Polyurea Spray-Applied Systems for Concrete Protection
Polyurea coatings combine extreme application properties such as rapid cure (even at temperatures well below 0 øC), and insensitivity to humidity, with exceptional physical properties such as high hardness, flexibility, tear and tensile strength, and chemical and water resistance. This results in good weathering and abrasion resistance. The systems are 100% solids, making them compliant with the strictest VOC regulations. Due to its specific curing profile and exceptional film properties, the polyurea spray coating technique has been introduced into many areas, including corrosion protection, containment, membranes, linings and caulks.

Definitions
The term 'polyurea' has been wrongly used in the past. Urethane coatings chemistry can be divided into three segments: polyurethane coatings; polyurea coatings; and hybrid polyurethane/polyurea coatings. All three are linked to different isocyanate reactions (Figure 1). Each of these segments deals with systems that can be aromatic, aliphatic, or a blend of both aromatic and aliphatic. Pigments, fillers, solvents and/or additives can be introduced to all of them.
The water/isocyanate reaction also produces urea-groups at the end of the process. However, this reaction should not be considered to be a polyurea reaction as the mechanism is a two-step process, which is controlled by the much slower isocyanate water reaction and produces carbon dioxide.

The Polyurethane Landscape
The choice between the different polyurethane (PU) technologies is based upon different parameters (Figure 2). Polyurethane presents the best compromise between cost and quality, but is limited by the application performance. The polyurethane system is susceptible to blistering when the substrate contains more than 5% humidity. This is due to competition between hydroxyl-polyols and water for the reaction with an isocyanate group. The humidity content of the environment and the application temperature are limiting factors for polyurethanes and other chemically reacting systems.Hybrid systems already have a larger scope for application conditions, but the presence of catalysts in hybrids makes them more sensitive to humidity than "pure" polyurea systems. Moreover, because the catalyzed polyol/isocyanate reaction behaves differently from the amine/isocyanate reaction to changing application temperatures, the system becomes less robust.
Polyurea can be used in extreme conditions. When it is used on substrates almost saturated with water, polyurea will not provoke blistering nor will blistering occur when the air contains high amounts of humidity. Even at very low temperatures (as low as -20 deg C) the polyurea coating will still cure. Polyurea coatings combine high flexibility with hardness. They are the most suitable coatings when the following is required:
- high curing speed;
- application under high humidity and/or at low temperatures;
- extreme abrasion resistance;
- impermeable membranes;
- high thickness build up;
- chemical resistance.

Applications for Polyurea Coatings
A good understanding of the properties of polyurea spray coatings is required to specify the right application. Table 1 provides a general overview of the physical and chemical properties that can be expected of polyurea spray products. Polyurea systems are known to be very tough. They combine high elasticity with high surface hardness, resulting in very good abrasion resistance.The market development started in the United States, followed by Asia, with very strong growth during the second half of the 1990s. In the first stage of development, polyurea was used as a protective layer over polyurethane insulation foam for roofing applications. In Europe, the polyurea spray coatings market only started to develop in the last few years.
The broad window of application conditions, with a high tolerance for humidity, both from the environment and from the substrate and temperature, makes polyurea a very suitable coating for concrete in construction applications such as roof repair, containment liners, membranes, car park decks, bridges and offshore. The high abrasion resistance leads to its application in liners for truck, bulk transport wagons, freighters and conveyor belts. Table 2 presents an overview of the application fields where polyurea is chosen based on one or more of its unique application and/or film properties.

Raw Materials
A polyurea spray coating formulation consists of five different elements: the isocyanate component; (reactive) diluent; polyetheramines; chain extenders; and additives, fillers and pigments.
Isocyanate
Since the most commonly used isocyanate is diphenylmethane diisocyanate (MDI), this paper focuses on MDI-based products. Aliphatic systems can be used where UV stability is an issue.Standard polyurea spray coatings use MDI prepolymers with an NCO content of 15 to 16%. In this NCO range, a good compromise between viscosity of the material and the reactivity of the system is obtained. Lower-NCO prepolymers have a higher viscosity, but give higher elasticity and slower reactivity. Higher-NCO prepolymers are lower in viscosity, which provides an effective mixture of the two components. However, they become much more reactive, with the risk of building up more internal stress. Higher-NCO prepolymers will be used if higher surface hardness is needed. Table 3 provides an overview of the main properties of the MDI prepolymers used for polyurea spray coatings in Europe.
Diluent

- improved shelf life of the isocyanate-prepolymer;
- a compatibilizer for the mixing of the two components in the mixing chamber of the spray gun;
- a viscosity reducer for isocyanate-prepolymers;
- improved leveling of the applied film.
In applications where contact with water cannot be avoided, the use of propylene carbonate should also be limited, as propylene carbonate is completely miscible with water, and unreacted propylene carbonate could be extracted, increasing the water permeability of the film.
Huntsman Petrochemical Corp. owns certain patents relating to the use of propylene carbonate in polyurea elastomers. Other solvents or viscosity reducers can be used if they are compatible with the isocyanate component. They may be considered as a VOC. However, they will increase the shrinkage effect.
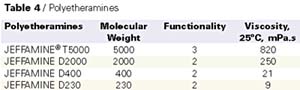
Polyetheramines
The amine blend used in polyurea spray coatings is a mixture of polyetheramines and chain extenders. The main component of the resin blend is a mixture of amine-terminated ethylene oxide and/or propylene oxide polyether with molecular weights varying from 200 to 5000 g/mole. The primary amine groups provide a very fast and reliable reaction with the NCO groups of the isocyanate component. Table 4 presents the properties of the polyetheramines commonly used in polyurea.
Chain Extenders
Diethyl-toluenediamine, or DETDA, is the standard chain extender used in aromatic polyurea spray coatings. DETDA contributes to the hard block and improves the heat resistance of the cured film. It is the most reactive amine in the resin blend but, because of the phase separation during the curing, it controls the reaction mechanism and makes it possible to spray a polyurea film.
Other chain extenders like dimethylthio-toluenediamine (DMTDA), N,N'-di(sec.butyl)-amino-biphenyl methane (DBMDA) or 4,4'-methylenebis-(3-chloro, 2,6-diethyl)-aniline (MCDEA) slow down the reaction significantly. Table 5 lists various chain extenders and their characteristics. Significantly slowing down the reaction also means that the competition with the water reaction becomes more important and precautions need to be taken.
Additives, Fillers, Pigments
Depending on the application, solvents, additives, pigments and/or fillers are introduced to the formulation. Adhesion promoters like silanes are used to enhance the adhesion on steel and concrete. UV absorbers are used to slow down the yellowing effect of aromatic polyurea systems. Fillers are added to lower the raw material cost and/or improve the physical properties of the coating. The addition of pigment and/or fillers is limited because the viscosity of the two components at the application temperature has to be kept under control. Higher amounts of fillers and reinforcement fillers can be added to the system as a third component.
Product-Application Specifics
The most important element of handling polyurea coatings is the mixing. Good mixing will be obtained in a suitable mixing module by impingement with mechanical purge. The operational pressure and temperature of the products will also help to optimize the mixing efficiency.Due to the high cure speed of polyurea and the short mixing time, the products are mixed by impingement at high pressure. Indeed, for field applications it is preferable to formulate the products on a fixed 1:1 volume-mixing ratio. The pressure used in the field will vary between 150 and 250 bar. The viscosity of the products at application temperature ideally needs to be lower than 100 mPa.s, and the viscosity of the two components needs to be at the same level. The properties of these prepolymers can be found in Table 3. The viscosity of the resin blend at 25 deg C is approximately 900 mPa.s, dropping below 100 mPa.s at application temperature.
Experiments prove that polyurea films produced at 65 deg C, 70 deg C and 80 deg C have different properties, and these properties improve with increasing temperatures. The spraying equipment has improved significantly. The new spray equipment allows different temperature settings for the two components, ensuring an optimum mixing in the spray head. Other features are easier variable ratio settings; easy output control; and easy monitoring of application parameters.
The index of a polyurea system is typically kept at a slight over-index of the isocyanate in the range of 1.05-1.10. As the isocyanate group reacts to humidity, the excess isocyanate compensates for the 'loss' of isocyanate groups during storage and/or application. The film properties of the 1:1 volume ratio sprayed system were measured for an index variation between 0.90 and 1.15. The test results indicate that the film performs best at an index of 1.05 and higher. Below an index of 1.05 the results can vary significantly and become unpredictable, even for small index shifts.
Aspects of Spray Polyurea Technology
Polyurea application had some problems during the initial start-up phase, which are at the origin of the still-existing misconceptions about polyurea technology. These problems can be attributed partly to the lack of experience at the time of the technology introduction, partly due to the lack of adequate application equipment, and partly to the fact that this new technology could not be applied in the same way as the current coatings systems.Initially, polyurea spray coatings looked too easy to apply. Polyurea is very fast - the coating can be put into service immediately after the application, and the final properties of the coating are obtained only a few hours afterwards. Polyurea is not water- or temperature-sensitive, and is easy to formulate and produce. The first systems on the market were indeed very fast, with a gel time of less than two seconds, and initially a number of problems were linked to the reactivity of the systems.
At first, substrate wetting was a problem. This problem was linked to the development phase of polyurea with the use of extremely fast spray systems. Development programs focusing on adhesion on concrete, with polyurea systems presenting gel times of three to four seconds, resulted in cohesive adhesion failure in the concrete. In practice, to limit the risks under variable field conditions, a multi-layer system is applied, made of a primer and a topcoat.
A second problem noted in the field was the lack of intercoat adhesion. Lab tests, with times between coats of several weeks, have shown that intercoat adhesion is very good. When problems occur with intercoat adhesion, most of the time they can be related back to problems with the raw materials, the manufacturing of the systems or the spray equipment. Spray equipment problems, or a disturbance of the feeding of one or both components towards the mixing module, can cause poor mixing. Adapting the machine settings of the spray can solve this.
Due to the high reactivity of the systems, the surface quality of the sprayed film was initially very poor. Fine-tuning the spraying equipment was a first step towards solving the problem. The use of non-VOC reactive diluents and the development of new MDI prepolymers with higher 2,4'-isomer content resulted in perfect surface quality without compromising on working time.
The cost of polyurea spray coatings technology is seen as a barrier to entry. "Pure" polyurea systems are more expensive, when considering raw materials cost alone, but can be applied in areas where all other systems will fail or where they are not suitable. Also the initial investment in equipment is rather costly.
However, when estimating the capital cost for a project, polyurea is more competitive when both the processing time and the waiting period before the coated substrate is put back into service are included.
As discussed above, the success of the project is very equipment- and applicator-dependent, and we believe that the high entry barrier can only guarantee quality services from specialized and skilled operators.
Construction-Related Aspects
Polyurea spray coatings can be used under difficult weather conditions because the chemistry is very fast and they do not experience negative side effects caused by the presence of humidity. They also cure at temperatures below 0 deg C. However, when using polyurea coatings, a number of precautions still have to be taken.
If primers are considered, a good approach is to start with the evaluation of existing primers with known performance on the substrate. It is important to determine the adhesion performance of the polyurea coating on the primers and to check whether the application conditions of the primer and the re-coating conditions for the primer still work for a system with polyurea as a finish.
Concrete Surface Defects and Surface Preparation
The low cost, high strength and structural properties of concrete make it the material of choice for the construction industry. Some of the typical properties of concrete like the limited chemical resistance, dust release and porosity or permeability make it necessary to put a protective and/or decorative layer onto the surface.The preparation of the surface is extremely important. Depending on the surface quality of the concrete, one or more of the following actions needs to be taken:
- water jet and/or solvent cleaning;
- grit blasting;
- bughole and crack filling;
- repair layer of concrete;
- priming.
Steel Surface Defects and Surface Preparation
The life cycle of steel construction coatings depends largely upon the protective system put in place. The life of the protective coating itself is strongly dependent on the surface condition prior to the application of the coating. The protection of the substrate is mainly obtained by ensuring a good adhesion. Two adhesion mechanisms are possible:- molecular attraction of the interfacial forces from both the coating and the substrate,
- mechanical bonding or anchoring of the coating on the substrate.
On freshly grit-blasted, dust-free steel with a surface roughness SA 2 1/2 to SA 2 according to the specification ISO 8501-1, very high adhesion values can be obtained for polyurea, even without the use of primers.

Mixing Efficiency
The mixing efficiency of the application equipment is of vital importance. When formulating a system or modifying an existing system, it is necessary to verify the mixing efficiency constantly. Figure 4 shows the influence of changing the mixing on the physical properties of a formulated product. The tensile strength almost doubles from 14 N/mm2 to 23 N/mm2, the angle tear increases from 75 N/mm to 85 N/mm and the elongation increases from 390 to 430 per cent. In this case, the influence on the other physical properties is limited.
System Index Influence on Final Film Properties
Earlier experiments reveal that a polyurea coating needs to be formulated at an index above 1.00, meaning with a slightly higher amount of isocyanate-groups than amine-groups. At an index of 1.00 or lower, the physical properties of the coating become unreliable. Figure 5 demonstrates that most properties have very good values at indexes from 1.10 to 1.30. Above an index of 1.30 the performance tends to drop again.
Taking into account that, in practice, slight variations might occur in the application parameters, related to the precision of the spray equipment and variations in application conditions, it is safer to work at a minimum index of 1.10 to 1.15.

Filler Influence on Physical Properties
Adding fillers to a polyurea system can be useful for different reasons, such as a reduction in the raw material cost or improvement in physical properties. Inorganic fillers have a different hardness and some will abrade parts of the spray equipment more than others. The most sensitive parts are the mixing chamber and the nozzle of the spray gun. The filled systems need to be carefully filtered before packaging.Depending on the performance of the spray installation, the dosing of filler can vary. We added up to 40% of filler to the resin blend. The main limiting factor for the processing is the increase in viscosity of the filled component. This can result in difficulties with the pumping unit and/or poor mixing due to big differences in viscosity for both components.
Adding fillers improves the surface hardness, the angle tear and flexural bending modulus but has a negative influence on the elongation and the tear propagation or trouser tear (Figure 6).

Water Absorption
For corrosion protection, the main consideration for good performance is adhesion to the substrate. Further testing proved that, even with very good adhesion, the resistance to cathodic disbondment for some systems fails. Since cathodic disbondment occurs over a longer term and certainly is not a simple test method, we measured the water absorption over a period of 10 days at 80 deg C and added 3% sodium chloride to the water.
As can be seen in Figure 7, the unmodified standard polyurea spray coatings give only a limited protection against corrosion. The water absorption drops significantly with increasing the NCO-content for the prepolymer. Further fine-tuning of the pre-polymer resulted in water absorption being well below 0.5% after 10 days. Cathodic disbondment testing on this system gave very satisfying test results.
Anti-Skid Performance
Flooring, car parks and sports floors are key applications for polyurea due to its high abrasion resistance, good mechanical properties and insensitivity to blistering during the curing process in humid conditions.
One example is the outdoor, impact-absorbing playground flooring. The system tested is based on SUPRASEC 2049, formulated to be applied in a 1 to 1 volume ratio. The film properties are 70 Shore A hardness, 600% elongation and 13 N/mm2 tensile strength. The results in Table 6 show that it is perfectly possible to formulate a system, which complies with the antiskid needs of a flooring system, in this case for a flexible substrate.
Conclusions
Polyurea spray coating technology is different from other coating chemistries and can expand the application range of coatings to areas and conditions where other coating systems will fail.Polyureas are very suitable for construction applications. The fast curing makes it possible to use when only very short disturbance periods are allowed. The fact that the isocyanate/water reaction is not affecting the physical properties of the applied film, expands the use of polyurea to high relative humidity conditions and does not set such stringent limits on the water content of substrates like concrete. Although they slow down at colder temperatures, polyurea coatings still cure at temperatures where other chemistries fail.
The formulation of polyurea spray coatings has to be approached similarly to any other coating system. Careful selection of raw materials for fine-tuning of the formulation and evaluation of the system, in the sometimes difficult conditions where the coating is to be applied, is still necessary.
Polyurea spray coating technology means handling reactive chemicals. While handling the chemicals during their manufacturing, packaging and application, the correct protective clothing should be worn at all times.
Acknowledgements
The author would like to thank Stefan Priemen and Domien Berden for the application and testing of the polyurea samples, Wesley Verbeke for the support with the testing of the systems and all other Huntsman staff who helped to realize this paper.For more information, e-mail Marc Broekaert at marc_broekaert@huntsman.com.
References
1 Van Tongelen, J. Untapped Potential. Eur. Ctgs. J. January-February, 2001.2 Godinich, C. Polyurea: A Market Overview. Eur. Ctgs. J. October 2000, p. 54.
3 Primeaux, J.D. II. Fast-Curing Polyurea Spray Elastomers Rapidly Spreading in Commercial Use. Urethanes Technol. October-November 2000, p. 37.
4 Primeaux, J.D. J. II. Spray Polyurea Versatile High Performance Elastomer for the Polyurethane Industry, Polyurethanes 89 - Proceedings of the 32nd annual technical/marketing conference, SPI, San Francisco, October 1989, p. 126.
5 Perez, A.; Johnston, J. A. Performance and Processing Enhancements of Aromatic Polyurea Elastomer Systems Prepared from High 2,4'-MDI Isocyanates, Proceedings of the Polyurethanes Conference 2000, October 8-11, 2000, Boston.
6 Broekaert, M.; Pille-Wolf, W. The Influence of Isomer Composition and Functionality on the Final Properties of Aromatic Polyurea Spray Coatings, Proceedings of the Utech 2000 Conference, The Hague.
7 Broekaert, M. Modified MDI-Prepolymers Improve the Initial Physical Properties and Reduce the 'In-Service' Time of Aromatic Polyurea Coatings, Proceedings of the 6th Nurnberg Congress - Creative advances in coatings technology, April 2-4, 2001, Nurnberg, p. 761.
8 Broekaert, M. Profits in the Pipeline, Polymer Paints Colour J. July 2001, p. 18.
9 Perez, A. Jr.; Shen, C. C. Performance Enhancements of Aromatic Polyurea Spray Coatings by the Use of Conventional Primer Systems, presented at the Polyurea Development Association in New Orleans, Louisiana, November 29-December 1, 2000.
10 Gusmer Corp., Direct Impingement Mixing for the Spraying of Polyurea, 1st Annual PDA meeting, 30/11-1/12/2000, New Orleans.
11 Broekaert, M. Polyurea Spray Coatings, the Technology and Latest Developments, Polyurethanes for high-performance coatings II, ECC, Berlin, March 14-15, 2002.
12 Johnston, J. A.; and Smith, S. Physical Properties of Aromatic Polyurea Elastomer Coatings after Exposure to Extreme Conditions, Polyurethanes Expo 2002, Salt Lake City, October 13-16, 2002, p. 291.
13 Livingston, G. Polyurea Coating on Highly Reinforced Concrete Structures, 2nd Annual PDA meeting, November 28-30, 2001, Orlando.
14 Weiss, A. Polyurea and Metal Coating, 2nd Annual PDA meeting, November 28-30, 2001, Orlando.
15 PMDI User Guidelines for Chemical Protective Clothing Selection, The Society of the Plastics Industry, ref. AX178, July 1994.
16 Technical Update on skin protection by Ansell, website: www.ansell.be.
SUPRASEC is a registered trademark of Huntsman International LLC. JEFFAMINE and JEFFSOL are registered trademarks of Huntsman Petrochemicals Corp.
This paper was presented at the Fourth European Congress on Construction Chemicals on April 10, 2003 in Nurnberg, Germany (parallel to the European Coatings Show 2003).
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





