The Future of UV Coatings is Now!
Can you think of any organization that has had 35 consecutive years of sales growth? A few come to mind that may fit that category, but you can count them on one hand. Statistics from past RadTech International shows indicate that the UV/EB formulated product growth for the North American market is just over 7% annually.
Fueling this growth is not just the push for “green” technology and increased productivity required in manufacturing, but also the availability of new raw materials from suppliers. These new materials allow formulators the ability to produce UV coatings with improved physical properties and better product performance, which opens up new capabilities for users.

Can you think of any organization that has had 35 consecutive years of sales growth? A few come to mind that may fit that category, but you can count them on one hand. Statistics from past RadTech International shows indicate that the UV/EB formulated product growth for the North American market is just over 7% annually.
Fueling this growth is not just the push for “green” technology and increased productivity required in manufacturing, but also the availability of new raw materials from suppliers. These new materials allow formulators the ability to produce UV coatings with improved physical properties and better product performance, which opens up new capabilities for users.
Two factors are primarily driving the growth of UV-cured coatings.
Resin
The resin can be thought of as the “film former,” which binds all the ingredients into a continuous system and forms a film when dried. Higher-molecular-weight synthetic resins are more generally referred to as polymers. Polymers are usually highly viscous, and solvent (or water) is incorporated to enable the blending of other ingredients and sprayability of the coating.
The term ‘binder’ is sometimes used interchangeably with resin, but the binder is, by definition, the nonvolatile portion of the liquid vehicle of a coating. It binds the pigment particles and other additives and the paint film as a whole to the material to which it is applied.
The binder provides:
Being a liquid, the solvent or water lowers the viscosity of the binder to allow application onto a substrate, which then evaporates to form a paint film. Solvents are measured in VOCs and are regulated by the EPA. Some, such as acetone, are considered “exempt” by the EPA and do not count as a VOC in regard to emission permits in most states.
Pigments
Pigments are solid particles that provide color and gloss control and remain insoluble in the binder. Typical pigments include: inorganic, organic and metallics.
Additives
Chemical additives are included in the paint formulation to provide unique characteristics and to improve the paint performance. Typical additives include catalysts, anti-blocking agents, defoamers, flow-control agents, gloss modifiers (high or low), storage stabilizers, UV-blocking agents, dispersants and rust inhibitors.
Oligomers provide bonding, pigment encapsulation, flow-out and the physical properties of the coating. Like solvent, the monomer reduces the viscosity, but also provides some properties similar to the oligomer, plus flexibility and crosslinking.
Monomer addition in most cases reduces the viscosity to a level that makes roll coating or rotary atomization an effective application method. For easier application or better flow out, UV coatings can be formulated at less than 100% solids by adding solvent or water. In this way, a conventional or HVLP spray gun can be used for application.
The photoinitiator acts as a catalyst to initiate the chemical reaction. Monomers and oligomers are converted to polymers, and crosslinking occurs during this free radical reaction.
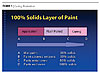
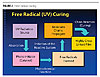 In comparison, the UV-cured coatings use a different method to form a cured paint film. When exposed to ultraviolet light, free radical-curable film formers crosslink into a paint film within seconds. These formulations are typically 100% solids, but also can be made with 5-50% solvent or water (see Figure 2).
In comparison, the UV-cured coatings use a different method to form a cured paint film. When exposed to ultraviolet light, free radical-curable film formers crosslink into a paint film within seconds. These formulations are typically 100% solids, but also can be made with 5-50% solvent or water (see Figure 2).
Specifically, this chemical process (chain reaction) forms a polymer from monomers and oligomers. UV cure may be accomplished in enclosed chambers saturated with high-intensity electrically generated UV light. The enclosed chamber is required because the presence of oxygen inhibits the curing of free-radical-initiated systems. The oxygen must be overcome with nitrogen or another inert gas. Due to the high cost of enclosed chambers, special formulations are commonly used to overcome the effects of oxygen that are typically present during curing. Today, UV systems rarely have enclosed chambers.
With regard to coating shrinkage, thermoset coatings are crosslinked with urea and/or melamine resin, which co-react resulting in only about a 1% shrink. In comparison, the reaction of free radical polymerization in UV coatings results in about a 20% shrink. When formulating, the chemist must compensate for this shrinkage.
In UV coatings, these accelerators or catalysts are called photoinitiators. For total curing to take place, the UV light must activate or “see” all of the photoinitiators. As a result, this imposes some limitations on the dry film thickness (~1 mil DFT) of the UV coatings, especially those containing pigments that can be UV-cured by a single pass. The pigment molecules act to absorb and reflect or block the UV light from reaching some of the photoinitiators.
As expected, black-pigmented coatings as well as dark colors are the most difficult to cure because of their characteristic to absorb light waves. The lighter, brighter pigments, in general, can be more readily cured.
UV light energy decreases with the square of the distance between the light source and the surface receiving the light. Therefore, the UV light source is kept as close to the coated part as possible. For this reason, UV cure is easier on flat surfaces. However, highly polished parabolic reflectors, strategically positioned light sources and/or rotation of the substrate (part) enable certain three-dimensional parts to be successfully UV coated, such as bicycle frames and hand tools.
UV curing occurs in a matter of seconds, which permits compact paint systems. The quick cure also reduces foreign particles on the surface and minimizes substrate heating, a great advantage for heat sensitive substrates.
UV light used in curing is in the spectral range between 240 and 450 nm (nanometers). This requires using photoinitiators to initiate polymerization within these wavelengths. Typically three distinct ranges of wavelengths can be achieved with the various light sources. A range of 240 to 320 nm (UVB and UVC) is obtained using an “H” bulb. This is a Mercury (Hg) “doped” bulb, which emits wavelengths that are shorter and primarily provide surface cure. These bulbs are typically used for non-pigmented, clear UV coatings.
A range from 320 to 400 nm (UVA) is obtained using a “D” bulb. This is an Iron (Fe) “doped” bulb, which emits medium wavelengths. A range of 400 to 450 nm is obtained using a “V” bulb. This Gallium (Ga) “doped” bulb emits longer wavelengths that can penetrate below the surface of the coating, down to the substrate. This provides superior adhesion and is necessary to fully cure pigmented coatings.
UV Lamps
UV lamps are controlled discharge devices that generate electromagnetic energy, giving off infrared and visible light, as well as ultraviolet radiation. The primary types of UV lamps are (a) electrode (medium pressure mercury arc lamp) and (b) electrode-less (microwave powered). Other UV lamps that are available include low-pressure mercury arc lamps (fluorescent) and high-pressure xenon arc lamps (both electrode type) and lasers, which are electrode-less.
1. Irradiance, watts/cm². This parameter indicates the intensity of the light or the UV lamp output needed; the greater the irradiance, the greater the depth of curing.
2. Wavelength of the irradiance, nm range (or UVB, UVC, UVA, UVV bands). This parameter helps determine the best bulb type for the cure (H, D or V). This wavelength also affects the photoinitiators used in the formulation and its absorbance.
3. Time, seconds or ft/min. The time needed for the formulation to cure will determine the lamp configuration. A static cure will specify the time in seconds and a dynamic cure, as in conveyored parts, will report speed in ft/min.
4. Energy, joules/cm². Energy is a measurement of irradiance and time. Logically, intensity of the light and the amount of time the coating sees this light, will have a great impact on the cure response. Typically, the paint line operator uses this measurement to monitor their process using a device called a radiometer.

Low Capital Investment
Several factors contribute to this such as:
It is highly recommended to design a 10- to 20-ft space after the paint application area and prior to the curing lamp to install infrared (IR) lamps. In some cases the heat and flash time is beneficial to flow out the 100%-solids coating and necessary when using less than 100% solids (water or solvent-based) UV coatings. It is very costly to retrofit the IR lamps and allocate space after the system has been installed.
Suited for Sensitive Substrates
Since the exposure to UV light is typically a matter of seconds, this system is well suited for substrates that cannot tolerate high temperatures. The increase in substrate temperature is minimal, with the ability to handle the part immediately after curing.
Lower Operating Costs
A major advantage of a liquid UV coating is the elimination of a costly thermal dryer or oven. In a liquid UV system, the oven is replaced with a small cluster of UV curing lamps. Costs to install these lamps are typically about half the cost to install a large capacity thermal dryer or oven. Annual operating costs of the oven versus the UV lamps are dramatically reduced as well.
For example, a large gas oven consumes ~1 MBTU/hr and requires large blowers to achieve a given production capacity. This same production capacity can be achieved with four UV lamps, requiring a total of only72 kW/hr.
Figure 3 illustrates typical operating costs per year. The fast curing of the UV process reduces the amount of time the wet coating is exposed to the environment. This reduces the likelihood of particles contaminating the coating, decreasing rejects and scrap.
Additionally, some defects cannot be found until the coating has completely cured. With the speed of the UV system, defects can be identified immediately so corrective action can be taken much faster, increasing the efficiency of the paint system.
 The length of the conveyor system is directly proportional to its replacement costs. Conveying systems passing through conventional thermal ovens are subject to accelerated wear as lubricants rapidly deteriorate and result in excessive bearing and guide wear. Staying with the 100-foot thermal oven example, conveyor maintenance costs for the curing portion of the line would average $8,000/year. Using a 10-foot bank of four UV lamps, conveyor maintenance costs would be only $400/year. Figure 4 illustrates conveyor chain replacement costs.
The length of the conveyor system is directly proportional to its replacement costs. Conveying systems passing through conventional thermal ovens are subject to accelerated wear as lubricants rapidly deteriorate and result in excessive bearing and guide wear. Staying with the 100-foot thermal oven example, conveyor maintenance costs for the curing portion of the line would average $8,000/year. Using a 10-foot bank of four UV lamps, conveyor maintenance costs would be only $400/year. Figure 4 illustrates conveyor chain replacement costs.
Since UV coatings do not dry until they are exposed to high-intensity UV light, reclaiming overspray is much easier, making for higher application efficiency.

In general, UV-cured materials are used as coatings, inks, adhesives and sealants for metal, plastics, wood, glass and composites. The following describes some of the major industries using UV-cured coatings and some of the products produced.
LED curing is defined by the longer wavelengths (greater than 400 nm, but with a much smaller range) that the LED bulbs emit. Some benefits that have been identified include lower energy usage, portability of the “lamps”, increased safety due to the wavelengths used and lower cost versus the current UV lamps. The biggest challenge facing the industry is to find photoinitiators that will initiate the free radical reaction at these longer wavelengths and still give the coating its desired properties. Research into this new technology is ongoing.
With the substantial growth of UV coatings in the last 15 years, a parallel product/technology comes to mind. Back in the early 90s, water-based liquid coatings were in a similar situation. Great technical strides were being made; enhanced properties were becoming available; emerging markets were ready to take advantage of any breakthrough, primarily because of increased regulatory restrictions. All this is why, “The Future of UV Coatings is Now.”
Note
This paper did not focus on EB (electron beam) cured coatings, since these coatings are used primarily in the graphics arts industry and use electron beams to induce curing, requiring a slightly different formulation as well as lead or concrete shielding for safety reasons.
Fueling this growth is not just the push for “green” technology and increased productivity required in manufacturing, but also the availability of new raw materials from suppliers. These new materials allow formulators the ability to produce UV coatings with improved physical properties and better product performance, which opens up new capabilities for users.

Can you think of any organization that has had 35 consecutive years of sales growth? A few come to mind that may fit that category, but you can count them on one hand. Statistics from past RadTech International shows indicate that the UV/EB formulated product growth for the North American market is just over 7% annually.
Fueling this growth is not just the push for “green” technology and increased productivity required in manufacturing, but also the availability of new raw materials from suppliers. These new materials allow formulators the ability to produce UV coatings with improved physical properties and better product performance, which opens up new capabilities for users.
Two factors are primarily driving the growth of UV-cured coatings.
- Environmental – At solids up to 100%, UV-coatings can provide a reduction or elimination of VOCs and HAPs.
- World-class manufacturing characterized by shorter cure times, automated processes, reduced lead time, reduced scrap, waste and rework, and just in time inventory.
Coating Components
Paint is typically defined as a liquid, containing a resin (or polymer), solvent (or water), additives and possibly pigment. For simplification, powder coatings and electrocoat will not be used for comparison.Resin
The resin can be thought of as the “film former,” which binds all the ingredients into a continuous system and forms a film when dried. Higher-molecular-weight synthetic resins are more generally referred to as polymers. Polymers are usually highly viscous, and solvent (or water) is incorporated to enable the blending of other ingredients and sprayability of the coating.
The term ‘binder’ is sometimes used interchangeably with resin, but the binder is, by definition, the nonvolatile portion of the liquid vehicle of a coating. It binds the pigment particles and other additives and the paint film as a whole to the material to which it is applied.
The binder provides:
- adhesion to the substrate;
- encapsulating and holding the pigment particles separate, preventing clustering;
- flowing out; forming a smooth, uniform film to optimize film properties, including gloss; and
- physical and chemical properties such as hardness, flexibility, durability, recoatability, abrasion and impact resistance, and water, UV, chemical and corrosion resistance.
Being a liquid, the solvent or water lowers the viscosity of the binder to allow application onto a substrate, which then evaporates to form a paint film. Solvents are measured in VOCs and are regulated by the EPA. Some, such as acetone, are considered “exempt” by the EPA and do not count as a VOC in regard to emission permits in most states.
Pigments
Pigments are solid particles that provide color and gloss control and remain insoluble in the binder. Typical pigments include: inorganic, organic and metallics.
Additives
Chemical additives are included in the paint formulation to provide unique characteristics and to improve the paint performance. Typical additives include catalysts, anti-blocking agents, defoamers, flow-control agents, gloss modifiers (high or low), storage stabilizers, UV-blocking agents, dispersants and rust inhibitors.
Thermal vs. Radiation Curing
Formulated UV-cured coatings can be looked at similarly, by replacing the words resin, solvent and catalyst with the words oligomer, monomer and photoinitiator.Oligomers provide bonding, pigment encapsulation, flow-out and the physical properties of the coating. Like solvent, the monomer reduces the viscosity, but also provides some properties similar to the oligomer, plus flexibility and crosslinking.
Monomer addition in most cases reduces the viscosity to a level that makes roll coating or rotary atomization an effective application method. For easier application or better flow out, UV coatings can be formulated at less than 100% solids by adding solvent or water. In this way, a conventional or HVLP spray gun can be used for application.
The photoinitiator acts as a catalyst to initiate the chemical reaction. Monomers and oligomers are converted to polymers, and crosslinking occurs during this free radical reaction.

Curing
Another way to distinguish between UV-cured coatings and solvent/water-based coatings is by the different types of polymers and how they cure. Solvent or waterborne polymers are often characterized based on how they form a paint film.- Lacquer – A lacquer is based on a synthetic thermoplastic film-forming polymer dissolved in organic solvent. It forms a paint film solely by solvent evaporation.
- Auto-oxidation – Polymers containing carbon-carbon double bonds that add molecular oxygen across the double bond to form a crosslinked paint film. Alkyds are typical of this type of reaction.
- Chemical curing – In this case, the system contains more than one polymer. One is a backbone polymer such as an alkyd or an acrylic and the other a crosslinking resin, such as melamine. Heat and a catalyst (internal or external) create a reaction, which causes the backbone polymer and the crosslinking agent to crosslink into a paint film.
- Emulsion-type film formers – These are similar to lacquers in that evaporation of the water (and coalescing solvent if present) results in coalescence of the polymer particles to form a paint film. These emulsions are usually acrylics; however, polyesters, polyurethanes, alkyds and epoxies are also available.

Radiation Curing
Radiation-curable coatings contain various accelerators or catalysts that are dormant, until exposed to UV light. The absorption of UV wavelengths triggers a free-radical reaction among chemical groups that, within seconds, results in crosslinking (curing).Specifically, this chemical process (chain reaction) forms a polymer from monomers and oligomers. UV cure may be accomplished in enclosed chambers saturated with high-intensity electrically generated UV light. The enclosed chamber is required because the presence of oxygen inhibits the curing of free-radical-initiated systems. The oxygen must be overcome with nitrogen or another inert gas. Due to the high cost of enclosed chambers, special formulations are commonly used to overcome the effects of oxygen that are typically present during curing. Today, UV systems rarely have enclosed chambers.
With regard to coating shrinkage, thermoset coatings are crosslinked with urea and/or melamine resin, which co-react resulting in only about a 1% shrink. In comparison, the reaction of free radical polymerization in UV coatings results in about a 20% shrink. When formulating, the chemist must compensate for this shrinkage.
In UV coatings, these accelerators or catalysts are called photoinitiators. For total curing to take place, the UV light must activate or “see” all of the photoinitiators. As a result, this imposes some limitations on the dry film thickness (~1 mil DFT) of the UV coatings, especially those containing pigments that can be UV-cured by a single pass. The pigment molecules act to absorb and reflect or block the UV light from reaching some of the photoinitiators.
As expected, black-pigmented coatings as well as dark colors are the most difficult to cure because of their characteristic to absorb light waves. The lighter, brighter pigments, in general, can be more readily cured.
UV light energy decreases with the square of the distance between the light source and the surface receiving the light. Therefore, the UV light source is kept as close to the coated part as possible. For this reason, UV cure is easier on flat surfaces. However, highly polished parabolic reflectors, strategically positioned light sources and/or rotation of the substrate (part) enable certain three-dimensional parts to be successfully UV coated, such as bicycle frames and hand tools.
UV curing occurs in a matter of seconds, which permits compact paint systems. The quick cure also reduces foreign particles on the surface and minimizes substrate heating, a great advantage for heat sensitive substrates.
Energy Sources
WavelengthsUV light used in curing is in the spectral range between 240 and 450 nm (nanometers). This requires using photoinitiators to initiate polymerization within these wavelengths. Typically three distinct ranges of wavelengths can be achieved with the various light sources. A range of 240 to 320 nm (UVB and UVC) is obtained using an “H” bulb. This is a Mercury (Hg) “doped” bulb, which emits wavelengths that are shorter and primarily provide surface cure. These bulbs are typically used for non-pigmented, clear UV coatings.
A range from 320 to 400 nm (UVA) is obtained using a “D” bulb. This is an Iron (Fe) “doped” bulb, which emits medium wavelengths. A range of 400 to 450 nm is obtained using a “V” bulb. This Gallium (Ga) “doped” bulb emits longer wavelengths that can penetrate below the surface of the coating, down to the substrate. This provides superior adhesion and is necessary to fully cure pigmented coatings.
UV Lamps
UV lamps are controlled discharge devices that generate electromagnetic energy, giving off infrared and visible light, as well as ultraviolet radiation. The primary types of UV lamps are (a) electrode (medium pressure mercury arc lamp) and (b) electrode-less (microwave powered). Other UV lamps that are available include low-pressure mercury arc lamps (fluorescent) and high-pressure xenon arc lamps (both electrode type) and lasers, which are electrode-less.
Measurement Parameters
Every UV-curable coating specification should include parameters for the amount of illumination onto a surface and a timeframe.2 The key parameters are as follows.1. Irradiance, watts/cm². This parameter indicates the intensity of the light or the UV lamp output needed; the greater the irradiance, the greater the depth of curing.
2. Wavelength of the irradiance, nm range (or UVB, UVC, UVA, UVV bands). This parameter helps determine the best bulb type for the cure (H, D or V). This wavelength also affects the photoinitiators used in the formulation and its absorbance.
3. Time, seconds or ft/min. The time needed for the formulation to cure will determine the lamp configuration. A static cure will specify the time in seconds and a dynamic cure, as in conveyored parts, will report speed in ft/min.
4. Energy, joules/cm². Energy is a measurement of irradiance and time. Logically, intensity of the light and the amount of time the coating sees this light, will have a great impact on the cure response. Typically, the paint line operator uses this measurement to monitor their process using a device called a radiometer.
Radiometers
Three factors are important to an efficient UV-cure system: irradiance, energy and wavelength. These can be measured by a radiometer, which is about twice the size of a modern cell phone. This device can be attached to the conveyor or part fixture and passed across the UV light, simulating the UV exposure to the part (coating). Values can usually be read from an LED screen. Once a satisfactory UV process is established, this instrument can be used to quantify the preferred exposure and ensure repeatability and reproducibility.
UV Coating Benefits
While gathering information and data regarding the investment into a UV coating line, the following benefits should be considered.Low Capital Investment
Several factors contribute to this such as:
- no drying oven required;
- less overhead conveyor length;
- smaller area for unloading painted parts; and
- smaller system footprint.
It is highly recommended to design a 10- to 20-ft space after the paint application area and prior to the curing lamp to install infrared (IR) lamps. In some cases the heat and flash time is beneficial to flow out the 100%-solids coating and necessary when using less than 100% solids (water or solvent-based) UV coatings. It is very costly to retrofit the IR lamps and allocate space after the system has been installed.
Suited for Sensitive Substrates
Since the exposure to UV light is typically a matter of seconds, this system is well suited for substrates that cannot tolerate high temperatures. The increase in substrate temperature is minimal, with the ability to handle the part immediately after curing.
Lower Operating Costs
A major advantage of a liquid UV coating is the elimination of a costly thermal dryer or oven. In a liquid UV system, the oven is replaced with a small cluster of UV curing lamps. Costs to install these lamps are typically about half the cost to install a large capacity thermal dryer or oven. Annual operating costs of the oven versus the UV lamps are dramatically reduced as well.
For example, a large gas oven consumes ~1 MBTU/hr and requires large blowers to achieve a given production capacity. This same production capacity can be achieved with four UV lamps, requiring a total of only72 kW/hr.
Figure 3 illustrates typical operating costs per year. The fast curing of the UV process reduces the amount of time the wet coating is exposed to the environment. This reduces the likelihood of particles contaminating the coating, decreasing rejects and scrap.
Additionally, some defects cannot be found until the coating has completely cured. With the speed of the UV system, defects can be identified immediately so corrective action can be taken much faster, increasing the efficiency of the paint system.

Improved Productivity
UV coating materials typically cure in seconds versus up to several hours in a conventional thermal oven, so the cycle time for each coated part can be cut dramatically. This allows quicker response to your customers’ requests and better delivery performance. Faster cycle time also leads directly to a reduction of Work in Process (WIP), lowering inventories and freeing up capital for other investments.Since UV coatings do not dry until they are exposed to high-intensity UV light, reclaiming overspray is much easier, making for higher application efficiency.

Reduced Environmental Impact
A 100%-solids UV formulation contains zero VOCs and no HAPs. This will have a positive impact on the allowances in your emissions permit. If required, non-VOC solvents, such as acetone, can be used for less than 100%-solids formulations. In addition, paint filters can be dried and disposed in the trash, as opposed to a costly hazardous waste hauler. Figure 5 summarizes the cost comparisons.Application Methods and End Uses
UV-cured formulations have been applied by a variety of techniques. These include roll coating, flow or curtain coating, dip coating, and spraying with either a conventional or HVLP gun.In general, UV-cured materials are used as coatings, inks, adhesives and sealants for metal, plastics, wood, glass and composites. The following describes some of the major industries using UV-cured coatings and some of the products produced.
- Graphic Arts – Sometimes uses the EB method for curing; products include screen, litho, jet print and overprint varnishes.
- Wood Industry – Products include shelving, cabinets, furniture and flooring.
- Optical Fibers – Two UV protective coatings are applied to prevent damage to the fiber through bending and handling. A hard outer (buffer) coating and a soft (cushion) coating, between the outer coating and the fiber, is used to achieve the necessary protection.
- Compact Discs – Also used as a protective coating on CDs and DVDs.
- Adhesives – Used to bind together various substrates.
- Rapid Prototyping – This is a new development that allows a three-dimensional model on a CAD system to be translated directly into a 3-D functional model. Products include mold castings, functional production models (engine manifold), damaged bone structure (surgeons) and teeth (dentists).
- Dental Industry – Dental resins for tooth repair, sealants, impression material and Rapid Prototyping.
- Composites/Plastics – Used on automotive parts, vacuum metallizing, aircraft parts, bottles, bicycles, cosmetics, glass fibers and carbon fibers.
- Industrial – Products include hand tools, metal cans, propane tanks and electronic circuit boards.
Future of UV Coatings
It is predicted that there will be a continued growth of 5-10% per year in UV coatings usage in North America. This will be driven by:- environmental compliance (increased VOC restrictions);
- increased speed/productivity;
- energy savings (lower costs);
- improved properties;
- ability to cure pigmented formulations;
- improved adhesion to metal and plastics;
- automotive industry (plastics); and
- new technology (LED curing).
LED curing is defined by the longer wavelengths (greater than 400 nm, but with a much smaller range) that the LED bulbs emit. Some benefits that have been identified include lower energy usage, portability of the “lamps”, increased safety due to the wavelengths used and lower cost versus the current UV lamps. The biggest challenge facing the industry is to find photoinitiators that will initiate the free radical reaction at these longer wavelengths and still give the coating its desired properties. Research into this new technology is ongoing.
Conclusion
Hopefully some of the mystery about UV coatings has been removed and has allowed you to consider some of the potential benefits that a UV system can bring to your paint operation and your products. Some benefits that have already been realized include: reduced operating costs from increased efficiency, compliance with new regulatory requirements, and entrance into new markets.With the substantial growth of UV coatings in the last 15 years, a parallel product/technology comes to mind. Back in the early 90s, water-based liquid coatings were in a similar situation. Great technical strides were being made; enhanced properties were becoming available; emerging markets were ready to take advantage of any breakthrough, primarily because of increased regulatory restrictions. All this is why, “The Future of UV Coatings is Now.”
Note
This paper did not focus on EB (electron beam) cured coatings, since these coatings are used primarily in the graphics arts industry and use electron beams to induce curing, requiring a slightly different formulation as well as lead or concrete shielding for safety reasons.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





