The Effects of TPO Composition on Adhesion and Proposed Chemical Mechanism
Adhesion of chlorinated polyolefin (CPO) and topcoat on various types of thermoplastic polyolefins (TPO) at three different baking temperatures, 180 ºF (82 ºC), 200 ºF (93 ºC), and 220 ºF (104 ºC), was investigated using a gasoline soak test. The results showed that co-monomer in the TPO and the base used in the topcoat were critical factors in adhesion. Also, a chemical mechanism for adhesion at the interface of topcoat-CPO and CPO-TPO was proposed. Results of preliminary study on chlorinated model compounds are also reported.
Adhesion of chlorinated polyolefin (CPO) and topcoat on various types of thermoplastic polyolefins (TPO) at three different baking temperatures, 180 ºF (82 ºC), 200 ºF (93 ºC), and 220 ºF (104 ºC), was investigated using a gasoline soak test. The results showed that co-monomer in the TPO and the base used in the topcoat were critical factors in adhesion. Also, a chemical mechanism for adhesion at the interface of topcoat-CPO and CPO-TPO was proposed. Results of preliminary study on chlorinated model compounds are also reported.
Several methods have been developed to improve adhesion to TPO. In general, they can be classified as oxidation methods and primer methods. In the oxidation method, the TPO surface is oxidized by treatment with flame, corona discharge,2,3 plasma,4 UV radiation5 or chemical oxidation.6,7 As a result, a more polar surface (by formation of an oxide layer or introduction of polar functionality) enables the adhesion of topcoat to TPO. There are several disadvantages of this method. First, coating must be done soon after treatment because of the reversibly short lifetime of the radical species. Second, chain scission on the surface of the TPO can occur due to over-treating. And third, non-uniform coverage can lead to cohesive failure of the TPO’s surface.8 In the primer method, an adhesion promoter is applied to the TPO surface to increase the polarity, which in turn promotes adhesion of topcoat to TPO. The most common adhesion promoters used in the North American automotive industry are based on chlorinated polyolefin (CPO). Although it requires an additional coating layer (adhesion promoter) to increase adhesion between TPO and topcoat, this primer method is the predominant North American method because it offers flexibility on diverse applications and types of TPOs and topcoats.
Previous efforts have been devoted to understanding the mechanism of CPO-TPO adhesion at their interface. The results indicated that CPO-TPO adhesion depends on many factors. Some key factors are type of test performed, baking temperature, strength of modulus, impact modifier, thickness of the interface and the residual solvent in the TPO. Studies by Williams and his coworkers showed that non-chlorinated polyolefins (NCPO) performed better than CPO in gasoline soak tests because they have less functionality and are therefore less soluble in organic solvent.9 The opposite is true for peel strength test.10 Temperature also plays an important role in adhesion. Higher bake temperature could significantly improve adhesion in the gasoline soak test for flexural modulus because there is more entanglement at the CPO-TPO interface.11
Another factor is the amount of impact modifier in the TPO. Ryntz and coworkers used lap-shear test to measure the strength of CPO-TPO interface adhesion. They found that increasing the impact modifier by 0.3% would increase the strength of adhesion almost two times.12 Besides the factors mentioned, the thickness of the CPO-TPO interface is also important. Theoretical and experimental studies by Mirabella and coworkers found that in order to have good adhesion between topcoats and TPO, the range of thickness for the CPO-TPO interface should be 11-400 nm.13
The residual solvent in TPO before and after baking also influences the CPO-TPO adhesion. Increasing solvent before baking allows or facilitates diffusion of the CPO into TPO surface. If less solvent is present after baking, the CPO-TPO adhesion increases.1 If the CPO-coated TPO is exposed to solvent after baking, the TPO matrix swells, which results in disruption of CPO-TPO adhesion.
Obviously, the mechanism of CPO-TPO adhesion is complex. Currently, the general hypothesis suggests that CPO-TPO adhesion is the result of diffusion of CPO into the surface of TPO and both become entangled after baking. This only explains the phenomenon physically. We believe the chemistry at topcoat-CPO interface and CPO-TPO interface would also be critical factors. In this study, we proposed a chemical mechanism for adhesion and explored the effects of chemical composition of various TPOs on adhesion. A set of chlorinated model compounds was also used to support our mechanistic concept.
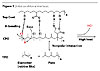
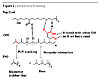 At the CPO-TPO interface, there exist non-polar interactions between aliphatic parts of the two layers. When the system is heated to high temperature, some chlorides in the CPO are unstable and the weak base in the topcoat can diffuse into the CPO and catalyze the elimination of HCl from CPO (dehydrohalogenation). This would result in a formation of some alkenes in the CPO layer. The –OH in the topcoat can also attack the anhydride in the CPO to create an ester and a carboxylic acid in the CPO. Overall, the system lost one H-bond between the topcoat-CPO interface but gained a covalent ester linkage and at least two more H-bonds from the newly formed carboxylic acid in the CPO (Figure 2).
At the CPO-TPO interface, there exist non-polar interactions between aliphatic parts of the two layers. When the system is heated to high temperature, some chlorides in the CPO are unstable and the weak base in the topcoat can diffuse into the CPO and catalyze the elimination of HCl from CPO (dehydrohalogenation). This would result in a formation of some alkenes in the CPO layer. The –OH in the topcoat can also attack the anhydride in the CPO to create an ester and a carboxylic acid in the CPO. Overall, the system lost one H-bond between the topcoat-CPO interface but gained a covalent ester linkage and at least two more H-bonds from the newly formed carboxylic acid in the CPO (Figure 2).
 In addition, the alkene in the CPO enhances the interaction between CPO and TPO through π-π interaction if the TPO composition contains some rubber-like elastomers or other more polar groups. The overall result is better adhesion for the entire system.
In addition, the alkene in the CPO enhances the interaction between CPO and TPO through π-π interaction if the TPO composition contains some rubber-like elastomers or other more polar groups. The overall result is better adhesion for the entire system.
From this mechanism, the key to adhesion is the composition of the TPO and the use of base catalyst at the topcoat-CPO interface. In Figure 3, we selected three simple halogenated butanes instead of the CPO itself for the ease of analysis and characterization of the products. Each reagent was subjected to heating with and without base catalyst. The main purpose is detection of any alkene formation in the reaction mixture. Reactions without base in these model systems served as controls.

For the dehydrohalogenation concept as shown in Figure 3, three chlorinated butanes (from Sigma-Aldrich) were heated to corresponding bake temperatures with and without base (1 equivalent N,N-dimethylethanolamine). This base was selected because it has a high boiling point and is commonly used in our topcoat. Each compound (1 ml) was added into a glass vial with label. The vials were simultaneously placed into a heating mantle at 82 ºC for 30 minutes. After that, the IR of each mixture was measured by a Nicolet 5DXC FT-IR spectrometer (Serial No. 8701412) with OMNIC software. The same procedures were repeated for 93 ºC and 104 ºC.
 Each panel (4x6 inches) was half-sanded with sand paper (Gator Grit 100) and washed to remove any dust and let dried overnight. After that, the CPO was sprayed on each panel and air dried for 10 minutes. Following, the topcoat was sprayed to each panel and all was baked in the gas oven at 82 ºC for 30 minutes. After baking, the panels were allowed to cool to room temperature. The same procedures were repeated for 93 ºC and 104 ºC.
Each panel (4x6 inches) was half-sanded with sand paper (Gator Grit 100) and washed to remove any dust and let dried overnight. After that, the CPO was sprayed on each panel and air dried for 10 minutes. Following, the topcoat was sprayed to each panel and all was baked in the gas oven at 82 ºC for 30 minutes. After baking, the panels were allowed to cool to room temperature. The same procedures were repeated for 93 ºC and 104 ºC.
 Panels that were baked at 82 ºC were completely immersed into gasoline mixture (45 wt% isooctane, 45 wt% toluene, 10 wt% ethanol) for 30 minutes, 60 minutes and 90 minutes. Observations were recorded, and the panels that completely failed the adhesion test were removed at each time interval. The same procedures were repeated for panels that were baked at 93 ºC and 104 ºC. A fresh gasoline solution was used for each set of temperatures.
Panels that were baked at 82 ºC were completely immersed into gasoline mixture (45 wt% isooctane, 45 wt% toluene, 10 wt% ethanol) for 30 minutes, 60 minutes and 90 minutes. Observations were recorded, and the panels that completely failed the adhesion test were removed at each time interval. The same procedures were repeated for panels that were baked at 93 ºC and 104 ºC. A fresh gasoline solution was used for each set of temperatures.

 We next plotted the effects of TPO type, melt flow, density (top left to right), flex modulus and temperature (bottom left to right) versus adhesion percentage in gas soak test (vertical axes) (Figures 6-8). For the y-axes, 100% means complete adhesion of the system and 0% means complete loss of adhesion.
We next plotted the effects of TPO type, melt flow, density (top left to right), flex modulus and temperature (bottom left to right) versus adhesion percentage in gas soak test (vertical axes) (Figures 6-8). For the y-axes, 100% means complete adhesion of the system and 0% means complete loss of adhesion.
 Figure 6 showed that C2/C8 type had strongest gasoline resistance among the TPOs in this experiment. It also showed that the gasoline resistance increased as the bake temperature increased. However, as the time of immersion in gasoline mixture increased, the temperature became less effective (Figures 7 and 8). In general, the adhesion of all TPOs decreased as the test became more severe by longer immersion time. The homo-formulation type had the weakest gasoline resistance because of its single component composition (isotactic polypropylene). Without co-monomer, its high density, high flex modulus, and inert surface did not enhance adhesion.
Figure 6 showed that C2/C8 type had strongest gasoline resistance among the TPOs in this experiment. It also showed that the gasoline resistance increased as the bake temperature increased. However, as the time of immersion in gasoline mixture increased, the temperature became less effective (Figures 7 and 8). In general, the adhesion of all TPOs decreased as the test became more severe by longer immersion time. The homo-formulation type had the weakest gasoline resistance because of its single component composition (isotactic polypropylene). Without co-monomer, its high density, high flex modulus, and inert surface did not enhance adhesion.
Both C2/C4 and C2/C8 types had better gasoline resistance than the homo-formula type because of the addition of co-monomer. Addition of C8 produced higher gasoline resistance than that of C4 because C4-containing TPO has higher solvent absorption rate which, in turn, reduces adhesion of the system.14 Also, addition of C8 to the TPO may increase non-polar interactions at CPO-TPO interface and resulting in better adhesion for the overall system. This is in agreement with our mechanism as described earlier. A simultaneous decrease in polarity of CPO (forming of alkene) and an increase in polarity of TPO (addition of co-monomer) or non-polar interactions will lead to better adhesion. For the TPOs that contain 18% talc, the same trend was observed. Therefore, talc was not a factor.
 A limited decrease in polarity of CPO can be accomplished through the dehydrohalogenation process. The IR results showed that our model compounds did not undergo this process without the presence of base. However, when adding base, dehydrohalogenation was detected with the C=C-H stretching around 2800-3000 cm-1 as in the case of 1,2-dichlorobutane (Figures 9 and 10). Similar results were observed for the other two model compounds. There was little or no C=C stretching observed at 1600 cm-1 for all model compounds. This is probably due to trans conformation of the alkene products.
A limited decrease in polarity of CPO can be accomplished through the dehydrohalogenation process. The IR results showed that our model compounds did not undergo this process without the presence of base. However, when adding base, dehydrohalogenation was detected with the C=C-H stretching around 2800-3000 cm-1 as in the case of 1,2-dichlorobutane (Figures 9 and 10). Similar results were observed for the other two model compounds. There was little or no C=C stretching observed at 1600 cm-1 for all model compounds. This is probably due to trans conformation of the alkene products.
Presented at the 10th International Coatings for Plastics Symposium 2007, Chicago, IL. For further information, contact timothy.december@basf.com and/or david.choi@basell.com.
Adhesion of chlorinated polyolefin (CPO) and topcoat on various types of thermoplastic polyolefins (TPO) at three different baking temperatures, 180 ºF (82 ºC), 200 ºF (93 ºC), and 220 ºF (104 ºC), was investigated using a gasoline soak test. The results showed that co-monomer in the TPO and the base used in the topcoat were critical factors in adhesion. Also, a chemical mechanism for adhesion at the interface of topcoat-CPO and CPO-TPO was proposed. Results of preliminary study on chlorinated model compounds are also reported.
Introduction
Thermoplastic polyolefins (TPO) are widely used in the automotive industry because they are durable, cost effective, easily molded into complex shapes and can be recycled. However, the major challenge in using TPO is the poor adhesion of paints (or topcoats). This is due to lack of functionality on the TPO that leads to poor wettability, inertness and low surface energy.1Several methods have been developed to improve adhesion to TPO. In general, they can be classified as oxidation methods and primer methods. In the oxidation method, the TPO surface is oxidized by treatment with flame, corona discharge,2,3 plasma,4 UV radiation5 or chemical oxidation.6,7 As a result, a more polar surface (by formation of an oxide layer or introduction of polar functionality) enables the adhesion of topcoat to TPO. There are several disadvantages of this method. First, coating must be done soon after treatment because of the reversibly short lifetime of the radical species. Second, chain scission on the surface of the TPO can occur due to over-treating. And third, non-uniform coverage can lead to cohesive failure of the TPO’s surface.8 In the primer method, an adhesion promoter is applied to the TPO surface to increase the polarity, which in turn promotes adhesion of topcoat to TPO. The most common adhesion promoters used in the North American automotive industry are based on chlorinated polyolefin (CPO). Although it requires an additional coating layer (adhesion promoter) to increase adhesion between TPO and topcoat, this primer method is the predominant North American method because it offers flexibility on diverse applications and types of TPOs and topcoats.
Previous efforts have been devoted to understanding the mechanism of CPO-TPO adhesion at their interface. The results indicated that CPO-TPO adhesion depends on many factors. Some key factors are type of test performed, baking temperature, strength of modulus, impact modifier, thickness of the interface and the residual solvent in the TPO. Studies by Williams and his coworkers showed that non-chlorinated polyolefins (NCPO) performed better than CPO in gasoline soak tests because they have less functionality and are therefore less soluble in organic solvent.9 The opposite is true for peel strength test.10 Temperature also plays an important role in adhesion. Higher bake temperature could significantly improve adhesion in the gasoline soak test for flexural modulus because there is more entanglement at the CPO-TPO interface.11
Another factor is the amount of impact modifier in the TPO. Ryntz and coworkers used lap-shear test to measure the strength of CPO-TPO interface adhesion. They found that increasing the impact modifier by 0.3% would increase the strength of adhesion almost two times.12 Besides the factors mentioned, the thickness of the CPO-TPO interface is also important. Theoretical and experimental studies by Mirabella and coworkers found that in order to have good adhesion between topcoats and TPO, the range of thickness for the CPO-TPO interface should be 11-400 nm.13
The residual solvent in TPO before and after baking also influences the CPO-TPO adhesion. Increasing solvent before baking allows or facilitates diffusion of the CPO into TPO surface. If less solvent is present after baking, the CPO-TPO adhesion increases.1 If the CPO-coated TPO is exposed to solvent after baking, the TPO matrix swells, which results in disruption of CPO-TPO adhesion.
Obviously, the mechanism of CPO-TPO adhesion is complex. Currently, the general hypothesis suggests that CPO-TPO adhesion is the result of diffusion of CPO into the surface of TPO and both become entangled after baking. This only explains the phenomenon physically. We believe the chemistry at topcoat-CPO interface and CPO-TPO interface would also be critical factors. In this study, we proposed a chemical mechanism for adhesion and explored the effects of chemical composition of various TPOs on adhesion. A set of chlorinated model compounds was also used to support our mechanistic concept.

Proposed Chemical Mechanism
Much attention in TPO adhesion was focused on the physical aspect of the substrates at the interface. However, to fully understand the mechanism of adhesion, one must examine the chemical aspect of the system, especially the chemistry at the interface of layers (topcoat, CPO and TPO). To our knowledge, this is the first time a detailed chemical mechanism has been proposed. In this mechanism, initially there are H-bonds between the polar groups in the topcoat (-OH, -CO2H) and the CPO (-Cl, anhydride) (Figure 1).

From this mechanism, the key to adhesion is the composition of the TPO and the use of base catalyst at the topcoat-CPO interface. In Figure 3, we selected three simple halogenated butanes instead of the CPO itself for the ease of analysis and characterization of the products. Each reagent was subjected to heating with and without base catalyst. The main purpose is detection of any alkene formation in the reaction mixture. Reactions without base in these model systems served as controls.

Experimental
Model SystemsFor the dehydrohalogenation concept as shown in Figure 3, three chlorinated butanes (from Sigma-Aldrich) were heated to corresponding bake temperatures with and without base (1 equivalent N,N-dimethylethanolamine). This base was selected because it has a high boiling point and is commonly used in our topcoat. Each compound (1 ml) was added into a glass vial with label. The vials were simultaneously placed into a heating mantle at 82 ºC for 30 minutes. After that, the IR of each mixture was measured by a Nicolet 5DXC FT-IR spectrometer (Serial No. 8701412) with OMNIC software. The same procedures were repeated for 93 ºC and 104 ºC.

Practical System
To test our hypothesis, 11 TPOs with different composition were used (Table 1). The TPOs were grouped by formula composition: homo polypropylene, plastomers (C2/C4 and C2/C8 types) and 18% talc filled polypropylenes or plastomers. A typical TPO composition is shown in Figure 4.


Results and Discussion
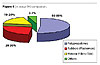
Using MiniTabTM software, we first explored whether the roughness of the TPO surface (by sanding) has a strong effect on the adhesion. The analysis showed that it had minimal effect of only 5%, which is the same as the percent error (Figure 5). The major effects are from TPO substrate (44%) and bake temperature (20%). The interaction between TPO substrate and bake temperature accounted for 25% effect.

Both C2/C4 and C2/C8 types had better gasoline resistance than the homo-formula type because of the addition of co-monomer. Addition of C8 produced higher gasoline resistance than that of C4 because C4-containing TPO has higher solvent absorption rate which, in turn, reduces adhesion of the system.14 Also, addition of C8 to the TPO may increase non-polar interactions at CPO-TPO interface and resulting in better adhesion for the overall system. This is in agreement with our mechanism as described earlier. A simultaneous decrease in polarity of CPO (forming of alkene) and an increase in polarity of TPO (addition of co-monomer) or non-polar interactions will lead to better adhesion. For the TPOs that contain 18% talc, the same trend was observed. Therefore, talc was not a factor.

Conclusion
The results of this study indicated that the type of co-monomer in the TPO and the base used in the topcoat were critical factors in gasoline resistance. Simultaneous decrease in the polarity of the CPO and increase in the polarity or non-polar interaction of TPO will improve the overall adhesion. This can be done by altering the composition of the TPO by addition of co-monomer to the TPO, and addition of a base to the topcoat. Due to the limitation of number of substrates used in this experiment, our results may not be indicative of other types of TPOs that may or may not conform to our hypothesis. A more comprehensive study of this subject is the objective of our future study.Presented at the 10th International Coatings for Plastics Symposium 2007, Chicago, IL. For further information, contact timothy.december@basf.com and/or david.choi@basell.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




