UV-Curable Systems for Sensitive Applications
Introduction
 Credit: BASF SE Credit: BASF SE
|
| Figure 1 Click to enlarge |
UV curing has become a well-accepted technology, mainly due to unique environmental and economic benefits compared to conventional drying processes. Solvent-free systems, low-energy consumption and high production rates explain the fast growth of this technology in various industrial sectors such as printing inks, protective coatings, adhesives and composites.
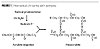
In a UV-curing process, a liquid resin is transformed within a fraction of a second into a solid polymer with outstanding mechanical properties. The cure reaction is initiated by means of a photoinitiator that decomposes when absorbing light (Figure 1).(1,2)
UV Curing in the Packaging Industry
High productivity and the superior quality of UV-printed over conventionally printed materials have made this technology especially successful in the graphic arts industry. However, to gain full acceptance by the packaging industry, a well-known UV-curing issue still has to be tackled: the migration of additives, e.g., photoinitiators, from the packaging into food. Indeed, after exposure to UV light, the photoinitiator has not been completely photolyzed. Significant amounts remain trapped in the printed ink and can be further extracted from the layer and transferred to the contents of the package.
 Credit: BASF SE Credit: BASF SE |
| Figure 2 Click to enlarge |
Type I photoinitiators, shown in Figure 2, directly initiate the UV-curing process by a-cleavage.(3,4) Here, concomitantly with the photolysis of the photoinitiator, photodecomposition products are generated, as shown for a-hydroxyketone HK-1.(5) Volatile molecules release the film during or just after exposure to UV light and in some cases give off a strong odor, while components with a higher molecular weight remain trapped in the cured ink and, like the photoinitiator, can be extracted when entering into contact with an organic or aqueous phase.
 Credit: BASF SE Credit: BASF SE |
| Figure 3 Click to enlarge |
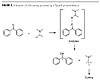
A second type of initiating process, shown in Figure 3, involves Type II photoinitiators, which do not undergo a-cleavage when exposed to light but go through a bi-molecular initiating reaction in the presence of hydrogen donors. Type II photoinitiators are essentially based on aromatic ketones (e.g., benzophenone, thioxanthones or camphorquinones). Polymerization is initiated by the amino alkyl radical, whereas the ketyl radical does not initiate polymerization of acrylate double bonds.(6,7) Since they do not generate secondary breakdown products, they were, until recently, regarded as low-emission photoinitiators but were unfortunately found to migrate significantly. This can be explained by the fact that they are not grafted or attached in the final network.
UV-printed packages are essentially used in indirect food contact applications. Some migrating species are occasionally supposed to diffuse from the printed exterior of the packaging to the inside, but the most probable explanation for migration lies in the production process and storage method of packages. Once printed and rolled, the inside of the packaging comes into direct contact with the printed outer surface. This is probably the crucial stage at which small molecules are transferred to the inside, where they will later come into direct contact with the foodstuffs.(8)
The objective of the following discussion is to demonstrate that a combination of the right initiator package and the right processing conditions can actually address this issue and provide a safe solution to the UV-curing industry, which is seeking low-emission and low-migration systems.
Experimental
Products
 Credit: BASF SE Credit: BASF SE |
| Figure 4 Click to enlarge |
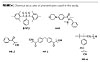
The structures of photoinitiators mentioned in this paper are shown in Figure 4.
Formulation
Table 1 shows an overprint varnish (OPV-1) formulation used for reactivity assessments. This formulation was further modified to provide a special overprint varnish (OPV-2) with reduced odor. The UV-curable formulation was applied using a 6 mm-thick wire-wound bar coater.
 Credit: BASF SE Credit: BASF SE |
| Table 1 Click to enlarge |
For migration tests, radical photoinitiators were incorporated in a radically curable blue flexo UV ink at concentrations ranging from 2 wt% to 6 wt%. The flexo UV-curable ink was applied on aluminum foil using a Prüfbau printing machine.
UV Irradiation
UV exposure was performed on an IST UV belt line equipped with a medium-pressure mercury lamp (from 80 to 200 W/cm) at different belt speeds and under different atmospheres (air or nitrogen with 500 ppm residual oxygen).
Migration
Test samples were immersed in ethanol 95%, placed in a pre-heated oven and left there for 2 hours at 70 °C. These conditions correspond to the extraction of the photoinitiator from the printed ink placed in direct contact with the food simulant, which does not give the real levels of migration but provides a worst-case scenario, allowing differentiation between several photoinitiators. Extracted photoinitiators were quantified by using reversed phase HPLC methods with UV detection. All experiments were performed in triplicate.
Curing Efficiency
Cure speeds were measured by determining dry rub resistance (DRR), by transfer testing and by performing the KMnO4 test (optical density measured after 1 min contact). Chemical modifications resulting from acrylate crosslinking were monitored by IR spectroscopy with an ATR unit for surface measurements (Digital FTIR Excalibur Spectrometer FTS 3000 MX). The reaction of the acrylate double bonds was determined quantitatively by monitoring the disappearance of the IR band at 1410 cm-1 and 810 cm-1 – bands characteristic of the acrylate double bond.(9)
Results and Discussion
Current Offer
 Credit: BASF SE Credit: BASF SE |
| Table 2 Click to enlarge |
The ability of a photoinitiator to migrate depends on its type, as reported in Table 2. The results of extraction tests performed on fully cured printed ink containing different photoinitiators show that, at a comparable molecular weight, the amount of extracted photoinitiator can vary by a factor of more than five, depending on type.
A second factor controlling migration levels is clearly molecular weight. With their enhanced mobility, it is mainly small molecules that are involved in contaminated beverages.
 Credit: BASF SE Credit: BASF SE |
| Table 3 Click to enlarge |
Thus, increasing the molecular weight of the photoinitiator is one alternative way of reducing its mobility and consequently its ability to migrate. However, this is achieved at the expense of reactivity (Figure 6), as shown with high-molecular- weight benzophenone photoinitiators whose structure is shown in Figure 5.
Those results associated with the molecular weights reported in Table 3 show that there is a direct link between cure speed and molecular weight (HMW-1 and HMW-3), and that one way to address the loss of reactivity is to incorporate lower-molecular-weight species (HMW-1 and HMW-2). However, this alternative carries with it the risk of losing the benefit of using high-molecular-weight components, as again these small species are able to migrate even at low concentrations.
 Credit: BASF SE Credit: BASF SE |
| Figure 5 Click to enlarge |
In all cases, however, reactivity is significantly lower than that of HK-3 and HK-4, which themselves are ranked as low-emission photoinitiators. Reducing their concentration could allow migration levels to be reduced while achieving a similar cure speed to that of HMW photoinitiators.
These results clearly show that thus far no suitable product, i.e., a photoinitiator with good reactivity and low/no emission, is available on the market.
Development Work
 Credit: BASF SE Credit: BASF SE |
| Figure 6 Click to enlarge |
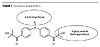
The structure of HK-3 was modified as shown in Figure 7 to give a high-molecular-weight photoinitiator called MacroPICS so that, once exposed to light, it generates heavy and/or crosslinkable breakdown products with very low migration potential and highly volatile breakdown products that simultaneously minimize the migration potential of the photoinitiator and of its by-products and ensure that there is no persistent odor. The molecular weight of MacroPICS lies between 900 and 1200 g/mol.
In the first step, reactivity of MicroPICS was assessed and compared to that of HK-2 (worst case in terms of emission but used as a reference for reactivity), HK-3 and a combination of benzophenone and HK-4. The reactivity of MacroPICS under these conditions is basically very similar to that of standard photoinitiators (Table 4), confirming that it is practically possible to combine high molecular weight, low polydispersity index and adequate reactivity. Such a product could allow a reasonable photoinitiator concentration, whereas concentrations of up to 15% of the HMW photoinitiators mentioned above are usual.
 Credit: BASF SE Credit: BASF SE |
| Table 4 Click to enlarge |
Extraction experiments were conducted in parallel on UV-printed samples containing MacroPICS and compared to HK-3: the results reported in Table 5 confirm that the increase in molecular weight significantly reduces the ability of the photoinitiator to be extracted from the film under drastic extraction conditions.
 Credit: BASF SE Credit: BASF SE |
| Table 5 Click to enlarge |
Odor developing from films containing MacroPICS and other photoinitiators was also assessed. Two curing conditions were defined: in one case, all films were thoroughly cured; in the other, films were cured at the maximum speed that still gave tack-free surfaces (borderline conditions). Again MacroPICS shows by far the best behavior (Table 6).
Oxygen Inhibition
 Credit: BASF SE Credit: BASF SE |
| Table 6 Click to enlarge |
Developing highly reactive HMW photoinitiators is the key to minimizing migration, but it is also essential to select the right curing conditions. We examined the influence of the curing atmosphere, which directly affects the efficiency of the curing process. Indeed, atmospheric oxygen interferes with free-radical UV-curing processes: the free radicals formed by the photolysis of the initiator are rapidly scavenged by O2 molecules to yield peroxyl radicals,(10,11) which do not polymerize the acrylate double bonds and can therefore not initiate or participate in any polymerization reaction. To surmount this issue, which directly impacts cure speed, additional amounts of photoinitiator are traditionally introduced into the formulation, allowing the photoinitiator to compete more efficiently against oxygen scavenging.
Another well-known practice to overcome this problem, which is unfortunately not sufficiently used on an industrial scale, is the utilization of an inert atmosphere, usually nitrogen or carbon dioxide.(12) Working under inert atmosphere has many advantages:
- it allows a significant reduction in the amount of photoinitiator required, while maintaining stable cure speed;
- free radicals are more likely to participate in the curing process and to be grafted to the network;
- a limited number of by-products is generated; and
- last but not least, crosslinking density is much higher.
All these elements help to reduce photoinitiator migration levels.
 Credit: BASF SE Credit: BASF SE |
| Figure 7 Click to enlarge |
The behavior of MacroPICS was thus investigated under inert conditions. In particular, the concentration required under nitrogen to match the cure speed reached when exposed under air was determined.
Figure 8 shows that working under nitrogen allows the photoinitiator concentration to be reduced by a factor of more than 10, as acrylate conversion is close to 90% for the same UV dose, with both 5% MacroPICS cured under air and 0.2% MacroPICS exposed under oxygen-reduced conditions. This very low concentration combined with a low-emission photoinitiator leads to extremely low amounts of extractables.
 Credit: BASF SE Credit: BASF SE |
| Figure 8 Click to enlarge |
Conclusion
Until now, the only solutions suggested were not satisfying in terms of emission or reactivity, and the poor purity of commercial high-molecular-weight photoinitiators entailed risks that converters were not aware of. Along with a high molecular weight, which makes it unlikely to migrate extensively, the newly developed MacroPICS photoinitiator allows a significant reduction in concentration while still maintaining good reactivity, especially under inert conditions.
These results provide a real understanding of the value that can be extracted from UV-curing technology by the printing industry when combining the right photoinitiator with the right processing conditions. The packaging industry gains a new vision, as it becomes possible to achieve very low emission levels using this technology. This thorough work should open up new application fields to UV-curing and allow significant growth for food applications in the flexible packaging, folding carton and label markets.
Acknowledgments
The authors would like to thank Dr. G. Weidenbrück and A. Metzger for checking the article, as well as B. Spony and G. Haller for the experimental work.
This article is based on the presentation “UV-Curing Systems for Sensitive Applications” given at the European Coatings Congress, 2009, in Nuremberg, Germany, by Katia Studer, BASF SE.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






