Improving Flexibility of Bis-Phenol A Epoxy Paint
To answer this need, scientists at Dow Corning Corp. set out to develop a new class of organo-functional siloxane resins to serve as epoxy resin crosslinkers that would provide bis-phenol A epoxy coatings with the flexibility and the recoatability unavailable with existing organo-functional silicone polymers.
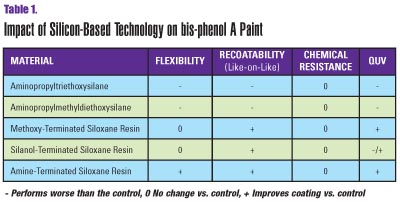
In their study, the scientists compared modifications of a bis-phenol A epoxy paint prepared on a Cowles blade dispersator. After reduction with methyl isobutyl ketone to 30 seconds per #4 Zahn cup, the coatings were modified with a variety of silicon-based technologies, including aminopropyltriethoxysilane, aminopropylmethyldiethox-ysilane, a methoxy-terminated siloxane resin, a silanol-terminated siloxane resin and a novel amine-terminated siloxane resin.
The control paint was formulated at a 1:1 amine hydrogen-to-epoxy ratio using a standard, cycloaliphatic amine curing agent. The modified coatings were formulated by incorporating the silicon-based materials at four levels ranging from approximately 3 percent to 35 percent of the binder system.
For the silicon-based materials containing amine functionality, the level of organic crosslinker was reduced to maintain the 1:1 amine hydrogen-to-epoxy ratio. The level of organic crosslinker was maintained for the silicon-based materials that did not contain amine functionality. For all samples, the pigment-to-binder ratio was maintained at a 2:5 ratio.
The samples were mixed for 30 min on a mixing wheel and then allowed a 20-min gestation period before application. Next, 6-mil and 10-mil drawdowns were made on chromated aluminum panels and allowed to ambient cure for 14 days. After 14 days, the samples were evaluated for flexibility and recoatability, plus other important attributes, including gloss, chemical and solvent resistance, hardness, QUV exposure, adhesion and dry time. The outcomes are as follows.
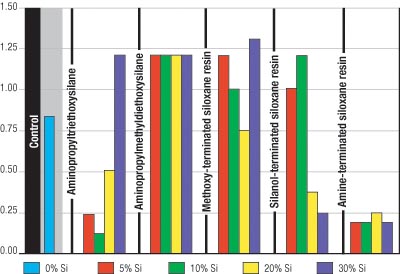
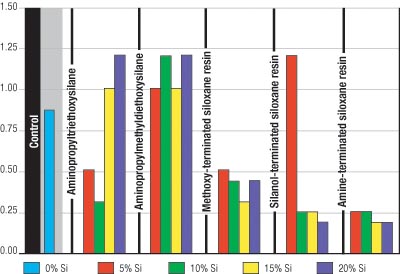
The flexibility of each coating was tested at 6- and 10-mil wet film thicknesses. Figures 1 and 2 illustrate that the amine-terminated siloxane resin significantly improved coating flexibility. Further, film thickness did not negatively affect the flexibility of the coating.
The methoxy- and silanol-terminated siloxane resins improved the flexibility on the 6-mil coatings. However, the 10-mil coatings were significantly less flexible. The coatings containing aminopropyltriethoxy- and aminopropylmethyl-diethoxysilanes were very brittle and, therefore, did not improve flexibility.
5: Excellent wetting and flow; no film defects such as craters and fisheyes
4: Slight dewetting and/or edge crawling; minimal defects
3: Moderate dewetting and/or edge crawling; minimal defects
2: Extensive dewetting and/or edge crawling; few defects
1: Extensive dewetting and/or edge crawling; numerous defects
0: Severe dewetting and/or edge crawling; extensive defects
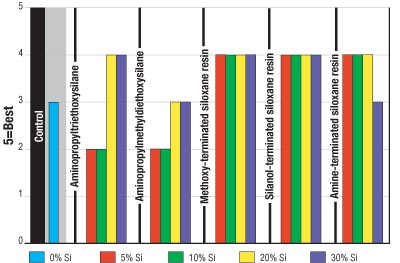
Figure 3 shows that the "like-on-like" recoatability of the samples improved with the addition of methoxy-, silanol- and amine-terminated siloxane resins. Addition of the silane-modified samples demonstrated a decrease in the recoatability of the coating.
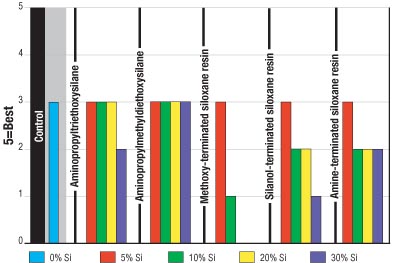
Figure 4 indicates that the silicone resins had a negative effect on recoatability with the control paint, whereas the silanes did not have an impact on the recoatability of the coating with the control.
Chemical and Solvent Resistance: To test chemical resistance, the scientists placed a 4 cm filter paper on a coated aluminum panel, saturated the paper with five drops of a chemical (see below), and covered the paper with a 4 cm watch glass. After 24 hr, the panel was rinsed with water and rated as follows:
5: No effect
4: Slight color change or blisters
3: Moderate color change or blisters or 1 percent to 10 percent film loss
2: Severe color change or blisters or 11 percent to 30 percent film loss
1: 31 percent to 75 percent film loss
0: 76 percent to 100 percent film loss
Chemicals used included concentrated hydrochloric acid, glacial acetic acid, nitric acid (50 percent), sulfuric acid (50 percent), sulfuric acid (5 percent), potassium hydroxide (50 percent), and sodium hydroxide (50 percent).
The silanol resin performed similar to the control, whereas the other silicon-based materials showed a slight decrease in chemical resistance, which was caused by the degradation of the coatings from chemical attack by the glacial acetic acid.
To test solvent resistance, the scientists wrapped eight layers of cheesecloth around the ball end of a 24-oz ball peen hammer and secured it with copper wire. The cheesecloth was then saturated with methyl ethyl ketone and placed on a coated panel. No downward pressure was applied to the hammer. The hammer was rubbed back and forth perpendicularly in the direction of the drawdown. A forward and reverse stroke was considered one double rub. Double runs were performed until a breakthrough to the metal surface was noted or until 200 rubs had been completed. All of the coatings, including the control, exceeded 200 double rubs.
Modification with the aminopropyltriethoxy- and aminopropylmethyldiethoxysilanes had minimal effect on hardness. However, incorporation of the methoxy- and silanol-terminated siloxane resins reduced the hardness significantly. This reduction was a direct result of the silicone resins not crosslinking into the system.
The amine-terminated siloxane resin had no impact on coating hardness with the low addition level (~4 percent) at "4H" and moderate impact at 36 percent loading with "H" hardnesses, both of which are lower than the hardness of the control sample, which is "4H."
QUV Exposure: Panels of each formulation were placed in a QUV-B weatherometer for 1000 hr. Both silanes delaminated and crazed during exposure to the QUV-B. The control paint did not perform well either, experiencing severe discoloration and loss of gloss.
The methoxy-terminated siloxane resin, the silanol-terminated siloxane resin, and the amine-terminated siloxane resin performed better than the control. The silanol resin initially appeared to have the best resistance to UV exposure. However, after closer evaluation, pockets of pinholes were found in the coating. The pinhole ruptures were likely due to the evolution of water during the condensation cure of the resin at elevated temperatures. No other samples exhibited this defect.
Dry Time: To test coating dry time, the scientists made a 4" wide, 6-mil wet film thickness drawdown on a pane of glass. A dry time recorder was immediately positioned over the wet film and switched on. A stylus with a 12-gram weight scribed a circle and, after a complete rotation, the results were evaluated as follows:
Set-to-touch: During the first drying stage, the film was mobile and partially flowed back unto the scribed channel. The film was considered "set-to-touch" when the material no longer flowed back and the stylus began to leave a clear channel.
Surface-dry: When the stylus no longer left a clear channel but began to rupture the dry upper layer of the curing film, the surface was considered "dust-free."
Through-dry: When the stylus no longer ruptured or dented the film, but moved freely on the surface, the cross-section of the film was considered to have reached a "dry-hard" condition.
The test indicated that both the silicon-based coatings and the control coating were set to touch within 4 hr and thoroughly dry 6 hr after coating.
Reaching a Conclusion
The scientists concluded that incorporating a new amino-functional siloxane resin ensures the most significant improvement in flexibility of bis-phenol A epoxy paint at various thicknesses with minimal impact on other aspects of coating performance.Lisa A. Honaman is a coatings application engineer/technical service specialist and Gerald L. Witucki is an industry specialist at Dow Corning Corp., Midland, Mich. Detailed technical information on testing procedures and results are available online at www.dow corning.com/coatings.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!







