Additives for Paints and Coatings
Current State and Future Dynamics
I’ve been in the paint and coatings industry for 50 years this coming September, and have been privileged to be a close observer of the “ins and outs” of the paints and coatings value chain from a variety of perspectives, including basic R&D, applied R&D, technology development, supply chain, production, and senior management. If there is one thing that paint and coatings manufacturers from my early career have in common with today’s producers, it is that they are constantly striving to turn out the cleanest, simplest, and easiest-to-manufacture formulations with the least number of raw materials. That’s a long time for such a basic set of requirements to remain unaltered, and it is reasonable to assume that this is because it is a sensible goal to set for the entire organization. Nonetheless, it is interesting to note that the request to make formulations “cleaner,” “simpler,” and “easier-to-manufacture” is an ongoing organizational request 50 years later. Isn’t it reasonable to expect that formulations would have become “simplified” over that span of time? That prototypes would have long ago been created for all paint and coatings producers, so that their formulations would always contain the minimum number of raw material components, used at objectively optimum levels so that each formulation could be reproduced exactly the same way, at the minimum cost, with the same production steps, and final physical and chemical properties on an on-going basis. …?
…And for certain types of products, such as any number of “off-the-shelf” architectural paints, this is largely the case. For others, however, I fail to see any difference, over the course of 50 years, in the overall composition, number of chemical components, special instructions for addition of certain chemicals (often involving pre-mixing “Component A” with “Component B,” and sometimes even with “Components C & D,” as well), post-addition of various additives to fix problems such as cratering, crawling, color float, blistering, etc., et al., ad nauseam…One really has a right to ask, in fact, why paints and coatings even contain “additives” at all these days – the past five decades of polymer engineering, pigment surface treatment changes, more advanced milling equipment, and more sophisticated and controlled milling procedures have been aimed at refining many properties of the final product, but one of the main goals has always been “to eliminate additives.” This doesn’t appear to have happened, however, and one has a very definite right to ask, “why not?”
To reply that “it is not a perfect world” seems a bit glib, but it really isn’t all that far from the truth, which is that, as Robert Burns would have it, “things go bump in the night,” and we either need to minimize potential problems from happening by formulating preemptively or rid our paints and coatings of those bumpy things after-the-fact. Let’s be honest about this – paints and coatings are surprisingly complex mixtures of chemicals, and the miracle is that they work at all, much less work so well that we can spray the inside of a beer-and-beverage (“B&B”) can at a rate of five cans per second (0.2 seconds/can) with no voids, pinholes (“holidays”) or other film defects, and run full-gloss, smooth-flowing appliance finishes on coil lines that are applying the coatings at rates of 700+ feet/min.
It’s really mind-boggling what paints and coatings are asked to do these days, in terms of application and performance properties, and – while all of the components in the coatings system are responsible for the final result – none are typically as critical as that class of chemicals that we call “additives.” These are the mysterious materials variously referred to as “Snake Oil,” “Fufu Juice,” “Fairy Dust,” and any number of other colloquial variations, all of which are intended to convey the fact that certain materials seem to have magical powers when added to paint in small amounts, whether during the manufacturing process or following completion of the batch of paint when it is discovered that some property or properties need to be “tweaked.” So, what are these manufacturing marvels called “additives,” and what do they have (or do not have) in common? First of all, let’s take a high-level view at the global additives market, where we see at a glance that, while additives comprise slightly less than 4% of the weight of all of the paint sold around the globe, the value of those additives is 13% of the raw material cost of those coatings, indicating that they are considered to be significantly value-added components by the world’s coatings producers (Figure 1 and Figure 2).
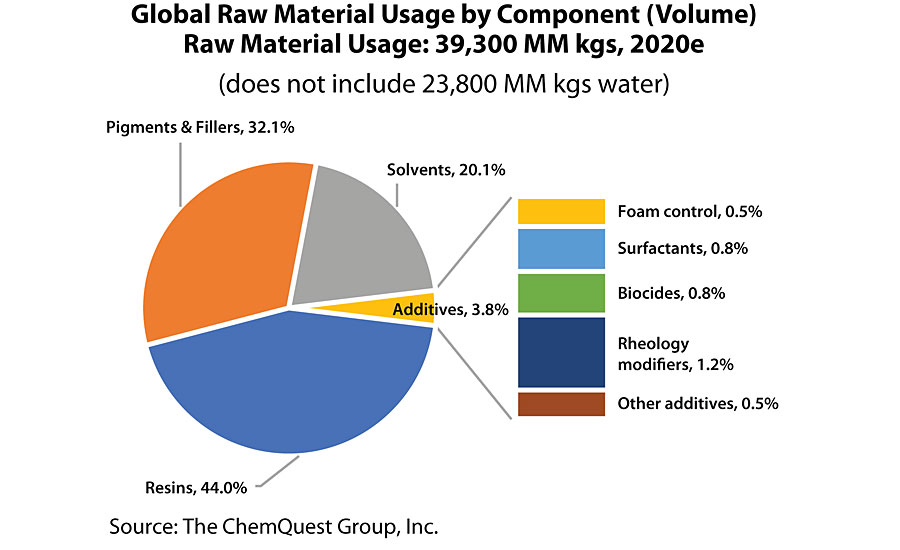
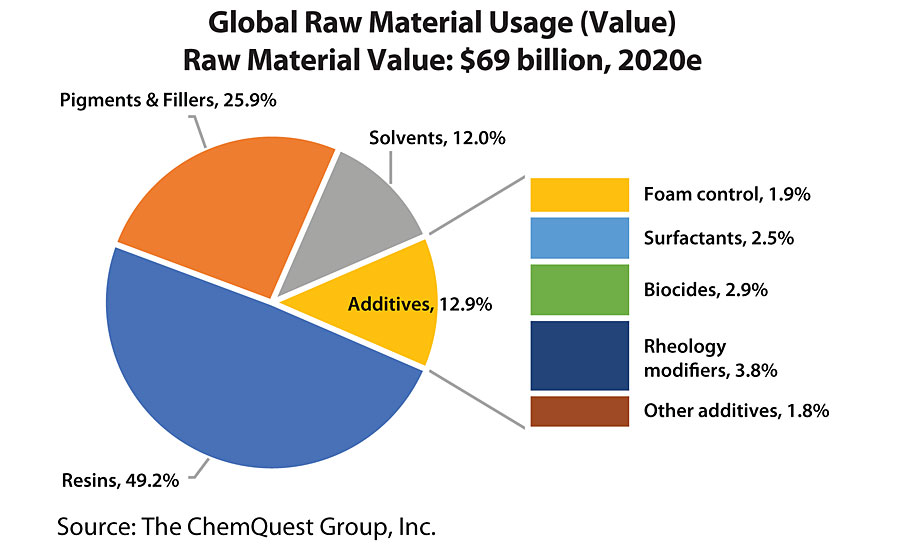
It is telling that, using the conventions of the World Coatings Congress, the “additives” are divided into just five categories, the smallest of which is “Other Additives” – yet it is precisely this category that contains the bulk of the different types of additives used in paints and coatings. Remember: The term “additives” is just a catch-all phrase for chemicals that typically have the ability to react either permanently or transiently with other chemicals in a paint and coatings formulation or the surface of the substrate onto which the paint is applied. Before getting into the details, it is helpful to consider a “family tree” approach to discussing additives. The following classifies the use of additives during the life of the coating, starting with how they aid in the production process, the role they play in maintaining the stability of the coating in its container, the benefits they provide during the application process, their role during the film forming process, and, finally, the functionality that they can provide during the lifetime of the coating (Figure 3).
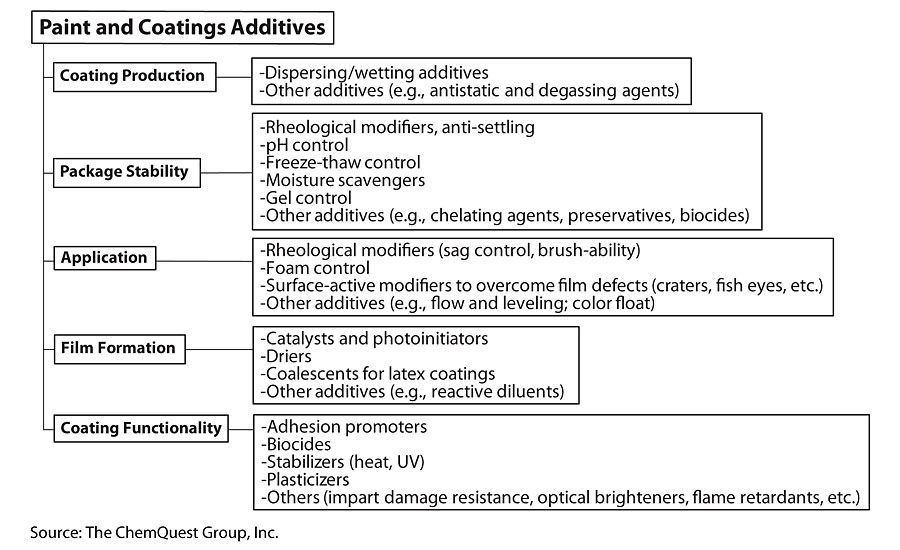
Now we are ready for some, but not an overwhelming number, of the details.
Coatings Production
The main category here are dispersing/wetting agents that are used in the most energy-consuming process in the paint making process. Pigments are supplied as agglomerates, which need to be separated and surrounded by a liquid medium. These materials are used to aid the dispersion of all pigments – and are key to the full color development of chromatic pigments. They also help to prevent settling of the dispersed pigments; help to prevent flocculation (pigment crystal corner-to-corner attraction), agglomeration (crystal edge-to-edge attraction), and aggregation (crystal face-to-face attraction).
Depending upon the pigments that need to be dispersed, dispersing/wetting additive molecules will have different chemistries, because they should ideally be able to have one part of the molecule attach to the pigment and the other end become affiliated with the polymer. In this way, the wetting agent will “link” the pigment particles to the polymer. Wetting and dispersing additives are often based on fatty acids, with an anionic, cationic or non-ionic end group that has an affinity to the pigment surface. These are typically low-molecular-weight polymers and are primarily used to stabilize inorganic pigments. Organic pigments are another story. Polymeric additives are preferred, because these additives contain a very large number of pigment-loving groups. These polymeric additives need to be compatible with the organic binder used in the formulation, otherwise the additive polymer chains collapse, and stabilization is lost. The compatibility of highly polymeric additives with various coating binders is considerably more restricted than that of a low-molecular-weight variety.
In aqueous systems that are based on emulsion binders, the dispersing/wetting additives are often ammonium salts of polycarboxylic acids and similar materials.
Package Stability
Producing the coating in an efficient manner is important, but, just as important, is making certain that the coating is stable in its package (i.e., gallon cans, 5-gallon pails, drums, totes, storage tanks). In the manufacturing process, much effort is made to disperse the pigments, but once the coating is filled into its container these same pigments will naturally begin to settle since their density is greater than the density of the liquid medium. Counteracting this natural tendency is often accomplished by introducing an additive that creates a thixotropic solution. A thixotropic solution is shear thinning, i.e., when not under shear, such as when a coating is sitting undisturbed in its package, the viscosity is higher than when shear is applied during agitation and application. There are several ways to create thixotropy, but three of the more common approaches are through the use of castor oil derivatives, organoclays or a fumed silica.
For waterborne coatings, pH control is critical, and several organic amines, ammonium hydroxide and sodium/potassium hydroxide are in common and widespread usage. For industrial coatings that utilize an acid catalyst for accelerating the crosslinking of a condensation reaction, using a small amount of neutralizer (i.e. “blocking the catalyst”) will minimize any reaction in the package.
In industrial coatings two-package (“2K”) urethanes, the presence of water in the isocyanate portion has a significant deleterious effect. Additives are used to scavenge acids, moisture or other chemicals (yes, water is very definitely a chemical), formed during storage, that would otherwise interfere with the performance of the paint or coating.
Application
Now it’s time to apply the coating. There are many means of applying paints and coatings, and additives play roles in two key areas: facilitating the application of the coatings, and overcoming issues associated with substrates.
During application, there are many opportunities for air to interact with the liquid paint which, if not addressed, can lead to foaming that creates film defects. Defoamers are used to ameliorate this condition, which is of far greater concern when waterborne coatings are being applied, compared to solventborne coatings.
Rheology control is another critical factor when applying coatings. Typically, a coating applies best when the viscosity is low. During a spray operation, the atomized droplets are smaller and sinter more effectively when the viscosity is low. Low viscosity, however, leads to sagging. The ideal coating is often one that shear thins during application (i.e., the viscosity is reduced during the application process), but quickly – although not immediately – recovers its viscosity to prevent drips, beads, sagging, etc. A host of additives are available, and function in different ways, depending on the specific additives and the resin system in which they have been incorporated.
During the application process, delivering the paint from the package onto the part is a complex process. The substrate itself presents a host of difficulties. If a metal substrate isn’t clean and pristine, surface defects such as cratering, crawling, and pin-holing are very likely to occur. Additives are typically incorporated into the paint to overcome these issues. Generally, the additives’ role in this case is to lower the surface tension of the coatings to compensate for low-surface-energy contaminants on the substrate, such as oil, wax, grease, etc.
As the film forms, “flooding” and “floating” may occur. Both conditions are surface appearance manifestations when two or more pigments are not uniformly distributed throughout the film during the drying/curing process. The former must typically be dealt with during the dispersion process, by changing either – or both – the dispersant(s) incorporated, and the method of mechanical dispersion used. The latter can generally be addressed via the use of silicone and/or acrylate surface-active agents.
Film Formation
Once the coating has been successfully applied to the substrate, film formation occurs. There are three principal types of film formation – air-drying, ambient-temperature curing and baking – and each has its own unique group of additives that are routinely used. In the case of bake coatings, acid catalysts are generally used to accelerate the reaction between the backbone resin and the crosslinker. In 1K blocked-isocyanate ambient-cure reactions, the catalyst is typically a metal salt. Metal chelate catalysts are often used in 2K isocyanate reactions. UV-cure coatings have unique chemistry, and catalyst is described as a photoinitiator.
For air-dry alkyd coatings, the catalyst is called a drier, and these are materials generally based on metal carboxylates or iron-ligand chemistry. Here the catalyst is accelerating the reaction between the alkyd resin molecules’ double bonds found on the oil chain.
Emulsion-based coatings utilize a coalescent that optimizes the ability of the emulsion particles to combine with each other to form a contiguous network – a process called coalescence.
Coating Functionality
What has been discussed so far has to do with producing paints and coatings, applying them, and helping them form a film. Additives play a crucial role along the way, but a large group of additives are used to impart a variety of functional properties to the coating. Of course, the resin system and the pigments play an important role in property development, but they can only go so far. Small amounts of additives can confer major beneficial properties to the coating film, either not achievable – or not as easily achievable – through other means, and there is a host of materials from which to choose:
- Biocides that serve as anti-fouling agents in marine environments, which prevent marine life, such as barnacles, from attaching to boat hulls or free-standing structures installed in a marine environment.
- Similar materials that prevent the propagation of bacteria, viruses, and fungi both “in the can” of (typically waterborne) paint, and sometimes to render the surface of the cured/dried paint “anti-bacterial,” as well.
- Rust preventatives that keep rust from forming in metal cans of waterborne paint – and prevent flash rusting from rearing its ugly head while waterborne paints are being applied to ferrous metal surfaces, such as cold-rolled steel.
- UV absorbers and Hindered Amine Light Stabilizers (HALS) that enhance exterior durability
- Adhesion promoters
- Additives that make the coatings more easily cleaned or allow graffiti to be readily removed
- Flame retardants
- Waxes that reduce coefficient of friction (COF) on the surface of dried/cured paints and coatings
- Plasticizers that boost flexibility
- Additives that minimize blocking, marring, scratching, and burnishing. These materials prevent coated articles from sticking to each other, a phenomenon known as “blocking,” a condition only too well-known to homeowners who push the couch against the newly-painted wall prematurely, or who close a recently-painted (but seemingly “dried”) window, only to discover that it will not re-open.
- Materials that produce unique surfaces, such as hammer finishes, wrinkles, and textured surfaces
- Materials that produce haptic surface properties
- Additives that produce either hydrophobic (water-beading) or hydrophilic (water-sheeting) effects, which can help painted surfaces remain clean
- Additives that depress gloss/sheen/DOI without the use of large amounts of traditional flattening pigments
- Materials that reduce static attraction for airborne, particulate matter
What types of chemicals, one might logically ask, bring about such a panoply of properties and effects when incorporated in “additive amounts” (typically ≤1-3%, on formulation weight, and often only fractions of a percent, depending upon the type of additive and the technology type of the backbone polymer) into paints and coatings? The answer to that question is by no means simple, because the additive space is vast, and much of it is proprietary. It is, therefore, reasonable for me, based upon my own experience, to opine that the following general types of chemistry are very likely to be the basis of the majority of all additives typically used in paints and coatings. This list is neither comprehensive nor does it attempt to address the many molecular variations that can be wrought within these general chemical classes, and which are typically responsible for producing the specific effects for which a commercial additive is sold:
Wetting/Dispersing
- Alkylphenol ethoxylates (APEO – in rapidly decreasing usage, due to health and environmental concerns)
- Phosphate salts and esters
- Aromatic ethoxylates
- Polyamine amides and similar
- Polyether amines
- Carboxylated fatty acids
- Soya lecithin and derivatives
- Sulfonates
- Sulfates
- Block copolymers
- High-molecular-weight polyurethanes/polyacrylates
Rheological Modifiers
- Hydrophobically modified alkali-swellable emulsions (HASE)
- Hydrophobically modified ethoxylate urethanes (HEUR)
- Cellulosics (both traditional and microfibrillated)
- Organo clays
- Fumed silicas
Foam Control, Surface-Active Modifiers, Flow and Levelling Agents
- Cellulose acetate butyrates (CAB)
- High-molecular-weight functional polymers – e.g., polyesters, polyacrylates and polyurethanes
- Benzoin
- Polysiloxines (silicones)
- Mineral oil
- Mineral spirits
Catalysts
- Sulfonic, carboxylic, and phosphoric acids
- Dibutyltin dilaurate (DBTDL), and dibutyltin diacetate (DBTDA)
- Organic peroxides
- 1,4-diazabicyclo[2.2.2]octane (DABCO)
Driers
- Metal carboxylates (naphthenates, neodeconoates, octoates)
- Iron ligand chemistry
Coalescents
- Propylene glycol
- 2,2,4-trimethyl-1,3-pentanediol monoisobutyrate (Texanol™)
- Various benzoates
Coating Surface Modification
- Waxes – high- and low-density polyethylene; polypropylene; Fischer-Tropsch; PTFE; microcrystalline waxes
- Polymethyl urea
- Fluorosurfactants with ultrashort fluorinated alkyl chains – C2, C3, C4
- Polyethers and polyacrylates
Adhesion Promoters
- Organofunctional silanes
- Organometallic salts, esters and chelates (titanates, zirconates)
- Functionalized copolymers
Biocides/Biostats
- Silver-bearing moieties, especially nanoscale
- Isothiazolinones
- Guanides
- Benzimidazoles
- Quaternary ammonium compounds
- Phenols
- Carbamates
Stabilizers
- 2, 2, 6, 6-tetramethylpiperidenes (HALS)
- Benzotriazoles (UV absorbers)
- Barium-zinc, organotin, and bisphenol-type heat stabilizers
Plasticizers
- Various phthalates (diisononyl-, diisodecyl-, dimethyl-, etc.)
- Polyester oligomers/polymers
- Epoxidized soybean oil
As one can see from just a glance at the preceding list of different types of materials recognized as “paint and coatings additives,” the majority of them are accounted for, in terms of global usage and value, under the category of “Other Additives”. Why so many materials in this one category? Does the usage of the additives in this grouping vary by end-use? By global region? By some other factor or factors?
A quick look at the usage of “Other Additives” by global regions speaks volumes, insofar as APAC uses a far greater percentage of the additives listed under “Other Additives” in its paints and coatings than either North American or Europe (Figure 4).
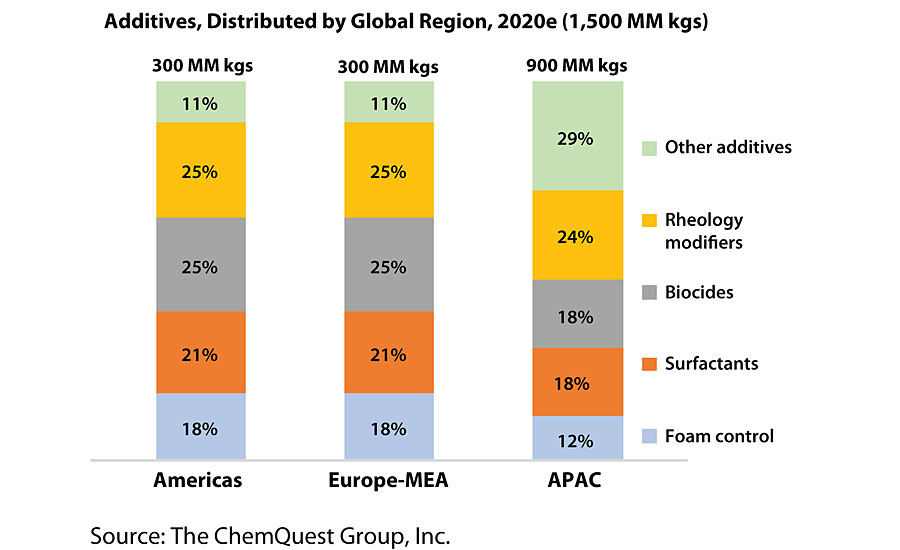
Since architectural coatings typically use larger amounts of a fewer number and type of additives (10% of total formula weight vs. 4-5% for most industrial OEM and protective coatings), it is probably reasonable to posit, from the APAC data, that this region of the world produces larger amounts of industrial coatings, and lower amounts of architectural coatings, than is the case in either the Americas or Europe. (So that the reader is not left in suspense, I can confirm that this is, indeed, a true supposition.)
At the End of the Day
Gaining an understanding of the additives space is not a trivial undertaking, and can only be accomplished by “hands on” experience in the art of formulation, although it certainly helps that many of the additive producers can provide at least a degree of assistance in matching the additive to the properties that are desirable in the coatings product. Nonetheless, the sheer profusion of potential additives, from a large number of companies – global, national, regional, sub-regional and sometimes even local – can defeat a well-intentioned formulator almost before he/she can even get a start on a new product development, or solve a performance issue with an existing product. Is the goal to take a new additive that claims to be able to provide a highly desirable property (or set of properties) to paints and coatings and use it to drive the composition of the final formulation? Or is the composition of the already existing formulation going to dictate the range of additives to which the formulator can turn for help? Or will a unique problem that may only occur once in a hundred batches, with an unknown cause, dictate a selection of additives to address the problem? Or will the formulator be limited to “what’s on the shelf” or “what’s on the Approved Additive List” within the organization? When it comes to pigment, resin and solvent use, there may be choices, but those choices are generally well-contained, and the list of any given type of paint component is not all that long. This same thing can be said about additives, as well – but with the caveat that the overwhelming number of available additives can only be “cut down to size” by someone who has had a lot of experience working with them, under a variety of conditions.
This brings us, yet again, back to the “experience” aspect of using additives, and the importance of understanding that they are all chemicals that interact in specific ways in paint and coating systems. Because this is a complex field of contenders, it is typically easiest for a formulator to adopt the attitude that many physicians take, which is to impose an arbitrary limit on the number of drugs that they will prescribe to 50-60 with which they have had “success” over the years. Formulators do this, as well, and most of the time, this approach works well. It is also the reason why, when an employee is hired from a competitor, he/she often ends-up being a local hero, because “outsiders” are able to solve certain long-running problems by introducing different additives into the formulating and production process from those that are “tried and true” within the company. Each paint and coatings producer (or personal care product producer, adhesive producer, lubricant producer) develops a “formulating philosophy” over the years, and part of this philosophy includes the use of certain raw materials but not others. Each producer chooses, over time, the pigments, solvents, polymers, and additives that it will use to make its products, and only occasionally are the company formulators ever allowed – or even tempted – to wander outside of the protective circle drawn around these materials, binding them to the formulating staff and production personnel. (Hence the fabled “stickiness” of additives, in general.)
Where from Here?
From where I sit, there are only two imperatives facing the use of additives in paints and coatings – one which will need to be addressed by the paints and coatings formulators and the other by the additive suppliers:
- Formulators need to keep plugging away at obtaining more – and greater – positive effects from the use of fewer additives, which will drive additive producers. …
- … To engineer newer materials that are truly polyfunctional, insofar as one new additive can replace two or three existing additives.
These twin desires - to use fewer raw materials to achieve multiple properties in paints and coatings - reflects an ongoing trend that will continue to drive the development of new additives that can fulfill more than one role in a coating, if for no other reason than the fact that global consolidation in the paint industry requires raw material rationalization. This is just a fact in today’s M&A climate, and purchasing and inventorying the minimum number of high-value raw materials simply makes good sense. Everyone wants some different combination of properties delivered by so-called “multi-functional” additives, whether to improve pigment dispersion at the same time as it prevents foam during the application of finished paint or to suppress gloss without viscosity increase, while providing haptic properties to the finished film. While it is true that many currently available additives lay claim to being multi-functional, there is still a lot of work to be done to really achieve the goal. I have only personally worked with a small number of additives that were truly multifunctional – AMP, polymethyl urea, sodium dioctylsulfosuccinate and perhaps a few others – and even they only act in a multi-functional fashion in certain systems, under certain circumstances. There is plenty of room for innovative thinking, by both the additive producers and the coatings formulators, with regard to the subject of polyfunctional additives for paints and coatings. The challenge is here. . . .and the time to accept it is now.
For more information, contact George Pilcher at 513-262-8727 or e-mail gpilcher@chemquest.com. Or visit ChemQuest’s website at www.chemquest.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!









