Curing What Ails Concrete Floors with Polyaspartic Technology

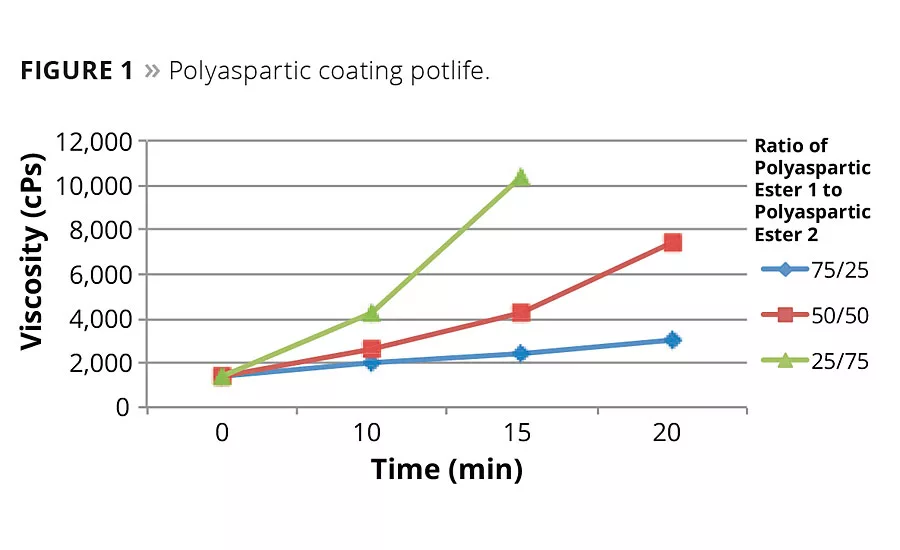
Figure 1
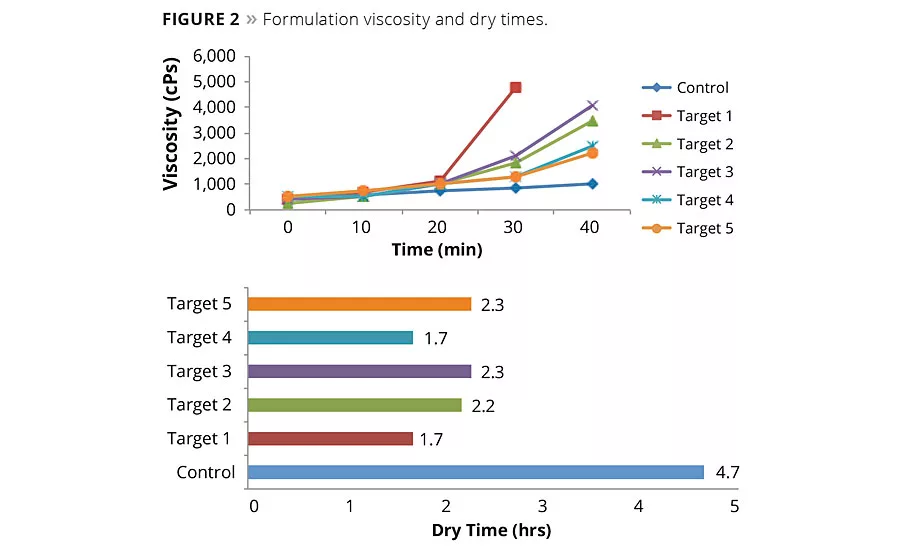
Figure 2
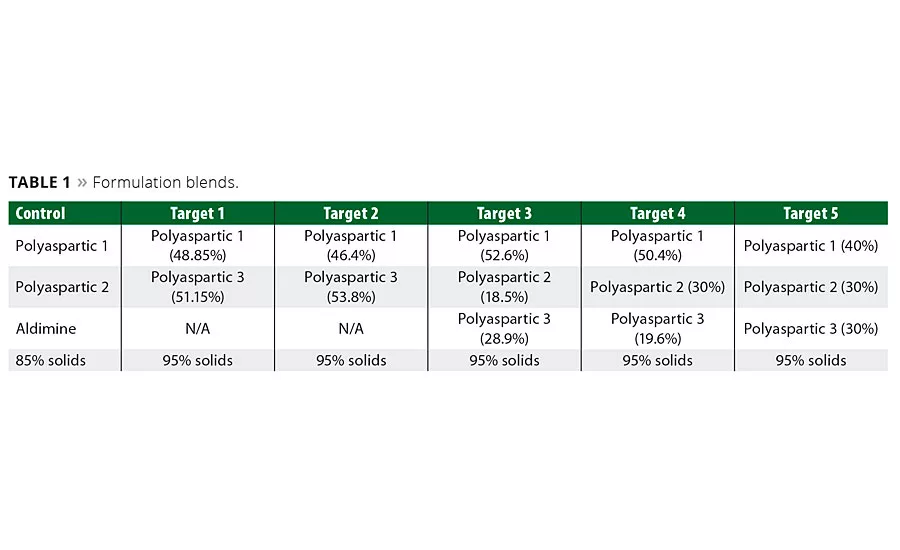
Table 1
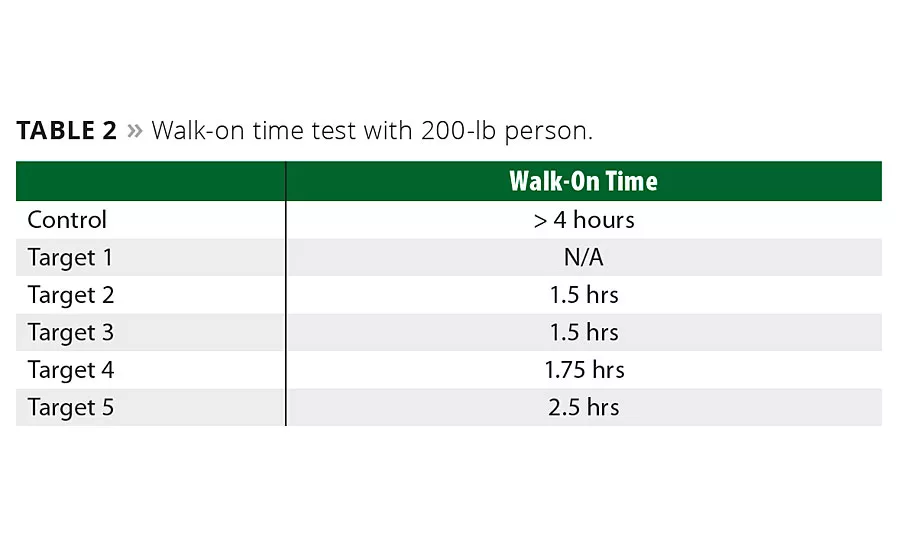
Table 2
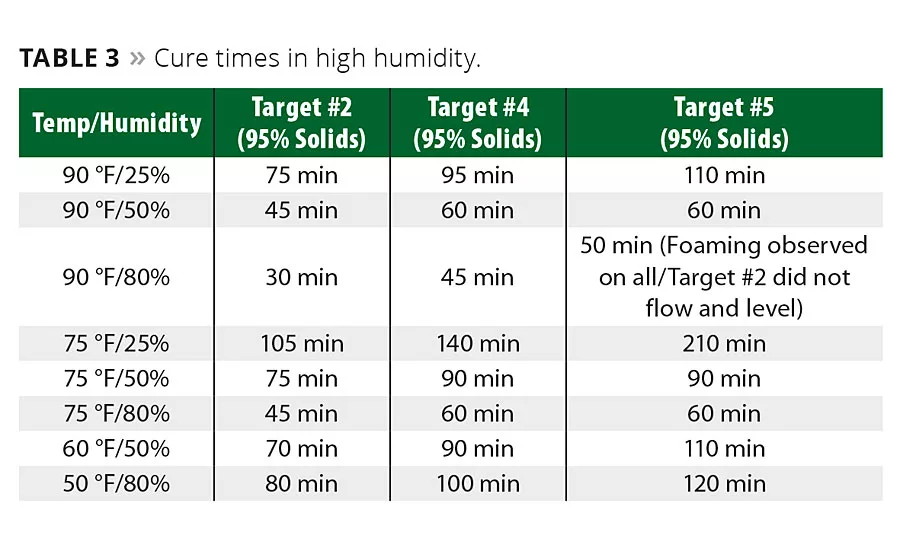
Table 3

Table 4
Concrete is a proven material for flooring and infrastructure. Under the right circumstances, concrete can last a half century or more, according to the International Association of Home Inspectors. Concrete’s durability is dependent on resistance to conditions such as moisture, chemicals and foot or vehicle traffic, to name just a few. The Grand Canyon is a stunning natural visual to the erosion power of moisture and other conditions. Of course, concrete floors are not expected to endure millions of years. Yet, adverse conditions can lead to structural and aesthetic issues for concrete floors. Enter a protective coating, which can significantly extend the functional life of concrete.
A protective coating or sealer is designed to enhance the color and aesthetics of the floor surface as well as protect it from wear, chemicals and staining agents. It also helps meet LEED and sustainability initiatives focused on lower volatile organic compound (VOC) content. For years, high-performance coatings have been a viable solution to protect industrial floors in production facilities, warehouses, laboratories and food processing plants. Additionally, a growing trend in the concrete flooring market uses a poured-in-place slab as a functional design element. This is achieved with a decorative staining treatment and a protective topcoat or a polished concrete procedure with a penetrating sealer.
During ongoing listening events with specifiers, contractors and owners we have learned of several preferences.
- Specifiers want low-VOC, LEED-compliant options with long-term, proven durability and national availability.
- Contractors want a product that is easier to apply with minimal temperature limitations, a long working time but a short walk-on time. They prefer a lower-odor option to minimize disruption at a client’s facility. They would also like to reduce labor costs by using faster-curing coatings or higher-build coatings that eliminate coating layer(s).
- Facility owners want minimal disruption to operations during a coating application. They want lower odor during application and quick curing for fast return to service. Visually, facility owners want the area to retain the original color and gloss over time, protect the floor from wear and contaminants, and be easy to maintain.
Polyaspartic esters, when formulated into protective concrete floor coatings, can address these desired attributes effectively by providing:
- Adjustable cure times for faster return to service;
- Thick, high-build finishes;
- Ultralow-VOC systems;
- Low viscosity;
- Low-odor systems that eliminate odorous, viscosity-reducing additives;
- Systems that are not greatly affected by ambient temperature; and
- Proven 15+-year durability record in industrial maintenance applications.
Polyaspartic Chemistry History
Developed in the early 1990s by Covestro, formerly Bayer MaterialScience, polyaspartic coatings are based on the reaction of an aliphatic polyisocyanate and a polyaspartic ester, which is an aliphatic diamine. The term aspartics is a generic term applied to the general chemical structural feature found in the amino acid – aspartic acid – hence the name polyaspartic ester for the raw material that contains multiple alkyl ester adducts of the aspartic acid.
The first applications were polyaspartic esters as co-reactants for aliphatic hardeners in conventional two-component (2K) polyurethane solventborne coating formulations in which the polyaspartic esters were used as reactive diluents allowing for higher solids polyurethane coatings, according to Zwiener, et al. The polyaspartic esters could be blended with hydroxyl-functional polyesters and polyacrylate co-reactants, thus allowing for reduction of VOCs in relatively high-solvent-containing coating systems. More recent developments have concentrated on achieving low- or near-zero-VOC coatings where the polyaspartic ester is the primary portion of the co-reactant for reaction with an aliphatic hardener.
Today’s raw material ingredients allow a formulator to control the rate of reaction and cure time. They can be formulated with very high solids (70-100 percent) and applied at high film builds (up to 15 mils wet film thickness in a single coat). These attributes mean polyaspartic coatings can significantly reduce the amount of time waiting for a coating to cure, ultimately reducing costs for an in-shop painting operation. In fact, a two-coat (polyaspartic urethane and epoxy zinc) process can save as much as 18 hours compared with a three-coat system (polyurethane, epoxy and epoxy zinc). When compared with a three-coat coating process, a two-coat polyaspartic coating system can reduce painting costs by nearly 8 percent, according to research by Helsel, et al.
The unique and adjustable reactivity of the polyaspartic ester co-reactant is illustrated in Figure 1, where changing the ratio of the two polyaspartic ester products changes the viscosity profile (which is a measure of reactivity) of the two-component mixture. Traditional 2K aliphatic polyurethanes typically cure (defined here as dry-to-touch) on the order of 6 to 12 hrs, compared with polyaspartic coatings that typically cure in 1 to 4 hrs. The fast-curing feature of these coatings can provide significant money-saving productivity improvements. Other advantages include low-temperature curing, abrasion resistance and corrosion resistance.
Application of Polyaspartic Coatings
While spray application techniques include the use of plural-component spray equipment, many applications can be applied with conventional sprayers or even brush and roll techniques, making application much less complicated and less prone to error. The polyaspartics technology is often used as a topcoat due to its inherent resistance to color and gloss changes because of ultraviolet exposure. The polyaspartic coatings, for example, can be formulated to very high solids (70- to 100-percent solids) and can be applied at higher film builds (up to 0.4 mm, or 15 mils, wet film thickness in a single coat) than typical 2K aliphatic polyurethanes. This is especially important when considering concrete floor coatings since it may allow for the elimination of a coating step in the process yet still achieve the desired total film build thickness.
Polyaspartic coatings can be formulated to achieve a 1:1 or 2:1 (by volume) mix ratio. This is accomplished by adding a higher-molecular-weight polyisocyanate or lower-molecular-weight reactive diluents, or solvents to the formula. Specifier preferences and current regulations are leading to a move away from formulations using solvents that may contain VOCs.
Due to their reactive nature, coatings made with polyaspartic esters can be applied at temperatures below 50 °F, which extends the application season for commercial and residential projects. Typical polyaspartic coatings can be applied from start (basecoat) to finish (topcoat) in an 8-hr work day. Polyaspartic coatings are often used as a topcoat over other floor coating technologies due to their nonyellowing nature and excellent durability.
New Formulation Development
Polyaspartic technology continues to advance as new and better formulations are developed to better meet market needs. Based on the coating properties our customers have shared with us, we tested several formulations as part of a design of experiment (DOE). A new polyaspartic resin was developed to address the industry desire to lower VOCs, raise solids levels and optimize the workability time.
Our goal was to determine the best blend of raw materials to provide optimal drying performance that is walk-ready within a day or less for faster return to service. Ideally a polyaspartic coating would dry in less than 2 hrs, have a viscosity of less than 1,000 cPs at 30 min with pendulum hardness greater than 75 seconds within one day and 120 seconds within two days. The DOE also tested for curing under different environmental conditions at 70 °F and 50 percent humidity to better understand how location would influence the formulation. Competitive technologies dry within 24 hrs and face difficulty offering low viscosity with high solids (90 percent or more) because of the viscosity of current polyaspartic resins.
We tested for viscosity, dry time and pendulum hardness (one day and seven day). Because properties of polyaspartic coatings are controlled by the structure or combination of resins, performance variables can include various curing rates and viscosity levels. Viscosity is very important for floor coatings to enable an evenly applied coating. This has led to a movement toward higher solids, which demonstrate a high capacity for even application.
Our formulations sought optimal properties through a mixture of polyaspartic resins, as detailed in Table 1. The first two formulations or targets blended close to 50:50 of Polyaspartic 1 and Polyaspartic 3. The additional three formulations used 30-50 percent of Polyaspartic 1 with the remaining amount being a mixture of Polyaspartic 2 and 3.
Figure 2 demonstrates that while our formulations build viscosity faster, they also dry faster than the control. Additionally, we determined that Polyaspartic 1 concentration is crucial to early walk-on times as detailed in Table 2. Early hardness development is critical to gaining enough strength to allow for walking on the coating in a rapid manner. For this reason we determined a high concentration of Polyaspartic 1 is critical to early walk-on time.
When testing high-solids (95 percent) formulations in low temperature (50 °F) and high humidity (80 percent), the formulations achieve cure in an optimal time period ranging from 80 min to 2 hrs (Table 3).
When testing for workability (Table 4), we noted that the system containing higher concentrations of Polyaspartic 3, such as in the Target 5 formulation, offers better workability or work life. This is due to the extremely low initial viscosity of the Polyaspartic 3 resin as supplied. Polyaspartic 3’s initial viscosity is 100 cPs as compared to other polyaspartic resins >1000 cPs. This lower viscosity allows for better reflow properties of the system while still having a rapid cure time. We also noted that higher concentrations of Polyaspatic 1 yielded poor workability due to the rapid reaction rate. The formulations can achieve similar work times to commercially available systems at higher solids.
Summary
Our formulation experiments demonstrate that polyaspartic coatings can achieve optimal drying performance that is walk-ready within a day or less for faster return to service. Additionally, the work time of these polyaspartic formulations is comparable to commercially available systems but at higher solids, which is an important property for improving application.
Polyaspartics offer specifiers, contractors and facility owners adjustable cure times for faster return to service, thick, high-build finishes, ultralow VOCs and low-odor systems that are not greatly impacted by ambient temperature. When combined with the availability of color options and protection from wear, chemicals and staining agents, polyaspartic technology provides a proven solution for concrete coatings.
As for the Grand Canyon, it is going to have to continue to fend for itself.
Author Note
The author would like to acknowledge the contribution of Tina Kasardo and Peg Kendi to this work, who designed and executed the experiments leading to the results published in this paper.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





