industries. Specifically, the systems are softer and may not necessarily require any coalescent or plasticizer to form a film under their conditions of use. The older-generation systems, on the other hand, which do not necessarily facilitate the formation of film at room temperature, do provide excellent performance for different applications. One obvious solution would be the use of a low-VOC plasticizer/coalescent with the harder polymer systems to assist with film formation.
In 2011, a low-VOC plasticizer/coalescent platform was introduced specifically for use in architectural paint.3,4 This plasticizer platform is a blend of three dibenzoate plasticizers. The data presented indicated that one of the products, K-Flex® 975P,was particularly suited to the role of a plasticizer/coalescent in architectural coatings. In addition to the dibenzoate blends, a new monobenzoate was discovered and incorporated into this evaluation to test for feasibility in graphic arts applications.
The purpose of this article is to illustrate and present data5,6 that demonstrate the utility of K-Flex 975P as a low-VOC plasticizer/coalescent and to introduce the new monobenzoate as a plasticizer/coalescent in graphic arts applications.
Evaluation
A comparison of the properties of commonly used plasticizers/coalescents, new low-VOC materials and traditional coalescents was conducted by evaluating the characteristics in both the neat form and in a starting-point test formulation. This evaluation began by first determining the VOCs of the plasticizers/coalescents selected. Secondly, the effectiveness and efficiency of the coalescents with a basic polymer were determined using binary blends. The polymer selected for evaluation was a classic high glass transition styrenated acrylic emulsion that is often used in the graphic arts industry. Finally, the effect of the new plasticizers in a basic starting OPV formula was documented to demonstrate the likely effects of the new plasticizers in an actual OPV formulation.
The following plasticizers/coalescents were selected for this evaluation (in whole or part):
• 2,2,4-Trimethyl-1,3-pentanediol monoisobutyrate (TMPDMB): This is the historic coalescent of choice in paint and other coatings but not necessarily in OPVs.
• 2-Ethylhexyl benzoate (2-EHB): This coalescent was introduced a few years ago as a new lower-VOC type coalescent (Eastman product).
• Butyl Carbitol™ diethylene glycol monobutyl ether (BC) (DOW product).
• Carbitol™ diethylene glycol monomethyl ether (C) (Dow product).
• Butyl Cellosolve™ ethylene glycol monobutyl ether (BCL) (Dow product).
• Dipropylene glycol monomethyl ether (DPM) (Dow product).
• Diethylene glycol dibenzoate (DEGDB) (K-Flex DE made by Emerald Kalama Chemical).
• Di-n-butyl phthalate (DBP) (Generic).
• Butyl benzyl phthalate (BBP) (Santicizer® 160 made by Ferro).
• K-Flex 850S blend of diethylene glycol dibenzoateand dipropylene glycol dibenzoate (Emerald Kalama Chemical).
• K-Flex 975P dibenzoate proprietary triblend (Emerald Kalama Chemical).
• X-250 dibenzoate diblend (Emerald Kalama Chemical).
• X-613 new grade of monobenzoate (Emerald Kalama Chemical).
Testing was conducted on the materials, beginning with neat plasticizers. The plasticizers were then blended with the base polymer and finally in an OPV formulation to measure the relative performance and impact with the polymer alone and also on the end coating. Tests included:
• A TGA isothermal scan at 110 °C was conducted for one hour to determine the VOC levels of the neat plasticizers;
• MFFT and viscosity reduction were measured on binary polymer emulsion/coalescent blends;
• Viscosity response, MFFTs, gloss, water resistance, hardness, and dry-to-touch times were evaluated for the OPV test formulations.
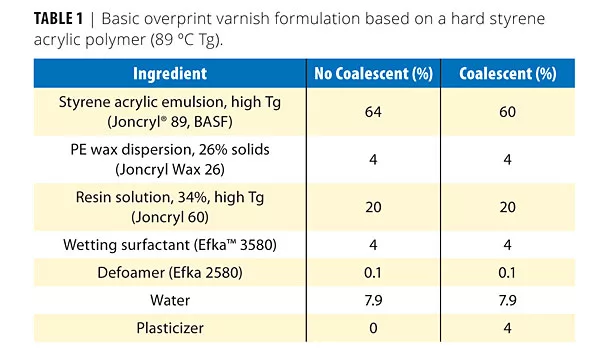
• The formulations and the details of the test methods are listed in Tables 1 and 4 respectively.
Results
Volatility of Neat Plasticizers/Coalescents
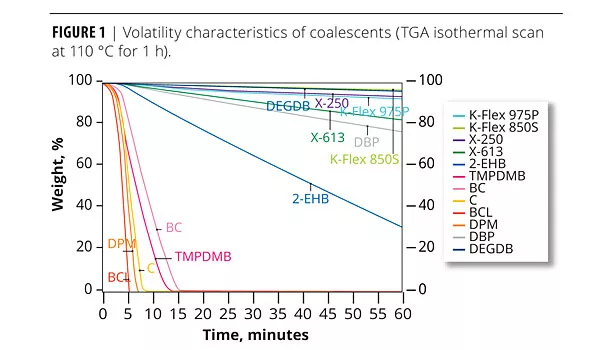
Volatility is an important characteristic, relating to the amount and rate at which VOCs leave the film. Figure 1 illustrates the volatility characteristics of coalescents in this evaluation. While traditional ether coalescents are more volatile, the plasticizers included in the evaluation are all low in volatility and would not contribute significantly to the overall VOC levels of a formulation at the typical levels of use. Obviously, the coalescents that are 100% VOC will contribute significantly to the total VOC release.
While more volatile than plasticizers, X-613 is significantly lower in VOCs than the older, more traditional high-VOC coalescents.
Binary Blends of Coalescent and Polymer
In this portion of the evaluation, coalescents were incorporated in the base emulsion at the same level of use as in the basic starting OPV formulation (4%). Important tests to see how coalescents affect the polymer are related to viscosity and water reduction (indicator of viscosity response), as well as MFFT. When adding a plasticizer or coalescent to a polymer, a change in viscosity may occur. The increase in viscosity of the plasticizer/polymer blend indicates the level of compatibility of the plasticizer with the polymer and the degree to which the plasticizer partitions into the polymer rather than the water phase. As shown in Figure 2, the higher viscosities of dibenzoate/benzoate chemistry also indicate a greater compatibility with the polymer than glycol ethers. The higher starting viscosity of the dibenzoate/benzoate chemistry may provide a benefit if the viscosity can be further reduced in the formulation of the OPV.
The viscosity of an OPV is normally reduced to a constant value to achieve consistent results for applications at the same viscosities. Water dilution levels can also have an economic impact on the cost in use of a given system, as water is naturally lower cost than the OPV formulation itself. The amount of water required to achieve the desired viscosity was determined and is illustrated in Figure 3. As expected, the amount of water required to decrease the viscosity was proportional to the difference between the initial and desired viscosity. This data demonstrate that the desired viscosity can be achieved by adjusting the OPV by diluting with water. Most of the coalescents tested could be adjusted by diluting with a similar amount of water, except for C and DPM, which required a lower level of dilution.
Minimum Film Formation Temperature (MFFT)
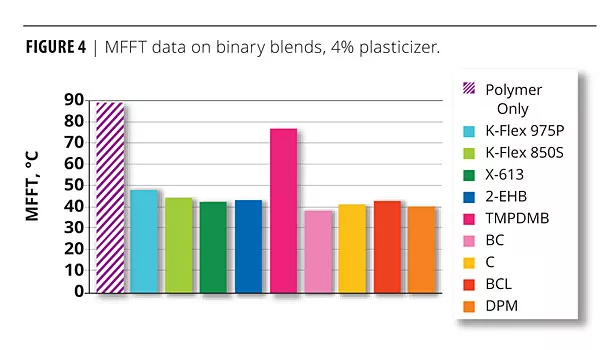
MFFT reduction is one of the key reasons for using a coalescent or plasticizer to assist in film formation. Figure 4 shows the MFFT of the binary blends with 4% wet plasticizer/coalescent added. Note that this low level of plasticizer had a very positive effect on the reduction of MFFT of the base polymer but did not bring it anywhere near room temperature. This is because this is an evaluation of a simple binary blend and not of a complete formulation.
As the MFFTs of the binary blends with 4% plasticizer were significantly higher than room temperature, it was decided to also evaluate the MFFTs of binary blends with 6% and 8% plasticizer levels. The results of that testing are illustrated in Figure 5.
The data developed were quite interesting and somewhat unexpected. The monobenzoate and dibenzoate blends decreased the MFFT significantly, as might be expected. Interestingly, the films formulated with TMPDMB had significantly higher MFFTs at all coalescent levels than the other films. It was also noted that the TMPDMB films were especially difficult to read on the MFFT bar due to the mottled film quality caused by the poor compatibility of TMPDMB with the styrenated acrylic polymer emulsion. Because of this obvious incompatibility, the use of TMPDMB was discontinued for the remaining portion of the evaluation.
After the 4% level, the Carbitol and Butyl Cellosolve did not decrease MFFT as would be expected. This would suggest a degree of incompatibility with the base polymer. A similar type of effect has been noted in other polymers when incompatibility exists.6 In the case of the cited reference, the effect noted was on Tg, not MFFT, but it is known that MFFT and Tg are related. The more hydrophobic the polymer, the closer MFFT and Tg temperatures are to each other. K-Flex 975P seemed to be particularly effective in MFFT suppression as was X-613.
The next step in this evaluation was to see if the experimental plasticizers/coalescents could perform as required in a basic starting formulation at the level tested in the binary blends.
Overprint Varnish
The coalescents were incorporated in the basic starting formulation listed in Table 1. Note the level of use of the plasticizer/coalescent was 4%. The viscosity response, as well as the dry film properties, were determined.
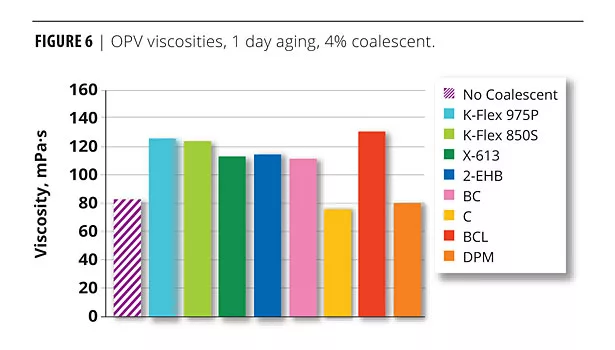
Figure 6 illustrates the OPV viscosity response with 4% plasticizer/coalescent. The viscosities of the dibenzoate OPVs were all in the expected range. The DPM and C OPVs viscosities were low. These coalescents are water soluble and did not partition (at least not completely) to the polymer.
The viscosity response to select dibenzoates was evaluated at a higher level of a 6% coalescent versus the 4% shown in the test OPV formulation in Table 1. Both the K-Flex 850S and K-Flex 975P OPVs were 250 mPa·s in viscosity. This evaluation confirmed that a relatively low concentration of plasticizer does have a significant impact on OPV viscosity with these types of plasticizers/coalescents.
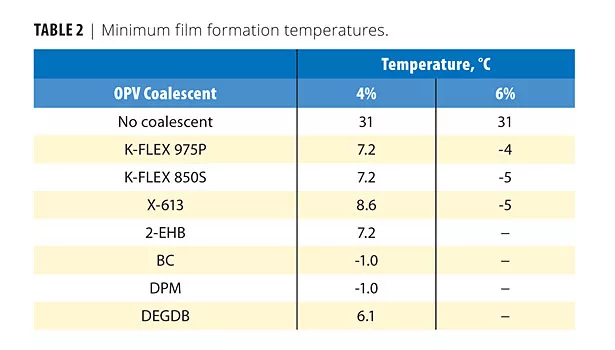
Table 2 lists the MFFTs of the OPV formulations. All of the formulations would form films well at room temperature conditions. The water-soluble coalescent types were more effective in MFFT suppression. As the MFFT depression was somewhat less for the dibenzoates than the ethers, the MFFTs of OPVs with loading at 6% wet of the monobenzoate and two of the dibenzoates were also determined. These results are also listed in Table 2 and indicate that less than the additional 2% would be necessary to achieve results similar to the ethers. Most likely this additional plasticizer would not be necessary to achieve the desired development of full performance characteristics.
Dry Time
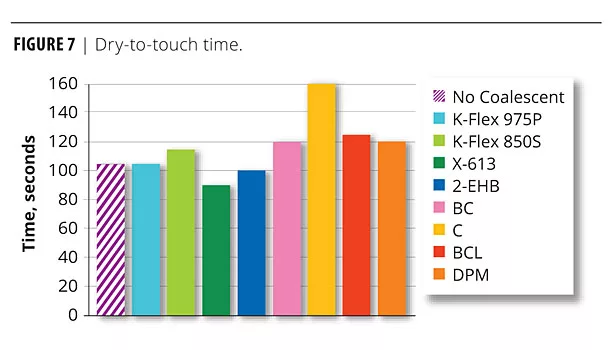
There was also interest in determining the effect on such parameters as dry time when replacing a volatile coalescent with a real plasticizer. The dry-to-touch time of the OPVs was determined and is shown in Figure 7.
With the exception of Carbitol, no significant difference in time to dry was noted between the volatile and non-volatile plasticizers and the coalescents.
Gloss
There was little difference in the 20° gloss values between the coalescents tested and the basic formulation without coalescent (all about 80). This is likely due to the use of the solution resin and its contribution to gloss in the total formulation, as well as the plasticizer/coalescent level.
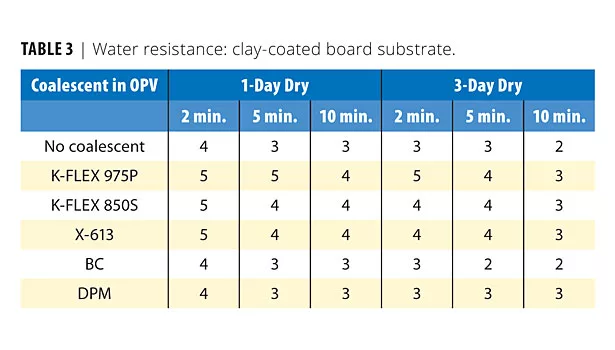
Water Resistance
Water resistance is an important feature in OPVs and other coatings. Table 3 lists the water resistance of the OPVs. The initial water resistance of the benzoate plasticized films was quite good. This is somewhat unexpected as the plasticizer does not leave the films as quickly as the volatile coalescents. After three days, the water resistance was similar to the initial data. The benzoate plasticizers, particularly K-Flex 975P, may provide benefits in water resistance by imparting its characteristic hydrophobic nature to the polymer film.
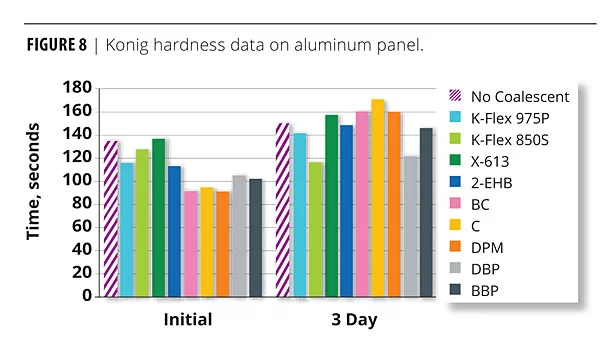
Hardness
Figure 8 displays the hardness data on the OPVs. There is a misperception that plasticizers will not perform as well in coatings because they are more permanent than very volatile coalescents and, as a result, they will stay and soften the film too much. The data prove that this is not true. The data presented in Figure 8 are contrary to the “too soft” statement. In a properly formulated coating that meets coalescence expectations, low-VOC plasticizers/coalescents will not necessarily soften too much. The 6% plasticizer films are somewhat softer but, in truth, they are somewhat over coalesced. Less would be better and acceptable. The 4% OPVs are all similar to the OPVs formulated with much more volatile coalescents. The films with plasticizers are not soft and do not fall short in performance as evidenced by the water resistance, gloss, hardness and other data in this study. This is consistent with conclusions from previous studies in which physical properties, such as scrub resistance, gloss retention and MFFT suppression, were found to be excellent in architectural coating formulations.5
Conclusion
The graphic arts and paper coating industries are seeking to reduce VOCs and select more environmentally friendly replacement materials in lieu of the higher-VOC materials currently in use. Several commercial dibenzoate and newer dibenzoate and monobenzoate coalescents that demonstrated performance benefits in other applications were screened in this study. These dibenzoates and monobenzoate offer the formulator solutions that are both lower in VOCs and offer alternatives as non-phthalates and non-ethers − key needs in the industry. Although this evaluation was not designed to serve as a comprehensive screen for low-VOC plasticizers/coalescents versus alternative choices, it did yield valuable insights into the effectiveness of low-VOC plasticizers functioning as coalescents in OPVs. The study clearly demonstrated that both dibenzoates and the monobenzoate are suited for graphic arts applications, in key parameters evaluated such as viscosity response, MFFT suppression, dry time, water resistance, gloss and hardness. Specifically, the K-Flex 975P, K-Flex 850S and X-250 dibenzoate blends are better suited than high-VOC ethers in OPV, as shown by the performance data in this article. The evaluation further demonstrated that the new monobenzoate X-613 is also an excellent lower-VOC plasticizer/coalescent.
References
1 de Wit, Jos, “Additives Improves Over-Print Varnish Coatings for Graphic Arts”, Paint & Coatings Industry, June 2011, Pages 36-38.
2 Arendt, W.D.; Strepka, A.; Lavelle, J.; Gallagher, J. “New Coalescent for the Graphic Arts Industry”, Proceedings of the 45th Annual Technical Conference, National Printing Ink Research Institute, October 17-19, 2001.
3 Arendt, W.D.; McBride, E. “New Dibenzoate Plasticizer/Coalescent for Low-VOC Coating Formulations”, Proceedings of the Thirty-Eighth Annual International Waterborne, High-Solids, and Powder Coatings Symposium, February 28 - March 4, 2011, Pages 419-429.
4 Arendt, W.D.; McBride, E. “New Dibenzoate Plasticizer/Coalescent for Low-VOC Coating Formulations”, Proceedings of the Thirty-Ninth Annual International Waterborne, High-Solids, and Powder Coatings Symposium, February 13 - 17, 2012 , Pages 442-460.
5 Arendt, W.D.; McBride, E.L. “New Dibenzoate Plasticizer/Coalescent Blend for Low-VOC Coating Formulations”, Journal of Coatings Technology, August 2011, Pages 26-33.
6 Arendt, W.D.; McBride, E.L. “Searching for a Better Dibenzoate Solution”, Adhesives & Sealants Industry, Vol 18, Issue 1, January 2011, Pages 23-27.
Disclaimer:
The information contained herein is believed to be reliable, however is based upon laboratory work with small-scale equipment and does not necessarily indicate end-product performance. Because of variations in methods, conditions and equipment used commercially in processing these materials, Emerald makes no representations, warranties or guarantees, express or implied, as to the suitability of the products for particular applications, including those disclosed, or the results to be obtained. Full-scale testing and end-product performance are the responsibility of the user. Emerald Performance Materials shall not be liable for and the customer assumes all risk and liability for use and handling of any materials beyond Emerald’s direct control. Nothing contained herein is to be considered as permission, recommendation nor as inducement to practice any patented invention without permission of the patent owner.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






