Without some type of protective surface coating, most wood will deteriorate very rapidly during outdoor exposure. Besides excellent resistance to weathering and UV, other features such as good adhesion, protection against liquid water ingress and a good balance between hardness and flexibility are key requirements for exterior wood coatings.
While solventborne alkyd binders have limited durability, they offer many of these properties and are therefore widely formulated into exterior wood coatings. Nevertheless, with increasing pressure to reduce the level of volatile organic emissions, acrylic-based waterborne systems are rapidly gaining popularity. Moisture protection, however, remains a critical parameter still to be optimized in the field of waterborne acrylic wood coatings.
While various additives exist to increase the short-term hydrophobicity of coatings, incorporating highly hydrophobic VeoVa™ vinyl ester monomers into an acrylic polymer is an ideal and durable solution for enhancing the final wood coating’s ability to repel water and resist weathering influences.1
Vinyl Ester of Versatic™ Acid 10
The vinyl ester of tertiary-branched mono-carboxylic acid containing 10 carbon atoms (VeoVa 10) is an ideal vinyl co-monomer for the production of latices for high-quality water-based coatings. VeoVa 10 finds its principal use as a hydrophobic comonomer in vinyl and acrylic polymerization. This vinyl ester molecule has a unique branched tertiary substituted a-carbon structure. Therefore, this alkyl neodecanoate (versatate) group is resistant to degradation by hydrolysis, as there is no hydrogen on the a-carbon, and in addition the bulky alkyl structure protects the ester function of the molecule. The branched tertiary structure with bulky and hydrophobic hydrocarbon groups provides the VeoVa 10 monomer (Figure 1) with a highly hydrophobic nature and low surface tension. Furthermore, the VeoVa 10 monomer does not degrade under the influence of UV light.2
The VeoVa 10 monomer polymerizes with various other comonomers through reaction of its vinyl ester functional group (Figure 1). In this way the specific properties of the VeoVa 10 monomer can be imparted to its copolymers.
VeoVa/Acrylic Copolymers (Veocryls)
VeoVa monomers readily copolymerize in emulsion with acrylate and methacrylate monomers to yield latices that are especially suitable for high-performance coatings. VeoVa monomers significantly upgrade key performance properties of acrylic coating systems, such as water and chemical resistance, adhesion and exterior durability.
Typical uses include wood stains and coatings, gloss paints, exterior masonry paints, roof tile coatings, anti-corrosion coatings and protective concrete coatings.
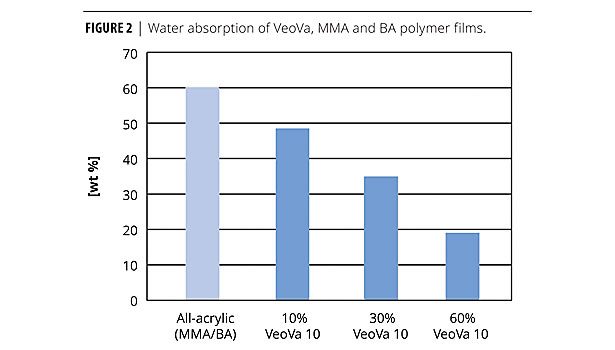
As can be seen in Figure 2, any incorporation of VeoVa 10 into an acrylic polymer leads to performance improvement, and properties further improve with increasing VeoVa content.3
Core-Shell Technology
The Core-Shell Process
A two-stage process whereby first all VeoVa monomers are reacted, followed by delayed addition of the rest of the acrylate monomers, creating a core-shell morphology, can be used. A small amount of ethylene glycol dimethacrylate (EGDMA) is reacted together with the VeoVa 10 to immobilize the core. Incorporation of specific functional monomers leads to stable one-pack self-crosslinkable polymers that will cure at room temperature upon drying of the coating.
A self-crosslinkable Veocryl polymer consisting of a hard core (Tg of 50 °C) and a soft shell (Tg of 0 °C) is a good example of a high-performance waterborne binder for wood coatings (Figure 3). Upon drying, a soft, flexible Veocryl matrix containing hard acrylic domains will be formed. The hard, pure acrylic core imparts good blocking resistance, and the combination with the soft-shell matrix provides very good balance between hardness and flexibility. With the hydrophobic bulky VeoVa monomer present in the shell, the polymer and the resulting wood coatings offer superior water resistance and water repellency, good adhesion and outstanding exterior durability.
Reduction of VOC
A careful selection of the monomers used in the shell enables the preparation of emulsions that form tough polymer films at temperatures as low as 0 °C. Wood coatings based on these polymers can be formulated with very low co-solvent levels and still form high-quality films at very low application temperatures.
As an additional benefit, these VeoVa/acrylic core-shell polymers are formaldehyde free and also have a low residual monomer content of < 500 ppm. This enables the development of a new generation of wood coatings with very low VOC levels.
Core-Shell Polymerization Procedure
Under nitrogen, demineralized water and surfactant were placed in the reactor and heated to 85 ºC. Then, the nitrogen flow was stopped, and demineralized water containing part of the initiator, potassium persulfate, was added to the initial reactor charge (IRC). This was followed by 2.5% by weight of pre-emulsion I. The temperature was maintained at 85 ºC, and a slight blue color and temperature increase were observed, which indicated that the polymerization was starting.
After 15 min, when this charge had polymerized, the remainder of pre-emulsion I was added over the course of 1 h. The remaining initiator solution was added simultaneously over a period of 3 h 15 min, via a separate line. Immediately after all of pre-emulsion I was added, pre-emulsion II was added over a period of 2 h. After 3 h of pre-emulsion addition, the reactor was kept at 85 ºC for 1 h post cooking. After post cooking, the latex was cooled down to 55 ºC and a conversion booster added in two separate doses, each lasting 30 min, followed by a 30-min post cook at 55 ºC. Low levels of residual monomer were achieved by using a redox booster based on tertiary-butyl hydroperoxide (tBHP) and the reducing agent Bruggolite® FF6 M in the presence of Fe III catalyst.
Finally, the latex was cooled to room temperature and filtered. During cooling, ammonia was added slowly to neutralize the latex. At room temperature, a biocide and crosslinker were added to the final polymer.
Composition and Latex Properties
A series of self-crosslinkable core-shell acrylic latices with a fixed core Tg of ~50 °C and a soft, flexible shell were prepared. The outer shell of the polymer particles was modified by incorporating either 2-ethylhexyl acrylate (2-EHA) or VeoVa 10 as hydrophobic monomers. In these latices, the monomers’ weight ratio of core to shell material was fixed at 30/70. For crosslinking, diacetone acrylamide was used at 3% m/m on total monomers, proportionally divided between the core and shell. The equivalent reactive adipic acid dihydrazide was post added to the latex at room temperature.
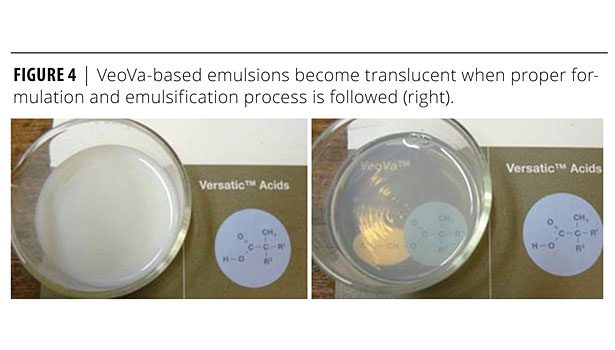
Emulsions with small particle size were expected to facilitate both the process of film formation as well as good penetration of the coating into the wood’s surface. The binder with the lower particle size (<85 nm) caused the latex to have a translucent aspect.4
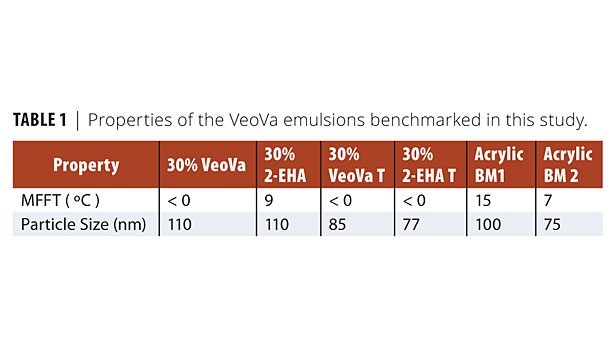
In addition, two representative commercial acrylic emulsion binders for wood coatings were evaluated (Table 1, Figure 4):
• 30% VeoVa: Core-shell polymer containing
30% VeoVa 10;
• 30% 2-EHA: Core-shell polymer containing
30% 2-ethylhexyl acrylate;
• 30% VeoVa: Core-shell polymer containing
30% VeoVa 10 translucent;
• 30% 2-EHA: Core-shell polymer containing
30% 2-ethylhexyl acrylate translucent;
• Acrylic benchmark 1 (BM1): A commercial, high-end acrylic emulsion polymer tailored towards the decorative wood coatings market;
• Acrylic benchmark 2 (BM2): A commercial, high-end acrylic emulsion polymer tailored towards industrial wood coatings market.
Formulation of Exterior
Protective Wood Stains
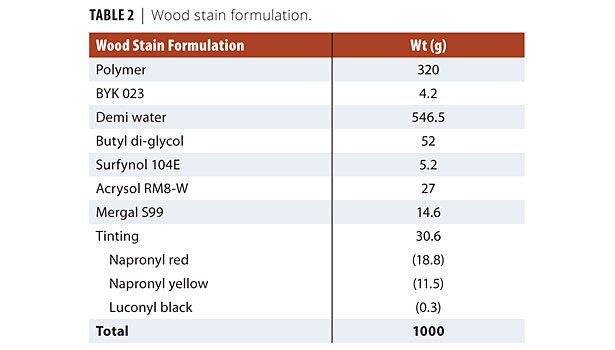
The different binders were formulated into wood stains (Table 2). A commercial (SB alkyd), high solids solvent-based alkyd stain was used as an additional benchmark. All paints were applied in three coats to Scots pine. Soft pine wood is one of the most demanding substrates because of its significant dimensional changes, which require the coating to adapt by expanding and contracting.
Experimental Procedure
Under gentle stirring, the latex was mixed with the defoamers (BYK 023 and Surfynol 104E), demineralized water, coalescing agent (butyl di-glycol) and fungicide (Mergal S99). At the end, the thickener solution was added step by step until a viscosity of about 100 KU was obtained. A mixture of the three colored pigments was made in order to obtain a consistent color. After 10 min of stirring, the predetermined amount of tinting mixture was added, still under slow stirring. Slow agitation was continued for another 15 min. The wood stains were allowed to equilibrate at 23 ºC for 4 days before application.
Blocking Resistance
The core-shell polymers prepared from the vinyl ester of versatic acid have equal or better blocking resistance than the commercial acrylic and alkyd benchmarks (Table 3). By adjusting the Tg of the shell, the blocking resistance can be further adjusted to meet specific needs.
Another important property for a wood coating is its adhesion to aged alkyds. All the emulsion polymer-based paints had excellent adhesion levels, in stark contrast with that of the solventborne alkyd, which clearly lacked adhesion under the applied conditions (ASTM D1653).
Liquid Water Permeability
Water absorption is a key driving force for expansion and contraction of wood, for mold growth and wood degradation. The liquid water uptake into wood was tested according to EN927-5 for wood stains applied on Epicea wood. The panels (6 replicates) were planed to a smooth and uniform finish by gentle sanding before three coats of wood stain were applied to the test face only. The front and sides of the panels were sealed with a solvent-based polyurethane white paint. The wooden panels were conditioned until constant mass was achieved. Before starting the absorption cycle, the panels were subjected twice to a leaching procedure.
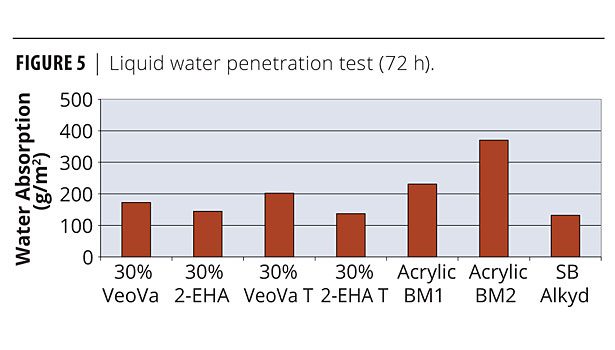
VeoVa-based wood stains have superior water barrier properties and significantly decrease the water uptake by the wood. From the water-based systems the VeoVa core-shell system has one of the lowest liquid water permeability values and is close in performance to the solvent-based alkyd. For the absorption test, the test panels were floated face down on the surface of the water for 72 h. The results were expressed as water absorption of coated wood panels in g/m2 test surface per 72 h (Figure 5). VeoVa-based wood stains have superior water barrier properties and significantly decrease the water uptake by the wood. From the water-based systems the VeoVa core-shell system has one of the lowest liquid water permeability values and is close in performance to the solvent-based alkyd.
Accelerated Weathering
Wooden articles intended for exterior use are exposed to severe UV radiation, rain and temperature changes. Coatings for exterior wood therefore need to have long-term weather stability. The different wood stains were evaluated in accelerated weathering for 2000 h according to EN 927-6 by an independent leading testing and certification institute for wood coatings (CATAS).
The weathering trial was carried out in a QUV Accelerated Weathering Tester from Q-Lab: 12 cycles of 24 h exposure to condensation (168 h total); 48 cycles of 3 h, consisting of 2.5 h of UV-A (340 nm) at 60 ºC and 0.5 h of water spraying 6-7 mL/min, which were alternated for a period of 2016 hrs.
The wood stain based on the VeoVa/acrylic core-shell polymer significantly outperformed the stain based on the 2-EHA-containing polymer and was superior to any of the other systems tested (Figure 6). After 2016 h, no sign of failure (blistering, cracking, flaking or chalking) could be observed for the VeoVa-based wood stain. Severe chalking and discoloration were observed for the wood stain based on the binder containing 30% of 2-EHA.
The good appearance retention of the VeoVa-based coatings after artificial weathering was not only visible, but could also be clearly quantified by minimal color change and excellent gloss retention of the formulated stain.
Again, the effect of VeoVa-10 on color protection is significant. The superior combination of UV stability with increased hydrophobicity as conferred by the versatic derivatives incorporated into the polymer are likely to have contributed to this improved appearance preservation.
Natural Weathering
Natural weathering was conducted in continental Europe weather conditions (Belgium). The VeoVa/acrylic core-shell systems have shown excellent performance during natural weathering (Figure 7). The tests are still ongoing, and have currently reached more than 2 years exposure without any significant performance loss.
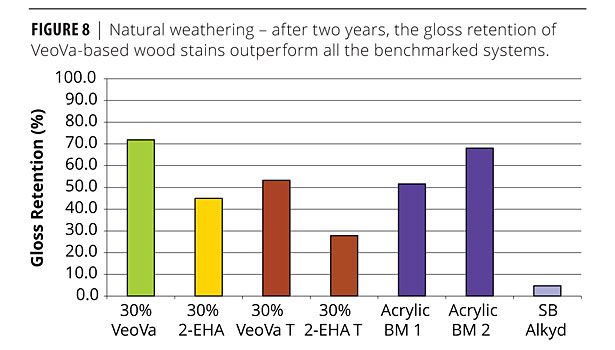
It is also noticeable that the panels coated with the VeoVa-based wood stain show the best gloss retention, surpassing several commercial grades considered industry standards (acrylic and alkyd benchmarks) (Figure 8).
Conclusions
From this work it can be concluded that water repellency and UV resistance of a binder are among the main components that govern the durability of the class of coatings studied. Of the hydrophobic monomers tested, it can be concluded that those based on the vinyl ester of versatic acid significantly enhanced the wood stains’ performance. This effect was demonstrated by a notable preservation in appearance of the coatings after artificial and natural weathering, as quantified by gloss retention and color change measurements.
VeoVa monomers contribute to the development of sustainable wood coating solutions characterized by:
• Superior and long-lasting water-repellent effect;
• Low liquid water up-take combined with good water vapor permeability;
• High exterior durability through excellent resistance to cracking, peeling and blistering, and good adhesion combined with adequate flexibility;
• Environmentally safe systems with low cosolvent demand, low residual monomer levels, and free of formaldehyde and APE surfactants. n
Note: Bruggolite® FF6M is a new reducing agent of Brüggemann Chemical. Visit www.brueggemann.com/english/restmonomere.html for more information.
References
1 Simal, F.; Hendrickx, H.; Verwee, A.; Nootens, C.; Decocq, F. Proceedings of the American Coatings Show, pp 1-6, 2008.
2 Corporate Web Site http://www.momentive.com/veova.
3 VeoVa Vinyl Ester Monomer – Momentive Technical Brochure 07/2011.
4 C. Koukiotis, I. D.; Sideridou. Progress in Organic Coatings, vol 63, 2008, pp 116-122.
For more information, e-mail david.vanaken@momentive.com.
This paper was presented at the 30th Biennial Western Coatings Symposium, October 23-26th 2011, Las Vegas, NV.









Report Abusive Comment