The Path to New Zinc-Free Anti-Corrosive Pigments
Identifying Specific Synergies Using Electrochemical Corrosion Investigations





The development of anti-corrosive pigments for the most diverse coating systems is extremely time consuming and expensive, owing to the necessary climatic testing, such as testing under salt spray exposure. To accelerate work on a new zinc-free pigment with improved protection, modern, electrochemical investigation methods have been used successfully and verified using traditional tests.1
Conventional Anti-Corrosive Pigments
The use of suitable anti-corrosive pigments has a significant influence on the protective properties of formulations for metal substrates (Figure 1). The mode of action of an anti-corrosive pigment is attributed to the following factors:2,3
- Increase in film strength;
- Prevention of rust creepage and underfilm corrosion at defect areas;
- General retardation of the corrosion process; and
- Cathodic and/or anodic passivation of the metal surface.
In the case of zinc phosphates, a very low water solubility results in the release of secondary phosphate ions to the coating, which are held responsible for the formation of inhibiting adhesive complexes on the metal surface and the associated anodic passivation. Another theory on the mechanism of action also describes the formation of tribasic ferric phosphate complexes, though only in weakly acidic media.4,5 As an ampholyte, zinc, or after hydrolysis zinc hydroxide, also demonstrates solubility behavior in acidic and alkaline media that is advantageous for corrosion protection. The modified zinc orthophosphate pigments and zinc polyphosphate pigments that are established on the market display a substantially increased chemical and electrochemical effectiveness as compared with traditional zinc phosphate, which enables very good protective properties to be achieved. In addition to aluminum, molybdate and organically modified types (ZPA, ZMP, ZPO), the universal WSA pigments ZCP PLUS and ZAM PLUS are of particular note.
Besides economic considerations, ecological and regulatory factors play an increasingly decisive role in the formulation of innovative coating systems. It is therefore no surprise that the call for zinc-free anti-corrosive pigments, or those that do not require labeling, has steadily increased in recent years. Zinc-free technology is not new; numerous pigments based on calcium, strontium, aluminum and magnesium phosphate have been available on the market for a long time. However, the real challenge is that it is only possible in rare cases to combine universal application with very good corrosion protection, as is the case with modified zinc phosphates. Although there are other causes, this is due primarily to the difference in solubility characteristics, as compared to zinc phosphate, of the corresponding compounds.
Even though the periodic table offers several alternatives to zinc that do not contain heavy metals, only a few metals qualify as suitable countercations. When making a selection, the emphasis is therefore on possible positive interactions between calcium and magnesium phosphate compounds. The requirements of the new zinc-free pigment technology were defined as follows:
- Highly effective anodic corrosion protection in solvent- and water-based systems;
- Stability and universal application;
- Very good dispersion properties;
- Cost efficiency.
Even at the early stage of the investigations, exceptional increases in performance were observed when using newly developed pigments with a different magnesium-calcium ratio, which also had a positive effect on the salt spray resistance.
Electrochemical Investigations
After extensive preliminary investigations, test conditions could be determined for two electrochemical methods – first, the rest potential analysis, and second, the electrochemical noise analysis – which enable comparative statements to be made on the protective effect of anti-corrosive pigments dispersed in an aqueous binder. For all electrochemical investigations, a round bar of unalloyed steel (C55, material no.: 1.1203) was used as a sensor or working electrode. As the electrolyte, an aqueous, organic coating dispersion was used, which was produced by dispersing the binder and the respective anti-corrosive pigment using a dissolver. As the organic coating dispersions were in fact aqueous but highly viscous, they were diluted in a 50:50 ratio with deionized water.
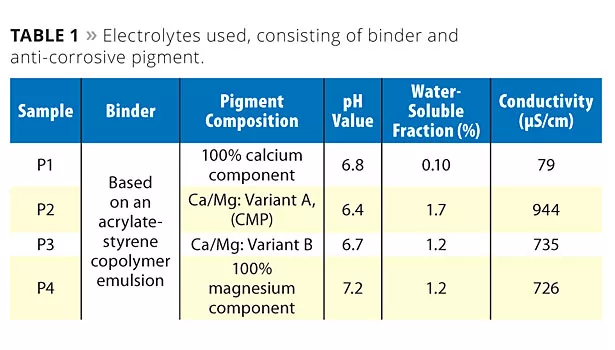
Table 1 shows the electrolytes used, consisting of an aqueous binder and four different anti-corrosive pigments or pigment combinations with a different Ca/Mg ratio. Variant B displayed a higher magnesium content than variant A. It shows that pigment P2 (CMP) displayed the lowest pH value at the greatest solubility and conductivity. This makes it difficult to predict the actual effectiveness of this pigment combination in real coating systems. But it is a first indication of changed electrochemical behavior.
Electrochemical Rest Potential Analysis
The rest potential analysis (RPA) is based on a rest potential measurement using a two-electrode arrangement, whereby a Ag/AgCl electrode was used as a reference electrode and the unalloyed steel C55 as a working electrode. The aqueous coating dispersion (Table 1) was used as the electrolyte, which was agitated during the entire measurement with a magnetic stirrer to prevent pigment settling. At defined time intervals, a 1M sodium chloride solution as a corrosion stimulator was added to the electrolyte by means of computerized pump arrangement. As a key measurement parameter, the potential curve was observed and evaluated by determining a critical quantity of chloride at which a significant potential drop occurred. For all measurements, the potential curve was first recorded over 60 min without the addition of chloride.
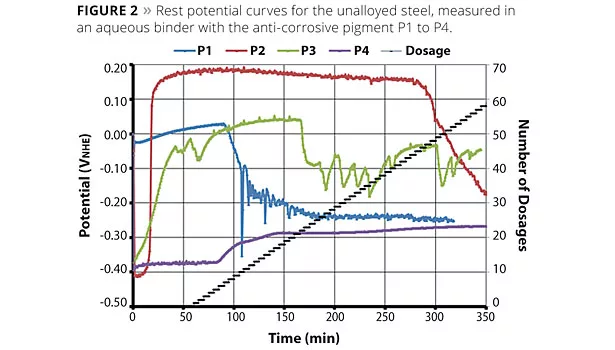
As a characteristic example of the rest potential analysis, in Figure 2 a rest potential curve is shown for each anti-corrosive pigment, P1 to P4. Using the potential curves, differences in the behavior of the pigments can be observed. For P1 to P3 a marked potential drop can be seen as a result of adding a defined chloride quantity. For P4 it is noted that the rest potential arose right at the start of the investigation at -400 mV. At this potential there was a strong metal dissolution, which was further accelerated after 60 min as a result of the chloride being added. The slight rise in potential to about -250 mV is attributed to the corrosion or secondary products on the metal surface and is not a result of inhibiting mechanisms.
Pigments P2 (CMP) and P3 are pigment combinations of pigment P1 and P4 in a different ratio. The pigment combinations first had the characteristic initial potential drop at about -400 mV, and then a potential increase depending on the quantity of pigment P4 added. The greater the quantity of pigment P4 (100% magnesium component) added to the pigment combination, the longer the metal needed to develop a passive-like surface state, and the longer therefore it took for the potential rise to occur.
Figure 3 illustrates the critical chloride quantities identified at the characteristic potential drop for the anti-corrosive pigments P1 to P4 dispersed in the binder. The mean value is shown in each case, calculated using a minimum of three individual measurements. Pigment P2 (CMP) displayed the highest critical concentration value, pigment P1 and particularly P4 the lowest.
Electrochemical Noise Analysis
Electrochemical noise analysis (ECN) is a very sensitive method for recording local corrosion processes and material changes, and has been successfully used for many years for various applications,6 including coatings testing.7 The measurements to investigate the dissolution behavior of the unalloyed steel in aqueous coating dispersions were performed without external current, using a three-electrode arrangement. For this purpose, two macroscopically identical working electrodes made from C55 were short-circuited via a zero-resistance ammeter and connected to a high-ohm potentiometer and a Ag/AgCl reference electrode. The measured noise signals were filtered by means of a band pass. This enabled them to be separated from the steady-state components (current and potential) and they could be amplified separately. After a test period of 20 min without chloride addition, 0.04 mL of a 1M sodium chloride solution was added to the electrolyte at 5-min intervals. The potential and potential noise as well as the current noise between the steel electrodes were measured and evaluated. Using the calculation of noise charge quantities and noise resistances, which were established in extensive preliminary investigations as characteristic values for the protective effect of the pigments, an even finer differentiation of the results could be achieved, as the charge quantities show a direct relationship to the pigment effect.
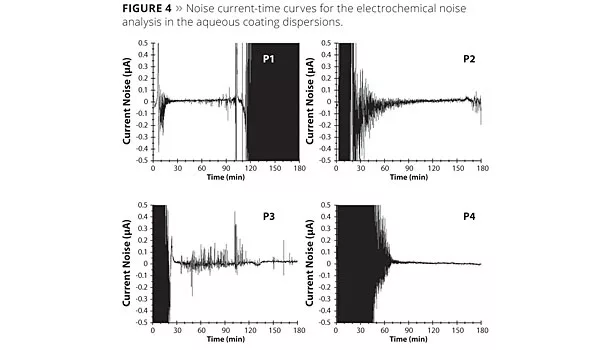
Figure 4 shows the noise current-time curves over the entire test period of 180 min for the unalloyed steel in aqueous binder with the anti-corrosive pigments P1 to P4.
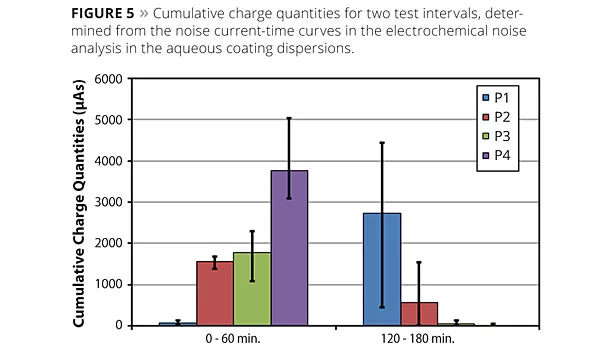
To illustrate the relationships and processes taking place at the metal surface, Figure 5 presents the cumulative charge quantities determined from the noise current-time curves.
Clear differences are shown between the various pigments. While P1 at the start of the measurement had low noise activity and, thereby, low metal dissolution, a strong initial activity could be identified in the first test interval for pigments P2, P3 and P4 due to the magnesium component. In the test period from 120 to 180 min, the noise activities of the pigment combinations P2 and P3 decreased greatly, while for P1 there was an increased dissolution. In fact, pigment P4 showed a sharp drop in the noise current and cumulative charge quantity, yet active metal dissolution continued, which could be evidenced by the detected noise resistances.
Verification Using Traditional Testing
To verify the evidence of the electrochemical investigation methods using conventional corrosion testing, cold-rolled steel sheets were coated with a formulation that is used in practice, also based on an aqueous styrene acrylate with the anti-corrosive pigments P1 to P4. The results after 408 hrs of aging in the salt spray test (DIN EN ISO 9227) are presented in Figure 6. CMP is HEUCOPHOS® CMP (calcium magnesium orthophosphate) produced by Heubach GmbH, Langelsheim, Germany. The combination of the calcium and magnesium component (P2) in the anti-corrosive pigment resulted in a significant increase in the corrosion protection.
To investigate the performance properties of pigment combination P2 (CMP) in other binder systems, salt spray tests were performed with formulations based on a solventborne short-oil alkyd resin (Figure 7) and a solventborne epoxy resin (Figure 8). For comparison, the pigment combination was tested against a zero sample without anti-corrosive pigment, magnesium phosphate, calcium phosphate and a reference sample containing zinc (zinc benchmark). The dry coating thickness in each case was 70 µm. To evaluate the degree of rust and rust creepage at the cross-section, the lower half of the coating was always removed after the salt spray exposure test.
The result of the salt spray exposure demonstrates the good performance properties when using CMP compared with magnesium phosphate. Even the protective effect of the reference sample that contained zinc could be outperformed using CMP. Magnesium phosphate exhibited serious problems regarding adhesion, rusting and blistering.
A significant improvement in adhesion and rust creepage at the cross-section could also be achieved in this system by using CMP.
Summary
A new, highly effective zinc-free pigment was developed. The investigations using rest potential analysis and electrochemical noise analysis show differences in terms of the pigment composition and its effect on the substrate dissolution. The amount of the magnesium proportion in the tested pigment variants P1-P4 had a decisive influence on the anti-corrosion performance of the overall system. Using the rest potential analysis, it can be clearly shown that an increasing quantity of the magnesium component in the anti-corrosive pigment causes the critical concentration value, which gives an indication of the anti-corrosion effect, to drop. A concentration of 100% magnesium component in the anti-corrosive pigment results in active metal dissolution and no anti-corrosive properties. On the basis of this investigation, P2 (CMP) was determined as an optimal pigment for this system with a defined Ca/Mg ratio.
With regard to electrochemical noise, first a trend is identified that the initial noise activity in the time interval from 0 to 60 min increases with increasing concentration of the magnesium component. This behavior points towards increased substrate dissolution in the first 60 min of the measurement at increasing magnesium concentration. After an extended test period and the addition of chloride, the pigment combination P2 displayed the best anti-corrosive properties. Pigment P4 with 100% magnesium component does in fact show a reduction in the noise activity. By including the determined noise resistances it could, however, be demonstrated that increased metal dissolution still takes place.
Results at a Glance
- The identification and use of synergetic interactions is of benefit when developing novel, highly effective anticorrosive pigments.
- The results of electrochemical investigations supported the choice of suitable synergetic components and considerably reduced the time it takes to carry out extensive preliminary investigations.
- The use of modern electrochemical investigation methods such as rest potential analysis and ECN was successful.
- The good result for the new pigment CMP in the electrochemical investigations could be confirmed using conventional corrosion testing.
References
1 Bender, S.; Babutzka, M.; Kirmaier, L. Moderne elektrochemische Korrosionsuntersuchungen gezielt eingesetzt, Farbe und Lack, (2014).
2 Kirmaier, L. Farbe und Lack, (2009), 115, p. 120-123.
3 Vogelsang, J. Basics about Anticorrosive pigments and Corrosion Inhibitors and Possibilities for their Usage, European Coatings Conference, Berlin, 2000.
4 Ruf, J. Organischer Metallschutz, Vincentz Verlag, Hannover, 1993, 260.
5 Hao, Y.; Liu, F.; Han, E.H.; Anjum, S.; Xu, G. Corrosion Science, 2013, 69, p. 77-86.
6 Heyn, A.; Göllner, J. Analysis and Monitoring of Corrosion using Electrochemical Noise - 5(th) Part, Materials and Corrosion (Vol. 64), No. 8, 2013, p. 663.
7 Plagemann, P.; Yezerska, O.; Brinkmann, A. [2] L. Kirmaier. Farbe und Lack, (2009), 115, p. 94-97
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






