Special Clays Deliver Key Advantages in Rheological Modification

There have been significant advancements in the use of special clays as rheological additives in paint and coatings systems. Special clays such as sepiolite, attapulgite and smectites comprise a particular silicate subgroup in the clay family. All of them, under the appropriate processing conditions, produce gel structures that are able to behave as shear thinning and thixotropic fluids. These properties make special clays unique in comparison to other rheological additives, improving specifically the stability and application properties of these systems. These special clays deliver good anti-settling and anti-sagging characteristics for paints and coatings, either in water or solvent systems.
Special clays also provide special absorption and adsorption characteristics. Absorption, surface activity and morphological factors allow the design of special functionalized additives that will work in a wide range of base systems and will provide new functions beyond the standard improvements that are seen in typical rheological additives.
Tolsa’s PANGEL® series of rheological additives - based on high-purity sepiolite technology - are used for water-based and solvent-based paints and coatings.
Special Clay Options
Special clays are a subgroup of the phyllosilicates mineral-ogical group that includes minerals used extensively in the industry as additives for a variety of different applications. The most widely known members of this group are sepiolite, attapulgite and smectites, who share a variety of chemical and structural characteristics. Despite sharing some basic structural details, they are different in the way in which the block units are spatially distributed. This makes a fundamental difference between sepiolite and attapulgite, with an acicular morphology, and the subgroup of smectites with predominant lamellar shapes.1 Morphological differences and the surface characteristics of the individual particles play a fundamental role in the interaction with the water and cations, and the creation of gel structures. These gels must be capable of retaining solids and flow properly under stress, without affecting the system chemically or physically.
Smectites are negatively charged aluminum-magnesium silicate blocks that are linked together through cations as sodium or magnesium. These blocks have a lamellar morphology and are normally arranged forming multiple stacks of many layers. Meanwhile, in sepiolite or attapulgite, the extension of the silicate basic unit is limited in one of the space directions and creates a discontinuity that is the origin of the acicular shape (Figure 1).
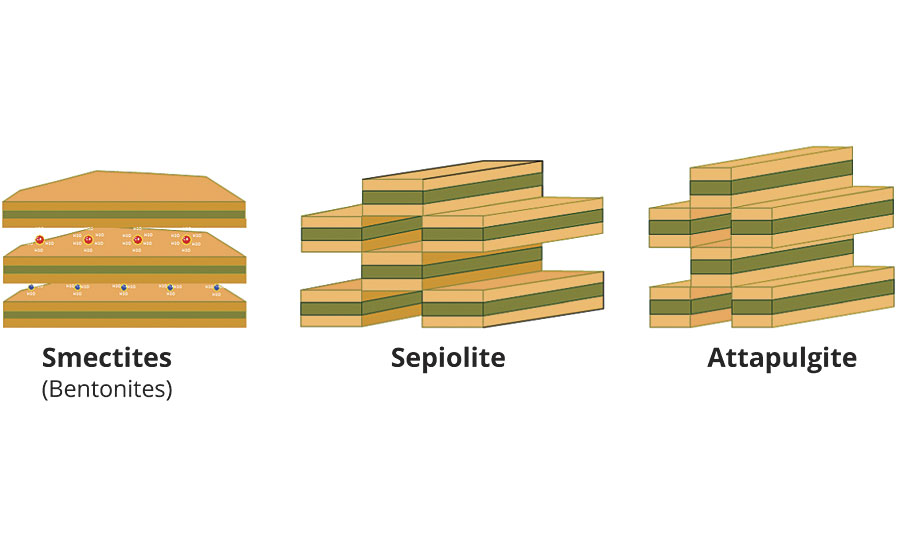
Magnesium silicate units in sepiolite and aluminum-magnesium in attapulgite are linked together through covalent bonds. Therefore, both minerals in contact with water maintain a very low electrical charge in contrast with the relatively high negative charge of smectites. Another important difference created by this morphology is the great surface area and hydroxyl (OH) density in both minerals – both key properties in their rheological behavior. This characteristic also allows both minerals to maintain high stability at high temperatures (up to 300 ºC) and extreme pH.
Tolsa’s product range is based on a wide variety of special clays, including sepiolite, attapulgite, montmorillonite and saponite, with each sharing the capabilities of easy build up, shear thinning and thixotropic gel structures. A key feature of PANGEL technology is its ability to yield fewer defects when incorporated into a coatings system, thus providing a competitive advantage for the end user. PANGEL products are characterized by the “3S” benefits they deliver: settling, sedimentation and syneresis.
Special Clays are Established Rheological Modifiers
Special clays, particularly smectites, are well known for their use in rheological modification since the 1940s, when J.W. Jordan2 developed the first organically modified smectite with enhanced hydrophilics for use in oil-based systems. This capability allowed the industry to get good dispersion of the flakes and conforming aggregates in their natural state. This advancement enabled these particles to create a tri-dimensional structure that produces a strong interaction with the solvent and other components in the coating system.
Morphological factors along with surface physicochemistry are responsible for the fast construction of a gelling structure.3 They are strong enough to maintain in suspension solid particles and weak enough to break down under the shear forces developed during application of the coating. This process is fully reversible and occurs in all three groups of clays. It is responsible for the shear thinning (pseudoplastic) and thixotropical behavior of fluids with these silicates.
Figure 2 compares the rheological behavior of the main families of rheological additives for waterborne systems. The data shows that special clays maintain the strongest pseudoplastic behavior in comparison with associative thickeners and traditional cellulosic or ASE thickeners. Special clays generate high viscosities at low shear stress where gravity forces are operating and affecting properties during settling of fillers and pigments, including sagging behavior and gel contraction processes. However, viscosity declines very fast as the shear rate climbs, therefore facilitating easy paint application and pumping operations. In comparison, the contribution of special clays to paint consistency and viscosity in water-based coatings is very small. Ideally, a flexible rheological system will combine special clays and organic thickeners with a less pseudoplastic behavior. Newtonian thickeners are recommended to control high-shear-rate processes in roller applications to avoid spattering, while clays will control sag and settling.

The second rheological characteristic, thixotropy, is important in order to attain good sag resistance while not adversely affecting the levelling behavior due to the right recovering speed of the gel structure.
All these characteristics are also maintained in solvent-based systems. In order to attain these rheological characteristics, it is important to allow the dispersion of hydrophilic particles in organic media. This is possible if the surface of the clays is treated with surfactants, normally quaternary ammonium salts that provide the appropriate polarity. In this way, the particles can be dispersed, thus creating a gel structure similar to the one built in water systems.
An Overview of Sepiolite, Attapulgite and Smectites
All special clays maintain common rheological properties, however the differences in basic structural and chemical characteristics introduce interesting variations that allow the formulator to provide the optimum additive in individual cases.
An important part of the dispersion process of smectites involves the swelling process of the interlamellar cations. This swelling occurs by means of the solvation of the cations that pull the particles apart. This makes it easier to disperse the particles and occurs in both water and solvent systems. In acicular clays, there is no swelling process, and the gel structure is obtained by the application of mechanical forces that disperse the particles, creating a randomly intermeshed structure. Microfiber length and the density of hydroxyl (OH) groups on the surface and their capacity to establish hydrogen bonds also create differences between the strength of the gel structure.
The result is that gels of sepiolite tend to be stronger in coating systems with higher solid content, but there still must be solvent available to allow a mechanical dispersion. In these cases, the anti-settling capacity will be at a maximum. In solvent-free coating systems, attapulgite is recommended because its particle sizes are smaller. More high-swelling smectites are also recommended. These two options used in solvent-free systems will provide an easier dispersion and a gel structure that is better formed.
Meanwhile, in the presence of water, the solvation process for smectites results in higher water retention, unlike sepiolite and attapulgite, which demonstrate faster water evaporation.
Special Clays Find Success in Coatings
Special clays have found effective use in water-based and organic coating systems. Today, waterborne paints utilize organic thickeners to control consistency of the paint with a high degree of efficacy and performance. However, products based on smectites, attapulgite or sepiolites are used to enhance specific properties of the paint combined with other organic additives. The most common combinations are with cellulose ethers, mainly HEC, and also with HEUR rheological additives.
In this case, a small dosage of special clays ranging between 0.1-0.5% helps to improve settling or sagging problems in paint. Also, they can be used to control syneresis during storage or to avoid sedimentation in pigment pastes.
Finally, special clay additives are particularly effective improving the workability and applicability of thick and viscous coatings due to the shear thinning behavior that can be provided in this type of system.
In solventborne or resin systems, the use of organoclays, particularly surface-coated hectorites, montmorillonites, or sepiolites, is more extensive. In water-based paints, special clays such as sepiolites are used as the main thickener, and are considered as a flexible and valuable tool due to the wide offering that can be found in the industry.
Every different clay and surfactant allows the construction of strong and applicable gel structures for production of a variety of paint and coating systems. Using pregel or direct addition procedures, they allow for easy handling in terms of production. These types of additives combine the capacity to control the system viscosity while anti-settling and anti-sagging characteristics are maintained and reinforced. With the exception of clear coats, most solvent-based systems can use special clays for controlling rheological characteristics.
Summary
Special clays have successfully demonstrated their use as rheological additives in paint and coatings systems. Sepiolite, attapulgite and smectites make up a particular silicate subgroup in the clay family. All of them, under the appropriate processing conditions, produce gel structures that are capable of behaving as shear thinning and thixotropic fluids. These properties make them unique, distinguishing them in comparison with other rheological additives used in paints. They are also useful in providing good anti-settling and anti-sagging characteristics in paints and coatings either in water or solvent systems.
The different morphologies of smectites, lamellar and sepiolite-attapulgite, acicular, allow the formulator to get the right rheology for a range of application needs.
References
1 Álvarez, A.; Santarén, J.; Esteban-Cubillo, A.; Aparicio, P. Current Industrial Applications of Palygorskite and Sepiolite. In: E.Galan and A.Singer, editors: Developments in Clay Science, Vol. 3, Elsevier, Amsterdam, The Netherlands, 281-298 (2011).
2 Murray, H.H. Applied Clay Mineralogy” Developments in Clay Science, Vol. 2, Elsevier, Amsterdam, The Netherlands, p.125. (2007).
3 Mezger, T.G. The Rheology Handbook. 3rd revised Ed. European Coatings Tech Files. Hanover, Vincent Network, p. 64 (2011).
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






