Non-Yellowing Catalyst for Uretdione-Crosslinked Powder Coatings
Cure temperatures can be lowered by 30 °C, to 170 °C, at very low use levels and by even more to 150 °C at higher catalyst levels. Physical properties of cured films in both clearcoat and pigmented systems have shown excellent chemical resistance, high gloss and good appearance. In addition, pigmented films have demonstrated little or no yellowing compared to uncatalyzed films under standard and overbake conditions.
By introducing an epoxy acid scavenger into the uretdione powder coatings, cure temperatures can be lowered even further, to 140 °C, at very low use levels. The acid scavenger prevents the carboxylic acid group on the hydroxyl functional polyester from inhibiting the catalyst activity.
The product is a white, free-flowing powder that can be easily incorporated into uretdione-crosslinked powder coatings; it is also an effective catalyst for use in caprolactam-blocked polyisocyanate powder coatings.
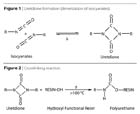
Powder coating technology continues to develop at an attractive rate, offering a viable alternative to conventional coating techniques. This is primarily due to its high efficiency, the durability of resulting finishes and very low environmental impact. In addition, powder coatings effectively resist chipping, scratching and fading, leading to high-quality finishes that are color stable. Since no solvents are involved in the process, the powder coatings can be recycled and reused, thereby reducing the emissions of volatile organic compounds and waste generated from the conventional liquid coatings. Powder technology finds widespread use in automotive parts and bodies, architectural furniture and many household appliances.
The current global powder coatings market is over 1 million metric tons, with the demand expected to exceed more than 1.2 million metric tons by 2008. The EU is leading the way with 43% of the market, followed by Asia with 26% and North America with 23%. By 2007, Frost & Sullivan predicts powder coating sales in Europe to reach $1.82 billion, driven by a compound annual growth of 7%.
In spite of its attractive end-use performance and environmental advantages, the relatively high curing temperatures encountered in the process limit its use to only substrates that can withstand high temperatures, such as metal.
Polyester-epoxy hybrids dominate the powder markets followed by epoxies, polyurethanes and acrylics. Epoxies generally have the best combination of properties including toughness, chemical resistance, corrosion and overall adhesion to the substrate, while polyesters possess better outdoor weathering properties than the epoxies, which generally tend to exhibit poor UV durability. Hybrids based on epoxies and polyester combine the desirable properties of each binder. Acrylics (functionalized with GMA) exhibit excellent film appearance and clarity, but are relatively brittle and expensive. Suitable catalysts are capable of lowering the cure temperatures of epoxy carboxy hybrids to as low as 120 °C.
Polyurethane powder coatings are well known for their exceptional weathering resistance, flow properties and overall finish quality. Since these systems are very reactive at room temperature and cannot remain stable in a one-package formulation, they are usually reversibly blocked with various blocking agents to provide good storage stability.
Several blocking agents are available based on the desired deblocking temperatures and end-uses, caprolactam being the most widely used in powder coatings. During the deblocking process, a large portion of the protecting group is released, leading to emissions of volatile organics.

Gedan-Smolka et al.3 described the use of zinc(II) acetylacetonate, which was a selective catalyst for the reaction of uretdione with a hydroxyl functional resin to produce an allophanate, a reaction that was selective at temperatures below 160 °C. Since the cured films were allophanates, the coatings were formulated with an NCO:OH equivalent ratio of 2:1. That dramatically increases the cost of coatings.
More recently, J.V. Weiß4 and coworkers at Degussa described catalysts based on tetraalkyl ammonium carboxylate. While a cure temperature of 140 °C was possible, the coating surface produced did not have the desirable flow properties of the conventional powder coatings.
We began our search for a more-efficient catalyst that can provide a lower temperature cure combined with desirable color properties. Herein we describe the development of a new uretdione catalyst that not only decreases the cure temperature, but also overcomes the yellowing problems associated with catalysts described earlier.

Materials
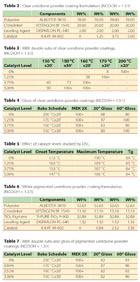
The raw materials shown in Table 1 were used to prepare uretdione powder coatings. ALBESTER 3870 and VESTAGON BF 1540 were used in initial lab evaluations to determine the activity of K-KAT XK-602 uretdione catalyst. The resins are supplied without catalyst. Two NCO:OH-equivalent ratios (1.3:1 and 1:1) were used to formulate the uretdione powder coatings.
Preparation of Powder Coatings
Uretdione powder components were premixed using a Waring Blender that was maintained at 2,500 rpm fo 2 minutes. The premix was melt-processed through a Prism Twin Screw Extruder set at 100 rpm. The temperatures of the four extruder zones were 30 °C, 35 °C, 90 °C and 110 °C. The extrudates were pulverized via a ZM100 Centrifugal Grinder from Glen Mills providing a less than 50 µm particle size. The final powder coatings were applied with an electrostatic powder-spraying unit at 60 kV onto degreased iron panels to obtain film thicknesses of approximately 60 µm. The films were then baked in an air circulated drying oven at temperatures between 130 °C and 200 °C.
Effect of Catalyst Levels
Four clear powder coatings (Table 2) were formulated with the NCO:OH-equivalent ratio of 1.3:1. The neat catalyst levels based on total resin solids were 0%, 1.25%, 3.75% and 5.00%. The catalyst is 80% active. Thus, the active catalyst levels were actually 0%, 1%, 3% and 4%.
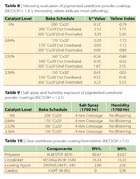
The coating containing 1.25% catalyst was fully cured at 170 °C when baked for 20 minutes (Table 3). The catalyst reduced the cure temperature by 30 °C compared with the coating without any catalyst. When the catalyst levels were raised to 3.75% and 5%, the coatings were fully cured at temperatures as low as 150 °C. There is a clear trend that the higher the amount of catalyst in the coating, the lower the cure temperature. The gloss and appearance of the cured films were excellent (Table 4).
The four powder formulations mentioned above were also evaluated by DSC (Differential Scanning Calorimetry) techniques. This again confirmed the clear trend that the higher the catalyst level, the lower the onset temperature (Table 5).
Four pigmented powder coatings (Table 6) were formulated with the NCO:OH-equivalent ratio of 1.3:1 as well. They comprised 33% of titanium dioxide. The neat catalyst levels based on total formulation were 0%, 0.84%, 2.52% and 3.36%.
The curing conditions and solvent resistance of these pigmented coatings are similar to that of the unpigmented systems described above. The higher the amount of catalyst in the coating, the lower the cure temperature (Table 7). This indicates that the pigment in the systems does not adversely affect the catalytic activity of the catalyst. High-gloss films were also produced in these experiments.
The coated panels were baked three times in an oven to evaluate the yellowing effect on overbake conditions (Table 8). The catalyst used in the coatings produces non-yellowing baked films and does not negatively affect the corrosion and humidity resistance of the coatings (Table 9).
Based on QUV experiments, durability of the tested coatings was not influenced by catalyst concentration.
Two clear powder coatings (Table 10) were formulated with an NCO:OH-equivalent ratio of 1:1. The coating with 5% catalyst was fully cured after a 20-minute bake at 160 °C, but not at 150 °C (Table 11). The solvent resistance of this coating was weaker than that of
the coating formulated with an NCO:OH-equivalent ratio of 1.3:1 (Tables 3 and 11).

VESTAGON BF 1320 uretdione crosslinker has higher functionality than VESTAGON BF 1540. Two clear powder coatings (Table 12) were formulated with the NCO:OH-equivalent ratio of 1:1 using VESTAGON BF 1320. The coating with 5% catalyst was fully cured at 150 °C when baked for 20 minutes (Table 13). It demonstrates that VESTAGON BF 1320 has higher reactivity than VESTAGON BF 1540, even with the NCO:OH equivalent ratio of 1:1 (Tables 3 and 13).
The unbaked (RT) and baked (140 °Cx20', 150 °Cx20' and 160 °Cx20') powder coatings mentioned above were analyzed by a Perkin Elmer Spectrum 2000 FT-IR instrument with DTGS (Deuterated Triglycine Sulfate) detector. The uretdione carbonyl peak of VESTAGON BF 1320 and the hydroxyl peak of ALBESTER 3870 are at 1764 cm-1 and 3544 cm-1, respectively. The coating baked at 140 °C for 20 minutes still contains a certain amount of unreacted uretdione and hydroxyl (Figure 3). The film was partially cured and achieved 66 MEK double rubs. When the coatings were baked at 150 °C and 160 °C for 20 minutes, all of the uretdione reacted and only a small amount of hydroxyl groups on the polyester remained. The films were fully cured and resulted in more than 200 MEK double rubs. The MEK double rub results in Table 13 were consistent with the IR analyses. Free isocyanate and urea were not detected. The free isocyanate and urea peak are at 2263 cm-1 and 1643 cm-1, respectively.
Effect of Epoxy Acid Scavengers
Carboxylic acid groups on the hydroxyl functional polyesters hinder the catalytic activity of K-KAT XK-602. TGIC (ARALDITE PT-810) and TMPTGE (ERISYS GE-30) were used as epoxy acid scavengers. Four clear powder coatings (Table 14) were formulated with an NCO:OH-equivalent ratio of 1:1 and an epoxy:carboxylic acid-equivalent ratio of 1:1. The amount of epoxy acid scavenger was calculated based on an acid number of 12, which was associated with the polyester used (ALBESTER 3870). Without acid scavenger, the coating requires 5% catalyst to be fully cured at 150 °C when baked for 20 minutes (Table 15). With acid scavenger, the coatings can be fully cured at 140 °C using 2.5% catalyst (Table 15). Clearly, the coatings with acid scavenger can be fully cured at lower temperatures with even lower levels of catalyst compared with the coatings without acid scavenger.
Caprolactam-Blocked Polyisocyanate Powder Coating
The catalyst was also evaluated in a caprolactam-blocked polyisocyanate powder coating. Two clear powder coatings (Table 16) were formulated with an NCO:OH-equivalent ratio of 1.1:1. VESTAGON B 1400 is a caprolactam-blocked polyisocyanate (NCO content: 13.3%; Tg 52 °C; Degussa Corporation). The coating with 2.5% catalyst was fully cured at 170 °C when baked for 15 minutes, which was better than the coating without catalyst (Table 17).
Conclusions
The uretdione catalyst presented in this paper has the following advantages:
- It can fully cure uretdione powder coatings formulated with an NCO:OH-equivalent ratio of 1:1 at 150 °C when baked for 20 minutes. Increasing catalyst level corresponds to decreasing cure temperature.
- Cure temperatures can be lowered even further, to 140 °C, with lower catalyst levels by introducing an epoxy acid scavenger.
- Cured films are non-yellowing.
- Gloss and appearance of cured films are excellent.
- Storage stability of the powder is excellent.
- There are no adverse effects on corrosion, humidity and UV resistance by using this catalyst.
- It is also an effective catalyst for use in caprolactam-blocked polyisocyanate powder coatings.
Acknowledgements
We thank Degussa Corporation, Hexion Specialty Chemicals, E.I. DuPont, Huntsman Corporation, CVC Specialty Chemicals and Kusumoto Chemicals for providing us with the raw materials.We appreciate Dr. Len Calbo for the guidance and support. We are grateful to Bob Coughlin, John Florio and Rui Gloria for evaluating the uretdione catalyst in powder coatings, and Mike Quintero for instrumental analyses.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!







