Novel High-Solids Systems Based On Silicone-Epoxy Resins

Corrosion protection for industrial, industrial maintenance and marine markets can be an expensive and overwhelming task. Protecting objects such as oil platforms, bridges, storage tanks, steel structures, concrete walls and floors, and ship decks from corrosion is labor-intensive, with high material costs. Still, one of the best methods for fighting corrosion is the use of protective coatings.
Coatings provide corrosion protection by one or a combination of three basic mechanisms: barrier, inhibitive and/or sacrificial. A physical barrier between the substrate and its environment may act by keeping oxygen and moisture away from the substrate. Examples include coal tar enamels and vinyl coatings. An inhibitive coating may act by passivating the metal and interfering with the corrosion process. Most such coatings are based on epoxies and urethanes. A sacrificial coating will itself corrode instead of the metal or steel structure it protects. Such a coating would be zinc-rich or metallized.
Coatings systems are typically organic or inorganic in nature. Traditionally, the industry has relied on protective coatings composed of organic resin systems. The variety of properties needed usually dictates a multiple coat system: For example, a zinc-rich primer, an epoxy mid-coat and a polyurethane topcoat. This multiple-coat system enables the formulator to provide multiple levels of protection by utilizing the sacrificial protection of the zinc-rich primer, the corrosion resistance of the epoxy and the weather resistance of the urethane topcoat.
Exploring inorganic silicone-based chemistry has led to the development of a silicone-epoxy hybrid polymer, which combines the properties of both organic and inorganic compounds in a new class of resins for protective coatings. This paper will discuss the benefits of these novel silicone-epoxy resin systems.
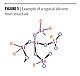
Polysiloxane Chemistry
As mentioned above, most protective coatings are based on organic resin systems. Unfortunately, they are subject to degradation by thermal and photo-induced oxidation, as well as chemical attack. The silicone inorganic systems are much more resistant to such degradation mechanisms.These silicone inorganic resin structures are comprised of polysiloxanes with a stable [-(Si-O)n-Si-] backbone (Figure 1). The Si-O inorganic groups in the polymer backbone provide a bond strength of 108 kcal/mole compared to the C-C organic bond strength of only 83 kcal/mole. Because it requires more energy to break apart the Si-O bond than a C-C bond, the silicone structures improve weathering and thermal resistance. And since the silicone is already oxidized, it is not subject to the same oxidative degradation that affects the C-C bonds.
Several properties and characteristics of silicone resins make them excellent components for protective coating technology:
· thermal stability;
· weather and UV resistance;
· color stability;
· low surface tension;
· hydrophobic nature;
· excellent release properties;
· good insulator; and
· low influence on health.
Coupling silicone resins with an organic component improves the properties even further. The next section outlines improved protective coating characteristics achieved by combining inorganic polysiloxane and organic epoxy polymer.

Silicone-Epoxy Resins
Due to their combination of organic epoxy functionality with inorganic silicone functionality, silicone-epoxy systems are generally referred to as “hybrid coatings.” The term “hybrid coating” reflects the nature of two binder systems that have distinct properties and curing mechanisms that work together to form one novel resin system.In this polymer system, siloxane forms the matrix with epoxy and alkoxy functionality in the side chains. The condensation reaction of an aliphatic epoxy with a polysiloxane produces the novel chemistry of the silicone-epoxy polymer. As reference to the “hybrid” nature of the polymer indicates, two reactions take place. The amine group of the aminosilane cures the epoxy resin in a typical manner. At the same time, a competing polycondensation reaction occurs between the silicone alkoxy groups of the hardener and resin. Figure 2 shows the silanol condensation reaction.
The advantages of the epoxy combined with the strength of the polysiloxane result in a two-component, ambient-cure thermoset coating. The low viscosity of this polymer enables formulation of a low-VOC, high-solids, non-isocyanate coating.

Results and Discussion
Reducing material and labor costs while maintaining corrosion resistance is a major focus in the protective coatings market. We tested a two-coat system consisting of a 75-micron zinc-rich primer top-coated with a 125-micron silicone-epoxy coating. We then compared the results to the traditional three-coat system consisting of a 75-micron zinc-rich primer, 125-micron epoxy mid-coat, and a 50-micron polyurethane top-coat (Figure 3).Having established the structure of a two-coat hybrid coating system and a traditional three-coat organic coating system, this paper will now present the design and results of two different experiments on these structures.
The first experiment was to evaluate how the two-coat zinc rich primer/silicone-epoxy technology performed against the traditional three-coat zinc rich primer/epoxy mid-coat/urethane top-coat technology.
The second experiment determined the performance of the first generation silicone-epoxy technology against the more flexible second generation silicone-epoxy technology. The main difference between the first-generation silicone-epoxy and the second generation is the alkoxy content on the silicone side. These changes are evident in the number of alkoxy units as well as the types of alkoxy groups used.
As you can see from the starting point formulation, the hybrid system requires very little solvent. Formulators may substitute other solvents, such as t-butyl acetate or acetone, for butyl acetate to lower the VOC content even further. The silicone-epoxy resin exhibits a low viscosity at a nonvolatile content by mass of approximately 98%. Due to the high solids content and low viscosity, it is possible to formulate a sprayable version with only a minimal amount of solvent – low enough to limit VOC content to less than 100 g/L.
To determine the performance of the two different silicone-epoxy resin systems as well as the traditional three-coat systems, we examined a number of different experimental tests. Comparative tests performed include physical properties, chemical resistance, corrosion resistance in salt spray and humidity testing, Florida exposure and QUV testing for weatherability.

The second-generation resin system exhibits better initial gloss, cross-hatch adhesion and longer pot life. The first generation has higher development of hardness as well as shorter cure time. This data helped confirm the theory that the modification of the alkoxy content develops a more flexible second-generation silicone-epoxy resin. Recoat time is also an important characteristic of these coatings. With their 48-hour recoat window, silicone-epoxy systems exhibit a certain degree of anti-graffiti properties due to the high crosslink density and the siloxane backbone. Therefore, as the crosslink densities of these systems build, the more difficult it is to apply multiple coats. Although the recoat time can make it difficult for the novice applying silicone-epoxy technology to a structure, these anti-graffiti properties are of benefit to many formulators for steel structures, bridges, rail cars, and other industrial and industrial maintenance coatings.

Chemical Resistance
Chemical resistance is critical for protective coatings. Not only are they exposed to rust and corrosion but also to a number of different acids, bases and solvents. Table 2 illustrates the differences between the traditional technology and the novel silicone-epoxy approach. This table also compares the first-generation silicone-epoxy to the second generation to determine if changing the alkoxy content produces any benefit or decreases performance.Table 2 shows that the silicone-epoxy coating systems outperformed the traditional three-coat system in resistance to most acids and bases. When testing various solvents all three systems demonstrated equally outstanding performance. Differences in chemical resistance became more pronounced in seven-day testing. There was only a minor decrease in chemical resistance to acids in the second-generation resin, but it still outperformed the traditional three-coat system.

Environmental Factors
Other important properties of the protective coating systems include salt spray, humidity and QUV testing. These tests establish the ability of each coating system to withstand the elements and protect against environmental factors such as corrosion and light (Tables 3-5).


Florida Exposure
Although many other experiments and studies were performed, the final study worth noting is the weathering data. Salt spray, humidity and QUV-B are all great forms of accelerated testing, but we also wanted to test these first and second generation silicone-epoxy resins in exposure to real-world conditions. As a result, we decided to prepare these two silicone-epoxy systems in the same experimental starting-point formulation subjected to accelerated testing and submit them to Florida exposure testing. Currently 42 months of Florida exposure has been completed. Figures 5-7 show the performance of the first- and second-generation silicone-epoxy systems.
Figure 6 shows exactly what we expected with regard to gloss retention. Gloss retention is much greater for the second-generation system than for the original silicone-epoxy system developed.

Following the trend of the gloss numbers in the charts shown in Figures 5 and 6, the second-generation silicone-epoxy outperforms the first generation with regard to ΔE. The difference between both resins, shown in Figure 7, is much more subtle, with a difference of approximately 0.2 after 42 months.
Conclusion
The novel “hybrid” chemistry achieved by combining an aliphatic epoxy with a polysiloxane allows the formation of a silicone-epoxy resin that performs better than an organic or inorganic polymer alone. This approach introduces a durable binder for the protective coatings industry. The first experiment shows the benefits of a two-coat system over the traditional three-coat system, while the second experiment shows how modifying the alkoxy content can achieve performance improvements with a more flexible, second-generation system. With this novel class of material, formulators can incorporate the following performance benefits in their products:· high volume solids;
· low VOC;
· excellent color and gloss retention;
· excellent corrosion resistance in two-coat systems;
· cost-effective, two-coat alternatives to multiple-coat systems;
· health and safety compliance;
· isocyanate free;
· excellent chemical resistance;
· a degree of anti-graffiti properties; and
· tolerance to high humidity due to nature of curing mechanisms.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





