Why Particle Sizing?
Controlling Product Quality
Particle size influences both the optical and bulk properties of pigment dispersions and therefore must be tightly controlled in order to deliver a product that meets customer requirements in terms of both finish and handling. The optical properties of the product are those that tend to define the finish achieved. These include:- Opacity;
- Tinting strength;
- Undertone;
- Gloss/film appearance; and
- Weather resistance.
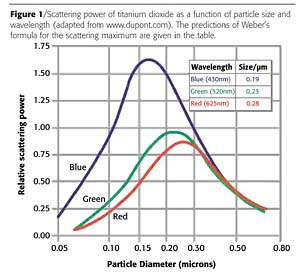
Optical Properties
Particle size directly affects the magnitude of light scattered by a pigment with a given dispersion. In turn, this scattering power determines the optical or visual properties of the finished film, and this is why particle size can be used so effectively to deliver specific product performance. The particle size at which the maximum scattering power is observed is related to the optical properties of the pigment and the mobile phase, as well as to the wavelength of the incident light, and can be predicted using Weber's formula:D = 2l/p(n1-n2)
Where D = optimum particle size for a high refractive index pigment.
l = wavelength of the light.
n1,2 = refractive indices of pigment and associated medium.
This formula shows that, in order to achieve the highest scattering power, the pigment particle size should be less than one micron (Figure 1), if not below 0.5 micron, although this does depend on whether high or low refractive index pigments are used. Maximizing the scattering power then allows other properties of the pigment dispersion to be controlled.
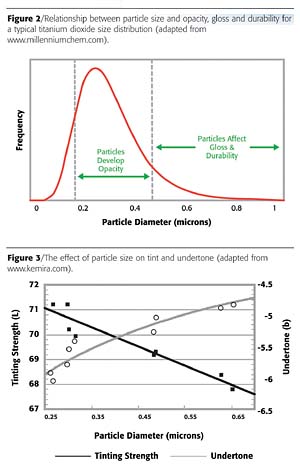
Opacity
The opacity, or hiding power, of a given pigment dispersion is a measure of its ability to completely mask the underlying substrate. As the particle size within the dispersion is decreased, the opacity will increase in relation to the scattering power (Figure 2). This relates to the increased probability that light entering the applied film will be reflected away from the substrate, effectively hiding it from view. A pigment with a fine particle size is therefore capable of producing a finished film whose opacity at lower concentrations is equivalent to that normally observed at much higher concentrations when using a coarser material. As pigment is frequently the most expensive element in a formulation, this effect can have significant cost implications.Tinting Strength and Undertone
Tinting strength and undertone are important properties within color mixing applications. The tinting strength defines the ability of a pigment to influence the color of a dispersion containing other pigments, whereas the undertone defines the hue obtained during tinting. Again, each relates to the scattering power of the pigment and, in the case of the undertone, how this varies as a function of wavelength.
The effect of particle size on the tinting strength and undertone is perhaps best shown for titanium dioxide (Figure 3). As the particle size decreases, the tinting strength improves. However, the undertone also changes, with a move towards a more-blue undertone being observed due to the increased scattering efficiency observed at the blue end of the spectrum for fine particles (as predicted by Weber's formula). A trade-off exists therefore between high tinting strength and an acceptable hue.
Gloss and Film Durability
The gloss and film resistance relate to the way that pigment particles pack within the applied coating film. Small particles with a narrow particle size distribution pack well at the film surface, yielding a uniform surface finish. This surface uniformity, in combination with the high scattering power associated with smaller particles, results in a high degree of gloss, a desirable outcome for many applications. Small particle size also improves the weathering characteristics of a coating since large particles or agglomerates can be more easily dislodged from the coating, resulting in the surface rapidly losing its gloss.Bulk Properties
The bulk properties of pigment dispersions are not only defined by the choice of mobile phase but also depend on the pigment particle size. Decreasing particle size enlarges the overall surface area for a pigment volume concentration, increasing the particle-particle interactions and the driving force for agglomeration. Thus, while achieving a fine particle size may be beneficial with regard to the optical properties of the film, it can cause problems with the functionality of the bulk dispersion.
Dispersion/Flocculation
A dramatic change in the dispersability of a given pigment will be observed as the particle size decreases. This is clearly shown in Figure 4, where the force of adhesion observed between particles is shown as a function of particle size.
For large particles, the adhesion force between particles is small and dispersion is easily achieved, provided that the pigment is completely wetted by the mobile phase. For particles below 20 microns in size, colloidal forces become important and the adhesion forces increase rapidly. The pigment dispersability therefore diminishes, and there is an increased likelihood of aggregate formation. In order to achieve the fine particle size required to yield the correct optical properties, it is therefore necessary to control the surface chemistry, either by applying pigment surface coatings or by changing the properties of the mobile phase. Inadequate dispersion will lead to aggregate formation, impacting the opacity and surface gloss and durability.
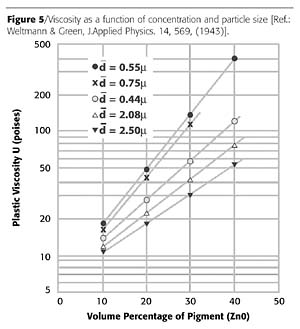
Viscosity
The rheological properties of the pigment suspension are important in defining how it behaves during storage and application. Often there is a requirement for the viscosity to be high when no shear is applied to the pigment system in order to ensure that settling does not occur during storage. However, this must be balanced against the requirements of the application process, where a lower viscosity may be required to ensure that a uniform film is applied. This requires the development of shear-thinning suspensions.
The particle size of the pigment is one factor that influences the rheological properties of a given pigment dispersion. As mentioned above, decreasing the size of the pigment will lead to an increase in the strength of any particle-particle interactions. As well as increasing the likelihood of agglomeration, this will also lead to an increase in the bulk viscosity, as shown in Figure 5. The change in viscosity with increasing pigment volume concentration will also become more pronounced. Again, control of the surface chemistry can allow the viscosity to be reduced.
Summary
It can be seen that the impact of particle size on pigment properties is both complex and far reaching. Particle size optimization and the control of any undesirable effects of selecting a given particle size are key steps in the development of effective and competitive products. Fully understanding the influence of particle size on both optical and bulk properties allows optimization to be carried out in a scientific and systematic manner.For more information, contact Alison Vines, Malvern Instruments Ltd., salesinfo@malvern.co.uk.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!



