Toughening of Epoxy Coating Systems with Novel Bio-Based Materials
The market is looking for possibilities to reduce weight by reduction of the thickness of metal sheets and maintain the protective properties. To meet these needs, there is a demand for more flexible, but tough coatings without compromising the other properties.
Epoxy systems have found widespread use in coatings, sealants and adhesives applications. These systems provide hard, highly crosslinked coatings with excellent protective properties. Pripol dimerized fatty acids have traditionally been used in epoxy resins, in polyamide curatives and in modified epoxies. The benefits that they add to these resins are flexibility, vibration resistance and stress absorption, hydrophobicity, water resistance, thermo-oxidative resistance, and compatibility with, for example, hydrocarbon resins.
For some years, Croda has had a range of polyester diol derivatives of dimerized fatty acids under the product name of Priplast. Recently, Croda has developed new bio-based polyester diacids based on renewable dimerized fatty acids. The carboxylic acid functionality allows these polyesters to be grafted on the epoxy as building blocks for epoxy systems.
Depending on the molecular weight and polarity of the polyester diacid in relation to the epoxy, micro-scale phase separation occurs. This reaction-induced phase separation leads to a morphology with low-Tg micro-segments that are homogeneously distributed inside a hard matrix, resulting in hard and, at the same time, tough systems. Next to bringing good toughening performance, the polyester diacids cause a reduction in moisture diffusion and enhance thermo-oxidative stability. The relatively low viscosity allows ease of formulation, handling and formulation flexibility that is combined with the enhanced environmental profile of a renewable impact modifier. As such, polyester diacids based on dimerized fatty acids can be used as relatively low-viscosity, built-in impact modifiers.
This paper describes incorporation of the polyester diacids as building blocks in epoxy systems to bring the combination of flexibility and toughness to coating systems. Initially, the influence of polyester diacids on the epoxy morphology was tested. The influence of polyester composition and molecular weight were examined. Subsequently, the properties of modified epoxies were studied, in comparison with a conventional rubber toughener and a traditional flexible epoxy system.
 Credit: Croda Incorporated Credit: Croda Incorporated
|
| Figure 1 Click to enlarge |
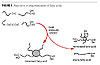
Dimer Fatty Acids
Natural oils and fats have for years provided the polymer chemist with a variety of building blocks, such as glycerin and castor oil. Less well known is the use of a fatty acid derivative, the so-called dimerized fatty acids. These are obtained by the conversion of unsaturated fatty acids (from sources like soybean oil or tall oil) by a combination of pressure, temperature and catalysis. This process generates a mixture of products, the most important being dimerized fatty acid. Others are trimerized fatty acid, and isostearic acid. Figure 1 gives an overview of the dimerization process.
Starting from the C-18 acids that nature typically provides, the dimer acid is a molecule with 36 carbon atoms, which makes it by far the longest dioic acid available. This hydrocarbon nature makes dimer acid and polymers in which it is included extremely hydrophobic. In addition, the combination of hydrocarbon character and non-crystallinity provides lubricity and flexibility, even at very low temperatures.
Dimerized fatty acids have found their application in such areas as polyamide epoxy curatives, polyamide resins, polyester and polyurethane coatings, adhesives, elastomers and foam. In all these applications, the value of the dimerized fatty acid is related to the features mentioned: flexibility and impact strength, lubricity and flowability, hydrophobicity and hydrolytic resistance. Dimer fatty acids for toughness modification of epoxies form yet a relatively unexplored area.
Epoxy Resins
Epoxy resins have a wide use in applications like coatings, flooring, sealants, grouts and adhesives. Epoxy systems are versatile because of the large amount of potential epoxy resins and the curing agent combinations, each giving a different structure to the final material. The broad spectrum of the potential epoxy/amine systems generally provides excellent adhesion to a wide range of substrates, resistance to a spectrum of chemicals and good mechanical properties. These properties and easily varied formulations allow the formulator to tune a resin system’s cure, handling and characteristics.
However with the high solids or solventless formulations, the challenge is for the formulators to get the potlife and cure temperature at the desired level, but another difficult challenge is to obtain highly flexible, yet tough materials for coatings, floors, adhesives, grouts and sealants. The general experience has been that improving one property normally means weakening several other properties.
Toughness and flexibility are important to distribute the stress that occurs during the duration of the epoxy system’s service life, especially in thick-film applications such as tank linings, floor coatings and adhesives. A number of approaches are available for the formulator to create flexibility and toughness, but rarely both to any significant extent.
One of the traditional ways to incorporate a flexible backbone is via the curing agents, which are available in many types: polyamides from dimer acids, adducts from ethylene- or propylene-oxide and polymeric anhydrides.
However these often mostly provide flexibility. Additionally, modified epoxy resins that have dimer acid incorporated in the backbone are used. These are normally used as a modifying agent at levels of approximately 10% on the epoxy component. They increase the flexibility and toughness modestly at these levels. At higher degrees of addition the flexibility increases but the toughness is lost.
Another way is through the use of thermoplastic polymer, which can be separated into products that exist as a separate phase like acrylonitrile-butadiene rubbers; those that separate into a second phase during cure e.g., carboxyl-terminated butadiene acrylonitrile (CTBN) rubbers and soluble polymers that exist as a separate phase in the cured system. These polymers work through phase separation in the cured system and provide an increasing toughness but have a small effect on the flexibility.
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Figure 2 Click to enlarge |
Dimer Acid for Epoxies
The dimer acids provide epoxy resins with enhanced flexibility, but the reduced glass transition temperature is not always favorable. Building of dimerized fatty acid into carboxylic acid-terminated polyesters makes it available for incorporation in epoxy resins as a larger-molecular-weight species (Figure 2). Due to the apolar character of the dimer-based polyesters, it can be expected that they will form a separate phase in the more polar epoxy matrix. As such, they can be introduced as a second-phase toughness modifier. By reaction-induced phase separation, the modifier forms soft domains within the hard epoxy matrix.
It can be expected that these dimer-based polyesters will have little impact on the glass transition temperature, bring better fracture toughness, enhance peel strength and provide a moisture barrier to the epoxy. A range of dimer-based polyesters with carboxylic acid functionality has been developed and tested in epoxy adhesives in order to substantiate the above-mentioned assumptions.
Initially, dimer-based polyesters with carboxylic acid functionality were developed with a range of polarities and molecular weights. They were introduced in epoxy coatings, and the influence of dimer-based polyester composition on the morphology was examined. This study was followed by testing the properties of these modified epoxies, in comparison with a conventional rubber-modified epoxy.
Bio-Based Polyester Diacids Polarity Effect on Morphology
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Table 1 Click to enlarge |
To investigate the morphology effect of the bio-based polyester diacids, a structural epoxy formulation was used. The formulations used were heat-curable and contained 66% of pre-reacted material, 25% high-molecular-weight Bisphenol A epoxy (Epikote©1001), 7% phenolic Novolac resin and a catalyst. These formulations were cured at 175 °C.(1)
Bio-based polyester diacids were produced from a combination of dimerized fatty acid and a low-molecular-weight dioic acid with glycol. The polarity was changed by varying the ratio of dimer acid to short dioic acid, in which the short dioic acid increases the polarity. The molecular weight was kept constant at about Mw 1500 (Table 1). The influence of these polyesters on epoxy morphology was examined using electron microscopy.
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Figure 3a 3b Click to enlarge |
Initially, polyester from dimerized fatty acid without short dioic acid was used as epoxy modifier. Figure 3a shows the micrograph of the material; the dimer-based polyester can be seen as large droplets of about 40 μm diameter. Due to the highly hydrophobic character, the material was too incompatible with the epoxy resin. No grafting reaction had occurred between the dimer-based polyester and the epoxy. The final resin was brittle, like unmodified epoxy, and sticky because of the separated polyester.
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Figure 4 Click to enlarge |
To enhance compatibility, the dimer-based polyester was made more polar by introducing a low-molecular-weight dioic acid. Polyester was made, combining 75 parts dimerized fatty acid and 25 parts short dioic acid. Figure 3b shows the micrograph of the epoxy modified with this polyester. The dimer-based polyester droplets were considerably reduced in size. Some grafting reaction had occurred, but still the components were not compatible enough. Again, the resin was brittle and sticky.
Another polyester was made, combining 50 parts dimerized fatty acid and 50 parts short dioic acid. Figure 4 shows two micrographs of the epoxy modified with this polyester. Full grafting took place, resulting in excellent phase separation on a micro-scale. The dimer-based polyester droplets were about 5 μm in size. The final resin was hard and tough. By reaction-induced phase separation, the modifier formed soft domains within the hard epoxy matrix, thereby toughening the epoxy.
Effect of Bio-Based Polyester Diacid Molecular Weight
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Table 2 Click to enlarge |
Polyester diacids were produced from a combination of dimerized fatty acid and a low-molecular-weight dioic acid at 50:50 ratio. As was discussed previously, polyesters of this polarity result in proper reaction-induced phase separation in the epoxy matrix. The molecular weight of the polyester was varied, and the influence on epoxy morphology was examined using electron microscopy. Increasing the molecular weight resulted in a reduction in size of the soft dimer polyester domains. Results are given in Table 2. Due to the larger size of the hydrophobic blocks that are built into the more polar epoxy, phase separation results in more finely distributed soft domains.
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Figure 5 Click to enlarge |
For all samples, the apolar dimer polyester particles were homogeneously distributed through the more polar epoxy matrix. The hydrophobic dimer polyester particles make the epoxy moisture resistant. Moisture diffusion was examined for these modified epoxies to explore the influence of morphology on moisture resistance. It was found that a more finely distributed morphology, with smaller soft domains, resulted in lower water diffusivity. This is shown schematically in Figure 5.
Preparation of Modified Epoxy Resins
Acid-functional dimer-based polyesters were grafted onto the epoxy matrix. They were pre-reacted with low-molecular-weight Bisphenol A epoxy (EPIKOTE 828) to obtain epoxy-functional material with 67% of the dimer-based polyester. In the case of the comparative reference formulation, a low-molecular-weight, carboxylic acid-terminated copolymer of acrylonitrile and butadiene, CTBN 1300 x 13, was pre-reacted in similar fashion. Additionally a flexible epoxy resin was prepared by reacting PRIPOL 1017 with low-molecular-weight Bisphenol A epoxy (EPIKOTE© 828). The evaluated formulations consisted of EPIKOTE 828, modified epoxy resin and amine curative Epikure© 3072.
Epoxy Modifier Effect on Coating Properties
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Figure 6 Click to enlarge |
A number of basic coating properties like flexibility and toughness depend on the viscoelastic behavior, physical transitions and relaxations. Coatings, as the polymers from which they are prepared, are viscoelastic in nature, that is, they behave both as viscous liquids and as elastic solids. The coatings have elastic recovery and yet will flow with time when placed under a stress. In general, viscoelastic behavior and mechanical properties are markedly affected when a coating enters the glass transition or relaxation. The effect on glass transition can be evaluated by e.g., differential scanning calorimetric methods. The ability to withstand stress can be validated by using bending and impact tests. This will be an indication towards flexibility, the ability of the coating to undergo a bend without failure, and towards toughness, the ability to withstand great strain in a short time without breaking or rupture.
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Figure 7 Click to enlarge |
The premise of the evaluation was to study the effect of the new polyester diacids in comparison to a dimer-modified and a non-modified epoxy resin. As reference a CTBN (Hypro1300X13) adduct, commonly used as a toughening agent in epoxy systems was included. For the evaluation of the epoxy system consisting of the Epikote 828, modified epoxy resin and curing agent were mixed at room temperature and diluted to 600 mPa.s with xylene/butanol (ratio 1/1). The impact of the addition of the modified epoxy resin to the Epikote 828 was measured.
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Figure 8 Click to enlarge |
Figure 6 shows the impact on the epoxy resin viscosity when incorporating the 5% rubber component in the epoxy system. The formulated epoxy resin with polyester diacids gives only a limited increase to the viscosity when compared to the CTBN-modified resin and is also lower then the dimer-modified resin. This allows the formulator to keep the solvent level low.
A range of formulations was made containing 0, 5, 10 or 15% rubber phase in the system. The resulting clear varnish was applied wet at 150 µ by a drawdown bar on the various test substrates. After initial screenings a selection was made with a focus on the use of 5% of the rubber component in the epoxy system.
Figure 7 shows the effect on hardness and Tg of using 5% of the rubber component in the epoxy system. Measured according to DIN EN ISO1522, one can observe that the hardness of the modified epoxy based on polyester diacid P02 and the PRIPOL 1017 did not lose the surface hardness, whereas the CTBN-modified, polyester diacid P01, and P03 had reduced hardness compared to the unmodified epoxy system.
When the glass transition is evaluated in relation to the hardness we see that the Tg of the PRIPOL 1017-based coating only decreases a few degrees, which would normally reduce the hardness of the coating. However one has to keep in mind that the degree of crosslinking in this system is higher than from the CTBN and polyester diacids P01/02/03, which will increase the hardness. The lower Tg of the polyester diacids suggests that the coatings will demonstrate a more elastic behavior.
To check the elastic behavior, the coatings were tested by using the cylindrical Mandrel test (DIN EN ISO 1519). The incorporation of the rubber component in the epoxy system shows flexibility increase, as can be seen in Figure 8.
Combining the flexibility data with the hardness data of Figure 7, the polyester diacid P02 shows good flexibility but at the same time gives a surface hardness comparable to the reference epoxy system based on Epikote 828/Epikure 3072. This indicates that the polyester diacid P02 has the ability to resist crack formation during a deformation.
Compared to the CTBN-modified resin we see good flexibility, but the hardness drops. This is caused by incompatibility of the CTBN-modified epoxy in the hard matrix giving a bad phase separation. To a lesser extent, the polyester diacids P01/03 have lost some hardness, but improved on deformation resistance over the reference epoxy system. Most likely the polarity difference between the epoxy resins based on polyester diacids P01 and P03 didn’t give the optimum phase separation. The PRIPOL 1017-modified epoxy resin shows a retaining hardness and an improved flexibility but doesn’t reach the level of the polyester diacid P02-modified epoxy. It lacks the ability to absorb the strain imposed on the coating.
Impact resistance testing is an additional means of predicting energy storage and loss as a function of temperature. For good impact resistance a coating must consist of a polymer with strong intermolecular entanglements and flow, combined with energy dissipation. Impact tests were done according DIN EN ISO 6272 at 25 °C and after 24 h exposure to -25 °C. Figure 9 shows the test results of this evaluation.
 Credit: Croda Incorporated Credit: Croda Incorporated |
| Figure 9 Click to enlarge |
We can see that the coatings consisting of the modified epoxy based on polyester diacid P01 and P02 have the ability to absorb the inflicted deformation at room and lower temperature. The good phase separation of the rubber phase in the rigid epoxy phase enables the coating to dissipate the energy of the impact. The CTBN- and PRIPOL 1017-modified epoxy resins have comparable to lower impact resistance than the reference due to compatibility issues with the epoxy matrix giving a bad phase separation.
Conclusion
The newly developed bio-based polyester diacids based on dimer fatty acid can well be used for toughness modification of epoxy systems. Flexibility of the chemistry allows tailoring of the polyester molecular weight and polarity in order to fit the base epoxy resin. It allows the desired morphology to be exactly achieved.
Modifying epoxies with polyester diacids enhances the flexibility at equal surface hardness when incorporated at a 5% rubber component level on the epoxy system. Especially in the Epikote 828/Epikure 3072 system the polyester diacid P02 gives good performance due to the desired micro-phase separation. Additionally the lower viscosity of the polyester diacid-modified epoxies allows formulation flexibility. The lower viscosity is useful for the ease of handling and allows the use of more fillers.
Based on the results found for epoxy-toughened adhesives,1 it is expected that the bio-based polyester diacids, based on their renewable dimer fatty acid, exhibit moisture resistance due to their hydrophobic nature, and increase hydrolytic, thermal and oxidative resistance.
The information in this publication is believed to be accurate and is given in good faith, but no representation or warranty as to its completeness or accuracy is made. Suggestions for uses or applications are only opinions. Users are responsible for determining the suitability of these products for their own particular purpose. No representation or warranty, expressed or implied, is made with respect to information or products including, without limitation, warranties of merchantability, fitness for a particular purpose, non-infringement of any third party patent or other intellectual property rights including, without limit, copyright, trademark and designs. Any trademarks identified herein are trademarks of the Croda group of companies.
This paper was presented at the American Coatings Conference, Charlotte, NC, 2010.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!