A 'Two-For-One' Biocide For Latex Coatings Provides Effective In-Can and Dry Film Preservation


In the early 1970s, the most popular biocides for paints were based on phenyl mercury acetate because they had the appealing ability to serve in the dual capacity of in-can preservative and dry film mildewcide.6-8 However, the toxicity and environmental concerns of mercury-based biocides resulted in the paint industry discontinuing their use in the United States in the 1980s. They were replaced by biocides based on alternative chemistries, such as 3-iodo-2-propynyl butyl carbamate (IPBC) and 2-n-octyl-4-isothiazolin-3-one (OIT).9-10 Highly effective against fungi, the new chemistries proved to be very capable mildewcides.
These non-mercurial agents, however, did not protect against the bacteria that cause spoilage during storage. As a result, manufacturers were compelled to use preservatives to protect paints in the can. Among the products employed for this purpose were formaldehyde releasers and a number of isothiazolinone compounds introduced in the early 1980s. Although these biocides performed well as in-can preservatives, they were not effective as dry film mildewcides. Consequently, for the past 20 years, paint manufacturers have been compelled to use a combination of biocides for complete antimicrobial protection.
Dual-purpose ROZONE 2000 controls a wide range of microorganisms, including fungi, algae and bacteria. As far as fungi are concerned, this isothiazolinone product inhibits the growth of many species at low use levels. Its efficacy against Aureobasidium pullulans (the fungus predominantly responsible for the development of mildew in exterior paints) is particularly noteworthy. Equally important from the stand point of in-can protection, this biocide also is very effective against the bacteria, especially the Pseudomonas species, that attack paints, joint compounds, roof coatings, exterior insulating and finishing systems (EIFS), and clear finishes in the can.
In addition to its impressive antimicrobial capabilities, ROZONE 2000 has a number of other appealing features for formulators. Because of its isothiazolinone chemistry, it is non-persistent in the environment, breaking down readily into harmless compounds. In addition, this biocide does not release formaldehyde, does not require zinc oxide for mildewcide stability and is easily incorporated into paint and coatings formulations. The new biocide can be used in both latex- and solventborne paints. With all its performance benefits, ROZONE 2000 exacts no appearance or performance penalty. This compound will not cause a paint film to yellow, will not destabilize a paint or detract from its tint retention and is EPA-registered for use in coatings.

Stringent Testing
The performance benefits of ROZONE 2000 were supported by stringent Rohm and Haas Company tests that demonstrated the ability of this biocide to function in the dual role as an in-can preservative and mildewcide. A number of studies had already documented its ability to control the growth of numerous fungi and algae. Rohm and Haas scientists confirmed these findings and also established the antibacterial efficacy through a series of different tests of ROZONE 2000 containing 20% DCOIT. The procedures used to evaluate ROZONE 2000 included minimum inhibitory concentration (MIC) determinations, in-can preservative efficacy tests and dry film mildewcide tests.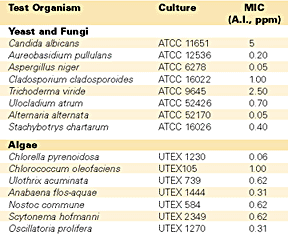
MIC Method and Results
The following procedure was used to establish MIC values. First, the test organisms were cultivated in three different media: nutrient broth for bacteria, potato dextrose for fungi and yeast, and Allen's medium for algae. In each case, the microorganisms were introduced to the medium and incubated at 25 deg C for seven days. An aqueous suspension of each test organism was then prepared by harvesting the growth from the medium in a test tube containing an aqueous solution of 50 ppm of Triton(tm) X-100 surfactant. The number of microorganisms in the suspensions was assessed with a hemocytometer and reported as colony-forming units (CFU) per milliliter.
The results of the testing clearly demonstrated the broad-spectrum antimicrobial efficacy of DCOIT. The MIC values of DCOIT against all organisms tested were under 20 ppm. As previous testing had shown, DCOIT was extremely effective against fungi and algae in the MIC tests, with values of generally less than 1 ppm (see Table 1). Equally impressive, DCOIT also provided efficient control of many bacterial species; MIC values for these microorganisms ranged between 2 and 20 ppm inclusive (see Table 2).


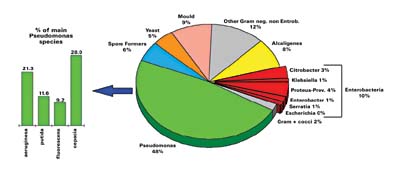
Collectively, approximately 60 to 70 percent of the bacteria identified were gram-negative (see Figures 2 and 3). The great majority of these, approximately 45% to 50% of the total contaminants identified, are Pseudomonas sp., with P. cepacia, P. putida and P. flourescens predominating. A number of other species, such as Alcaligenes sp., Citrobacter sp., Enterobacter sp., and Klebsiella sp., accounted for the remaining gram-negative species identified. It is these gram-negative bacteria, Pseudomonas in particular, that ROZONE 2000 controls so effectively.
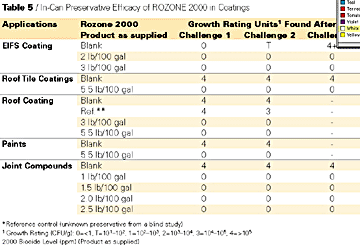
In-Can Challenge Test Results
To ascertain the efficacy of ROZONE 2000 for in-can preservation, the challenge method based on the ASTM Test Method D-2754 was followed.12 The investigators inoculated various paint and coating formulations with a mixture of freshly harvested Pseudomonas species consisting of P. aeruginosa, P. cepacia, and P. putida. All of these bacterial species were isolated from commercial paint and coating formulations. At various intervals after inoculating the paints and coatings (1, 3 and 7 days), the investigators analyzed samples of the test formulations for the presence of the Pseudomonas species.
The efficacy of ROZONE 2000 for in-can preservation was demonstrated in a wide variety of coating formulations, including tile coatings, roof coatings, EIFS coatings, joint compounds and paints (see Table 5). At no point in the testing of the samples - even in those containing the lowest level (1lb/100 gal) of ROZONE 2000 - were investigators able to detect any of the bacteria that had been introduced into them.


Dry Paint Mildewcide Performance
The ability of ROZONE 2000 to control the growth of mildew was evaluated per ASTM Test Method D-5590.13 Two samples each of paints containing biocide were brushed onto different Leneta charts and allowed to dry in open air for 48 to 72 hours. One of the two charts was then exposed to running water for 24 hours to simulate the leaching effect of weathering. One-inch squares were cut from each chart, placed on malt agar plates and individually swabbed with a fungal inoculum comprised of Alternaria sp., Penicillium sp., and Aureobasidium pullulans, three species commonly implicated in mildew. The treated squares were then incubated at 28 deg C for the remainder of the study. The plates were observed weekly for surface disfigurement due to mildew growth.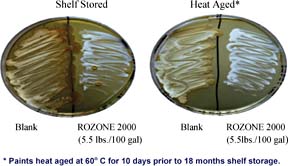

A Cost-Effective Option To Biocide Packages
ROZONE 2000 affords paint manufacturers a very effective option to conventional biocide packages for their formulations. For decades, manufacturers had to employ separate biocides to preserve paint in the can and prevent mildew growth on dry film because none of the available products had the broad-spectrum antimicrobial efficacy required to serve in both capacities. This microbicide, however, can meet both needs.
For more information on ROZONE 2000 Industrial Microbicide, contact David Sutton, Coatings Biocides Marketing Manager, Rohm and Haas Company, 100 Independence Mall West, Philadelphia, PA 19106-2399; phone 215/592.6855; fax 215/592.2178; e-mail davidsutton@rohmhaas.com; or visit RHCIS.com.
This paper was originally presented at the Federation of Societies for Coatings Technology's Annual Meeting Technical Program, ICE 2002 in New Orleans.
References
1 Buono F., W. J. Stewart, and M. Freifeld. 1973. Evaluation of latex preservatives. J. Paint Technol. 45: 43-54.2 Gillatt, J. 1992. Bacterial and fungal spoilage of waterborne formulations. J. Oil Colour Chem. Assoc. 10:387-392.
3 Gillatt, J. 1996. The use of biocides and fungicides in wood coatings and preservatives. Pigment and Resin Technology 25: 4-10.
4 Winters, H., G. Guidetti, and M. Goll. 1974. Growth of a typical paint bacterial isolate in aqueous emulsion paint. J. Paint Technol. 46: 69-72.
5 Wood, W. B. 1982. Prevention of microbial spoilage of latex paint. Journal of Waterborne Coatings 5(4): 2-4.
6 Environmental Protection Agency. 1972. Certain products containing mercury: cancellation of registration. Federal Register 37(61): 6419-6420.
7 Cadmus, E. L. 1974. Paint containing organic mercury compositions. Ventron Corporation (Canada): 1-14.
8 Machemer, W. E. 1979. Use of mercury in protective coatings. Dev. Ind. Microbiol. 20: 25-39.
9 Edwards, J. N., and W. A. Smith. 1974. Nonmercurial preservatives, their effectiveness and relation to raw materials in latex paints. J. Paint Technol. 46: 37-45.
10 Bergfeld, R. A., R. M. Hille, and R. E. Edwards. 1971. Efficacy of non-mercurial preservatives in aqueous coatings. J. Paint Technol. 43: 80-91.
11 Gastaldi, H. 2001. Overview of microbial contaminants found in latex and paint products from European Region. Rohm and Haas Technical Service Report # 01TC0037.
12 ASTM Test Method D 2574-97: Standard Test Method for Resistance of Emulsion Paints in the Container to Attack by Microorganisms.
13 ASTM Test Method D-5590-94: Standard Test Method for Determining the Resistance of Paint Films and Related Coatings to fungal Defacement by Accelerated Four-Week Agar Plate Assay.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




