Synthetic Resins for Wood Applications
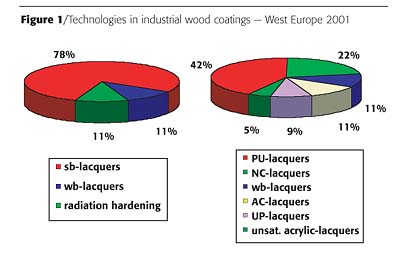
Even in highly developed and mature markets, such as Europe, solventborne wood coatings still account for 78% of all industrial wood coatings; the remaining part is equally shared by waterborne and UV-curing systems. The solventborne portion is fragmented into a number of different lacquer systems as outlined in Figure 1. As much as 6% of UV-curing systems are included in UP- and PU- lacquers.1

It is obvious from the graph that huge coating application sites, for instance automotive OEM plants, were the first to implement VOC-reduction measures. Consequently, their proportion is relatively small. For the small- to medium-size industry and the DIY sector, where the application is widespread, an improvement is only achievable if the coating technology - namely the resin system as a prime source of emissions - is changed to environmentally friendly systems. This presents a challenge to resin suppliers to serve the markets with tailor-made value propositions to meet different market demands.
Industrial Wood Coatings
A major part of solventborne industrial wood coatings is based on PU- and NC-lacquers. In terms of technical performance both systems can be formulated to exhibit:- rapid physical drying speed;
- blocking resistance;
- good appearance;
- easy handling;
- excellent wetting;
- good ratio between price and achievement.
In Asia Pacific, the formulation of a wood sealer creates a challenge to resin suppliers since often so-called oily wood types are in use. Wood ingredients tend to migrate to the top surface of the coating, causing discoloration and yellowing. Acidic, acrylic dispersions have been used in the past to prevent this kind of migration.
Current developments have led to a new waterborne, urea-modified amine-epoxy adduct that contains cationic stabilization. Applied as a clearcoat, it penetrates into the wood surface and shows excellent wetting and fast drying speed. Lab tests confirmed a superior prevention of the migration. One might speculate whether the cationic charges on this polymer enhance the affinity of the polymer to the cellulose fibers in the substrate, as this is known from cationic-stabilized methylolmelamines in the paper and fabric industry.
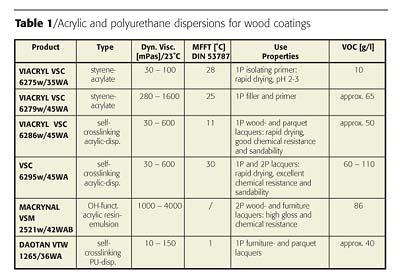
Special acrylic dispersions with low co-solvent content, solvent-free and with hydroxyl functional groups have been developed to formulate one- and two-component air-drying (and force drying) coatings for primer-, filler-, top- and clearcoats, as depicted in Table 1.
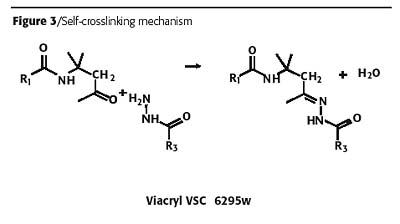
The self-crosslinking mechanism is based on an inter- and intra-molecular condensation reaction of a carbonyl compound with nucleophilic amines, as outlined in Figure 3. Water exists in large excess as supplied with the binder, consequently reaction proceeds onward only during and after the physical drying process.
This additional crosslinking process remarkably improves the film hardness and chemical resistance. Further enhancement can be accomplished by means of isocyanate hardeners. Formulations based on such kind of vehicles enable paint formulators to meet the chemical resistance requirements of DIN 68861 that are standard for the furniture industry.
Alternatively, polyurethane dispersions have been developed for air-drying and self-crosslinking 1K formulations that are able to provide additional flexibility and toughness to the film if applied as the sole binder or in a blend with acrylic dispersions.
It is common to blend PU dispersions with an acrylic dispersion in a ratio of 3:7, calculated on solids, to combine the positive aspects of both binder classes and to meet economical needs. A new development resulted in polyurethane-acrylic hybrids with built-in acrylic modification. These dispersions are suitable for 1K and 2K formulations and provide superior drying speed and hardness.
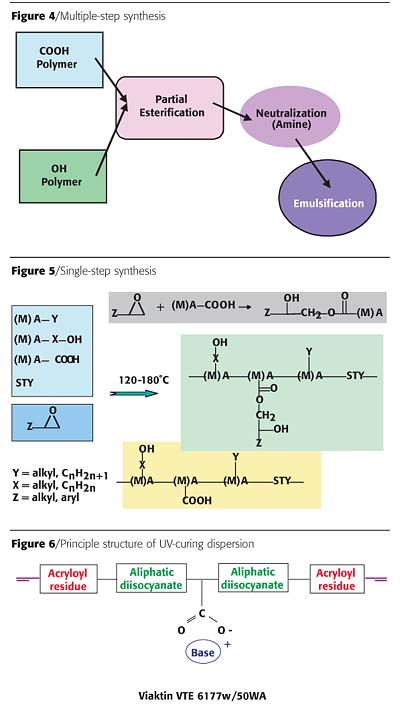
Surface Specialties spent considerable effort in optimizing the molecular design of correspondent acrylic polyols for this application.3 Initial developments were made according to the established process for the synthesis of alkyd emulsions in a multi-step process described in Figure 4.
In the first step, two independent intermediates with either an excess in hydroxyl or carboxyl functionality are synthesized, which are brought together in a controlled co-condensation step resulting in a carboxyl-functional pre-polymer. This pre-polymer is soluble into water after a neutralization step.
The advantage of this multi-step synthesis lies in a huge variety of options for the individual intermediates to trigger manifold performances. Commercialized products gained by this process sequence still showed some technical shortcomings in view of film hardness, application aspects and chemical resistance.
A new single-step synthesis is displayed in Figure 5. According to this concept a tailor-made polymer design is possible, and a number of properties were improved in the coatings, including:
- Scratch resistance;
- Chemical resistance;
- Film hardness;
- Pinholing (suitable to be applied in higher film thickness);
- Sagging (on vertical surfaces);
- Volatile organic compound - (VOC) - emission.
Consequently, a newer development trend is moving towards 100% and waterborne systems. Typically, in a UV-curing dispersion the molecular weight of the polymer itself can be altered more or less freely without impact on the viscosity. Therefore a higher level of chemical resistance is achievable that can be superior to their solventborne counterparts. An additional advantage is that these dispersions can be blended with compatible acrylic or epoxy dispersions and alkyd emulsions to enhance specific technical properties beyond the usual level. The principle structure of these dispersions is displayed in Figure 6.4
Since Surface Specialties has long-term experience in waterborne technology, focus was put on the development of dispersions that meet the customer's expectation in view of storage stability, because that was often a problem.
It should be noted that the water must be removed completely from the film before the UV-curing process can be conducted. The UV-curing line has to be modified accordingly. Another important aspect is that overspray is collectable and can be recycled by means of an ultrafiltration process.
An alternative approach is the use of UV-curing or ultra-low-curing powder coatings.5 This technology provides the lowest impact on the environment and reclaim of overspray, thus making it possible to establish nearly a 100% utilization of the coating material.
Powder coatings, after application, can be melted by IR burners that provide energy instantly to the polymeric material to minimize the temperature impact at the interface of the wood substrate and the organic layer. This prevents the emission of humidity. After a certain relaxation time for the flow-out of the powder, it will be cured in a few seconds by UV radiation. Although it seems difficult to match the technical requirements for the various substrate types, this technology will certainly capture a remarkable share of the industrial wood coatings segment in the future.
DIY Segment
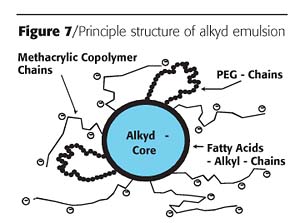
The DIY segment accounts for about 15% of the total solvent emissions in Europe; another 15% will originate from outdoor coating applications. In addition, safety and health considerations for applicators and end users encourages resin supplier R&D departments to seek environmentally friendly and safe solutions and binder systems.The focus on replacing solventborne alkyds with waterborne counterparts goes back decades and has resulted in the development of special core-shell alkyd emulsions under the trade name of Resydrol.6 These emulsions are produced according to the multi-step synthesis described for the acrylic dispersion previously. The principle structure of the alkyd emulsion is shown in Figure 7.
Combining the alkyd polymer with an acrylic modification to get optimum stability and drying speed is successful. In this self-emulsifying alkyd emulsion, the hydrophobic core is formed by the alkyd part surrounded by a more hydrophilic, non-saponifiable acrylic- or co-polymer shell.
The outer shell interacts with pigments, extenders and driers, and determines the rheology of the emulsion. Needless to say, adhesion to substrates is influenced to a great part by the acrylic modification. The idea of core-shell emulsions carrying alkyd segments in the inner part and acrylic segments in the outer part contributes to better understanding of the role of the alkyd segment with regard to film formation, drying properties, reticulation and film resistance.
Long-term experience and scientific investigations have proved that the penetration of such emulsions into the wooden surface structure is clearly superior and much more intense compared with acrylic emulsions. The inner part allows manifold modifications, for instance by isocyanates to improve film hardness and corrosion and water resistance; by epoxies to improve flexibility, water and corrosion resistance; and by grafted acrylic monomers to improve further drying properties and compatibility.
Self-emulsifying alkyd emulsions show remarkable resistance against shear stress and are suitable for direct incorporation of pigments and extenders. In outdoor exposures their performance does not differ from solventborne counterparts.
Special hybrid systems have also been developed to combine positive properties of typical alkyd resins with those of acrylic dispersions, resulting in faster physical drying speed and lower yellowing tendency. Since paints formulated on the basis of these Resydrol types can be applied by spray and brush, they represent the best choice for the DIY sector.
There have been public discussions about whether the development of waterborne systems simply shifts the problems of air pollution to water pollution. Consequently, the degradation of these binders has been studied at an early stage and, in so-called Zahn-Wellens tests, it was demonstrated that these polymers disintegrate within three to five days in nature.
It should be kept in mind that waterborne systems mainly consist of anionic-stabilized polymers or, in other words, are based on polyelectrolytes. Their functional groups can interact with a variety of metal ions, and complexes formed by these ions can destabilize the formulations. The use of de-ionized water is compulsory. Furthermore, these polymers are surface active, and water, as a solvent, performs different than organic solvents. Coating preparation, handling and application is different from conventional systems, and a learning period is needed.
Additives
It should not be overlooked that waterborne formulations require additives to develop the real potential of these binders. Defoamers, wetting and leveling agents are substantial parts of all formulations. Surface Specialties offers a full package of additives under the trade name of Additol and Modaflow to cope with all possible problems. A wrong choice in this part of the formulation can result in detrimental performances.
Moisture-Curing Systems
Due to the reaction mechanism - the chemical reaction of isocyanate groups with water/humidity coming from the ambient atmosphere - we do not expect to see a waterborne version in the future. These coatings are still quite popular because they combine rapid curing, radiant brilliance, superior abrasion resistance and excellent adhesion.The greatest concern, in view of EHS aspects, is due to the free isocyanate content. Apart from this, the content of aromatic solvents, namely xylene, was a major target for development as well.
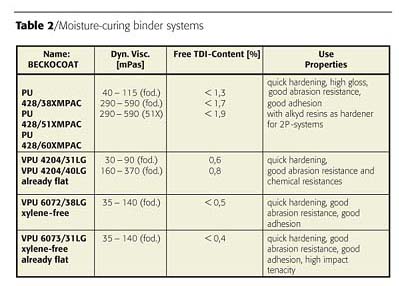
An overview of available products and correspondent-free isocyanate content is given in Table 2.
A comparison of different binder systems for parquet coatings is depicted in Figure 8.
Acid-Curing Systems
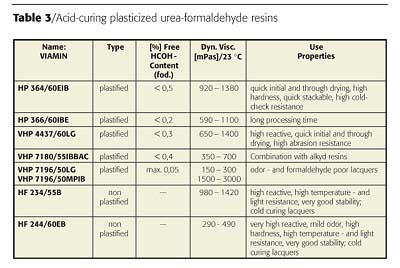
Acid-curing wood coatings are based on combinations of special alkyd resins with non-plasticized urea or melamine formaldehyde resins. This class represents the fastest drying coating type. The chemical reaction initiated by strong acid in high concentration as hardeners proceeds through a reaction cascade where it is difficult to control the amount of co-condensation between alkyd and amino resin and the self-condensation of the amino resin itself. As the name "urea-/ melamine formaldehyde resin" implies, the major concern today is based on a split off of free formaldehyde because these coatings are typically used in indoor applications.It has been shown that the development of waterborne systems is not useful, because the amino resin will undergo a rapid hydrolysis process, and under the acidic conditions release even higher amounts of free formaldehyde than solventborne systems. The technical performance and formaldehyde release can be optimized if the ratio of pure co-condensation could be increased over the self-condensation. This is achieved by the development of plasticized urea resins where the co-condensation occurs during the resin synthesis process. An overview of such plasticized urea formaldehyde resins for AC wood coatings is given in Table 3.
Polymers can be designed so that the content of free formaldehyde, as supplied, is as low as possible, but for practical purposes it is more relevant to focus on the actual figures of formaldehyde release. It is common to use the so-called chamber test where the formaldehyde emission is monitored by gas chromatography.
Release of free formaldehyde from films after different drying periods can be reduced and controlled below limits set by industrial standards.
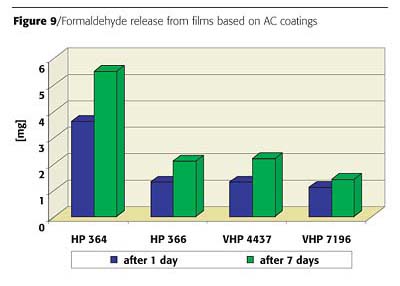
Measured results of accumulated formaldehyde emissions are exemplified by Figure 9.1 In general, one-third of the total formaldehyde emission is released immediately during the application; another 30% will be emitted from the film during the first 24 hours.
Summary
Wood coatings still represent an exciting and attractive field of coating application. Current and future developments are focused on environmentally friendly and safer coating systems for the paint manufacturer, applicator and end user. A huge variety of binder systems are offered to the market to support paint manufacturers in this task without sacrificing technical performance. This trend will continue.References
1 Neumayer, S. Internal communication.
2 Liebscher, H. "VOC-Verordnung in Deutschland und Oesterreich", Presentation, Graz, 2002.
3 Billiani, J.; Wilfinger, W. New Low-VOC Acrylic Polyol Dispersions for Two-Component Polyurethane Coatings. Surface Coatings Int., Part B., Coat. Trans. Vol 85, 169-242, 2002.
4 Gerlitz, M.; Awad, R. New Generation of Waterborne UV-Curable Resins. Presented at Int. Waterborne, High Solids & Powder Symposium, New Orleans, 2002.
5 Souris, Y.; Buysens, K. New UV Powder Systems for Metal, Wood and PVC. PCE, 2002.
6 Gobec, M. SLF Congress, Helsinki, 2000.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






