Paint Formulations and the Need for Zeta Potential
Zeta potential is a physical property exhibited by any particle in suspension. It can be quantified using electrophoretic mobility (electrophoresis) measurement and adjusted to optimize coating formulations. Studying zeta potential results enables the building of relationships between the chemical composition of formulated coatings and the required final physical properties and color. Today, zeta potential is easy to measure and aids in predicting long-term stability and optimizing product properties.
Electrophoresis
When an electric field is applied across an electrolyte, suspended charged particles are attracted to the electrode of opposite charge. Viscous forces acting on the particles tend to oppose this movement. When equilibrium is reached between the two opposing forces, the particles move with constant velocity. This velocity depends on electric field or voltage gradient strength, the dielectric constant of the medium, its viscosity and the zeta potential.
A particle’s velocity in a unit electric field is known as its electrophoretic mobility. Zeta potential is related to electrophoretic mobility through the Henry equation, which relates it to zeta potential based on physical constants such as the dielectric constant, viscosity and ionic strength of the medium. Current instrumentation makes zeta potential measurement easy and automatic.
Measuring Electrophoretic Mobility
The essence of a classical micro-electrophoresis system is a capillary cell with electrodes at either end, to which a potential is applied. Particles move towards the electrode, their velocity is measured and is expressed in unit field strength as their mobility.
Early methods involved direct observation of individual particles using ultra-microscope techniques. This procedure suffers from a number of disadvantages, including the strenuous effort required to make a measurement, particularly with small or poorly scattering particles. Modern methods, however, are fully automated. One of the most widely used systems is the Zetasizer Nano (Malvern Instruments), which combines laser Doppler electrophoresis with phase analysis light scattering (PALS) in the patented M3-PALS technique. This allows measurement of particle electrophoretic mobility even in samples with low mobility and also in high-conductivity samples. In addition, the low voltages involved avoid any risk of sample effects due to heating.
 Credit: Malvern Instruments Inc. Credit: Malvern Instruments Inc.
|
| Figure 1 Click to enlarge |
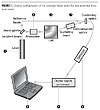
Optical Configuration
A zeta potential measurement system consists of six main components (Figure 1). First, a laser [1] is used as a light source to illuminate the particles within the sample. For zeta potential measurements, this is split to provide an incident and reference beam. The incident beam passes through the center of the sample cell [2], and the light scattered at a forward angle is detected [3]. When an electric field is applied across the cell, any particles moving through the measurement volume will cause the intensity of light detected to fluctuate with a frequency proportional to the particle speed, and this information is passed to a digital signal processor [4] and onto a computer [5]. The Zetasizer Nano software, for example, produces a frequency spectrum from which the electrophoretic mobility and hence zeta potential information is calculated. The intensity of the detected, scattered light must be within a specific range for the detector to successfully measure it. This is achieved using an attenuator [6], which reduces the intensity of the laser and, hence, reduces the intensity of the scattering. To correct for any differences in the cell wall thickness and dispersant refraction, compensation optics [7] are installed to maintain optimum alignment.
Stability and Interfacial Surface Effects
 Credit: Malvern Instruments Inc. Credit: Malvern Instruments Inc. |
| Figure 2 Click to enlarge |
Two fundamental mechanisms affect dispersion stability (Figure 2).
- Steric repulsion – where polymers added to the system adsorb onto the particle surface and preventing particle surfaces coming into close contact. When sufficient polymer adsorbs, the thickness of the coating is enough to maintain particle separation through steric repulsion between the polymer layers. At such separations the van der Waals forces are too weak to cause the particles to adhere.
- Electrostatic or charge stabilization is the effect on particle interaction due to the distribution of charged species in the system.
Each mechanism has its benefits for particular systems. Steric stabilization is simple, requiring only a suitable polymer. However, it can be difficult subsequently to flocculate the system if this is what is required. Expense may be a drawback, and sometimes it is not desirable to have a polymer present.
Electrostatic or charge stabilization works by simply altering the concentration of ions in the system – a reversible and potentially inexpensive process.
Zeta potential has long been recognized as a very good index of the magnitude of the interaction between colloidal particles. Zeta potential measurements are commonly used to assess the stability of colloidal systems.
Origins of Surface Charge
Most colloidal dispersions in aqueous media carry an electric charge. This can originate in many ways depending upon the nature of the particle and its surrounding medium. The more important mechanisms are the ones considered here.
Ionization of Surface Groups
Dissociation of acidic groups on the surface of a particle will give rise to a negatively charged surface. Conversely, a basic surface will take on a positive charge. In both cases, the magnitude of the surface charge depends on the acidic or basic strengths of the surface groups and on the pH of the solution. The surface charge can be reduced to zero by suppressing surface ionization – by decreasing the pH for negatively charged particles or increasing it for positively charged particles.
Differential Loss of Ions from the Crystal Lattice
As an example, consider a crystal of silver iodide placed in water. If equal amounts of Ag+ and I- ions were to dissolve from the surface, the surface would be uncharged. In fact silver ions dissolve preferentially, leaving a negatively charged surface. If Ag+ ions are now added the charge falls to zero. Further addition leads to a positively charged surface.
Adsorption of Charged Species (Ions and Ionic Surfactants)
Surfactant ions may be specifically adsorbed on the surface of a particle, leading, in the case of cationic surfactants, to a positively charged surface and, in the case of anionic surfactants, to a negatively charged surface.
The Electrical Double Layer
Based on the above mechanisms, latexes, pigments and in fact most materials suspended in aqueous media, carry a surface charge. The development of a net charge at the particle surface affects the distribution of ions in the surrounding interfacial region, resulting in an increased concentration of counter ions (ions of opposite charge to that of the particle) close to the surface. Thus an electrical double layer exists round each particle.
 Credit: Malvern Instruments Inc. Credit: Malvern Instruments Inc. |
| Figure 3 Click to enlarge |
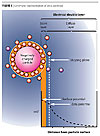
Zeta Potential
The liquid layer surrounding the particle exists as two parts; an inner region (Stern layer) where the ions are strongly bound and an outer (diffuse) region where they are less firmly associated. Within the diffuse layer there is a notional boundary inside which the ions and particles form a stable entity. When a particle moves (e.g., due to gravity), ions within the boundary move with it. Those ions beyond the boundary stay with the bulk dispersant. The potential at this boundary (surface of hydrodynamic shear) is the zeta potential (Figure 3).
The magnitude of the zeta potential gives an indication of the potential stability of the colloidal system. If all the particles in suspension have a large negative or positive zeta potential then they will tend to repel each other and there is no tendency for the particles to come together. However, if the particles have low zeta potential values then there is no force to prevent the particles coming together and therefore they will flocculate or coagulate.
What This Means for Coatings
 Credit: Malvern Instruments Inc. Credit: Malvern Instruments Inc. |
| Figure 4 Click to enlarge |
Historically, coatings have been formulated as solventborne (oil-based paints) or waterborne paints (latex, water-based paints). Environmental requirements limiting VOCs have mandated the reduction of solvent concentration in coating products. Other factors such as ease of use have helped minimize consumer use of oil-based paints. Therefore latex, waterborne paints command an ever-increasing share of the market. It is easy to study zeta potential in these coatings and, because of the additives and formulated nature of the products, manipulating zeta potential is highly relevant as an optimization tool.
Specifically, because water has a high surface tension, waterborne paints present unique formulation challenges. It is more difficult to wet the non-polar components of the formulation, such as pigments, etc. Also, because of the physical properties of the oil and polymers used in these aqueous based dispersions, the film formation characteristics of the latex paints themselves tend to be restricted. Therefore, a variety of additives including surfactants (wetting agents), processing aids and polyelectrolytes, play an important role in latex (waterborne) paint products.
As the particle concentration increases during wetting, spreading and coalescence of the coating, the viscosity increases. Based on this scenario, there is a point of phase inversion where the latex particles touch and deform into what will become the continuous phase – the film/coating. The process of film formation goes through four steps: (1) discrete emulsion particles; (2) concentration of the particles or evaporation of the water phase; (3) coalescence where the particles deform; and (4) maturation or diffusion of the chains.
Waterborne paints require additives to build the interfacial characteristics to achieve the desired stability for the storage and film formation characteristics needed in the final product. Amongst other properties these include good wetting, uniform film formation, zero defects, specific color, brightness, long-term adhesion and strength.
Zeta potential provides a measurement technique that allows the study of latex and pigment components, and also of additives and their possible combinations. Such data provides insight into the particle interfacial structure and allows optimization of each component in the formulation to give the desired final product properties.
 Credit: Malvern Instruments Inc. Credit: Malvern Instruments Inc. |
| Figure 5 Click to enlarge |
Figure 4 shows a latex or film former study as a function of a surface-active processing aid. These data represent an initial step in understanding how various additives interact with the surface of a film former in a paint formulation. The results indicate that this specific processing aid does adsorb to the surface of the latex, which has a -20 mV zeta potential initially without any additive. It reverses charge and becomes cationic with increased addition of a cationic processing aid. This type of data not only shows surface activity but also indicates the optimal concentration required for the processing aid to be effective.
Figure 5 shows a study of a series of paint formulations prepared with the identical latex and pigment components but having different additives. Labeling these formulations as Paint 1 through 4 in the order of the highest negative zeta potential to highest positive zeta potential, and relating zeta potential to coating defects of the final product, a clear trend arises. In this case it indicated that a cationic formulation resulted in fewer defects per 100,000 parts.
Conclusion
In conclusion, paints are multi-component systems formulated to produce specific product properties. The rate and uniformity at which these coatings wet spread and coalesce into a film can be controlled by the chemical components used in the formulation. Coatings, depending on their end use, are complex formulations with a minimum of four components added into a dispersed mixture.
Zeta potential measurements provide insight into the stability of the latex and pigments in the dispersion. Zeta potential also helps to elucidate the interfacial characteristics of the dispersed particulates and can be related to the required final product properties.
For more information, visit www.malvern.com or call 508/768.6400.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






