A Versatile Core-Shell Nanoparticle
A Solution to Coating Challenges


An exception to this is the work of Dr. Janos Borbely, Head of the Department of Colloid and Environmental Chemistry of the University of Debrecen in eastern Hungary. Borbely, a polymer chemist, has created an innovative nanoscale polymer particle technology, developed over many years, which he believes will offer significant benefits, not only for the coatings industry but other areas as well.
After recruiting a small number of dedicated investors, ElizaNor Polymer was formed to continue the funding of Prof. Borbely’s research. The basis of his technology is core-shell macromolecular particle morphology. The polymer particle is nanoscale in size, is soluble in most organic solvents and is heavily crosslinked. Core-shell polymer technology has been known some 20 years, having been used for industrial applications in coatings, paper manufacturing and adhesives.
Borbely’s technology, however, allows a manipulation of the polymer particle, which, heretofore, has not been known. For example, by manipulating the shell functional groups, it becomes possible to design a particle that will adhere to metallic surfaces as is required of a primer coat or replace these functional groups with those that will provide a gloss for use as a clear coat. At the same time, hardness, which largely resides in the core, is relatively unaffected by the manipulations in the shell. Furthermore, it becomes possible to design a polymer particle that will not only lower the VOCs in solventborne coatings, but the technology also lends itself to the creation of a particle for powder coatings.

Nanoparticle for Solventborne Coatings
Borbely and his co-workers have succeeded in creating a nanoparticle soluble in butyl acetate that allows a solid content of 75% with very good to excellent performance characteristics as follows: adhesion: 5B, (ASTM D 3359); hardness: 260 after 1 hr at110 deg C, (Persoz, ASTM D 4366); flexibility: no fracture (conical mandrel, ASTM D 1737). In addition to, and despite the high solids content, a remarkably low viscosity has been achieved; viscosity: 0.5-0.7 Pa.s. (AR 550 Rheometer, TA Instruments); shear stress: 260-280 Pa at 300 s-1 shear rate.
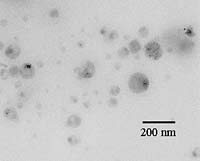
The low viscosity of Borbely’s preparations will result in positive performance characteristics. These effects appear to be intimately related to the unique nanoparticle properties, which, heretofore, have been largely unexplored. Because of the extensive crosslinking, the core-shell nanoparticle is globular in shape and the polymer chains possess covalent crosslinks that prevent free motion of the chains. This restricted motion results in the reduction of the viscosity. The nanoparticles have diameters of 30-150 nm, consisting of three basic size populations (Figure 2): small, ca. 30-50 nm; medium, ca. 60-90 nm and large, ca. 110-150 nm. Based on his studies, Borbely believes that the heterogeneous particle distribution allows good packaging and film formation and, in solution, excellent flow properties at high solids content. Although the molecular weight of the particle is approximately one million and greater, it behaves as though it were a much smaller molecule. Rheometric measurements have recorded viscosity as low as 0.7 Pa.s. at a solids content as high as 77% (Figure 3). Thus, despite the high solids content, coating by spraying could be readily accomplished without major modifications of equipment.
Polymer Nanoparticle for Powder Coatings
The versatility of Borbely’s core-shell polymer particle technology is clearly demonstrated by its ready adaptation to powder coatings (Figure 4). With this technology, significant reductions in both melting (120-130 deg C) and curing (170-175 deg C) temperatures have been achieved. Borbely believes that much lower temperatures can be achieved and that supplementation with crosslinking agents will allow even more dramatic temperature reductions. Stability studies have also demonstrated that the particles possess a lengthy shelf life.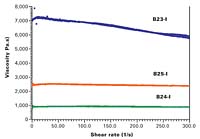
Future Research
Because of the experience gained through the design of core-shell polymer particles for coatings, and because of the pliable nature of the particle, Borbely and members of his staff are casting out into fields far removed from that of paint. For the future, they have successfully constructed a developing array of water-soluble core-shell polymer particles made of biodegradable molecules. ElizaNor Polymer will pursue the targeting of cancer cells with tumor-inhibiting compounds loaded into these engineered particles. This speaks to the versatility of this technology, capable of serving not only industry but medical science as well.For more information, contact John Hartmann, President, ElizaNor Polymer LLC, One Woodmeadow Lane, Princeton Junction, NJ 08550; phone 609.799.2812; fax 609.897.9660; or e-mail ElizaNor@aol.com.

Dr. Janos Borbely and ElizaNor Polymer
After receiving his Ph.D. in organic chemistry in 1980 Dr. Borbely was employed by Tisza Chemical Works of Hungary (the paint manufacturing segment has subsequently been acquired by Akzo Nobel). While there, he was involved in developing new acrylic polymers. He then became a faculty member at the University of Debrecen located in the city of the same name in eastern Hungary. He was awarded a Fullbright Fellowship in the early 90’s in the laboratory of Dr. David Tirrell who, at that time, was at the Polymer Institute of the University of Massachusetts, Amherst, and presently is chairman of the Department of Chemistry at Cal Tech. Borbely returned to Debrecen and resumed his position at the university, where he was promoted to his current status. Except for two years as Deputy Minister of Environmental Matters in the federal government in Budapest, Dr. Borbely has spent full-time developing his nanoparticle technology.ElizaNor Polymer, LLC, a company with headquarters in Princeton Junction, NJ, has funded Prof. Borbely’s work for almost three years, the fruits of which are now beginning to mature. The founder and president of the company, John Hartmann, Ph.D., a retired scientist from the Merck Research Laboratories in Rahway, NJ, became acquainted with Borbely in the late 90’s through Hartmann’s contacts with members of the Chemistry Institute at the University of Debrecen. Intrigued by his approach, Hartmann began supporting Borbely’s research.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





