Fine Tuning Zero-VOC Architectural Formulations with a Multifunctional Amino Alcohol

While certain characteristics, such as dirt pick-up resistance, are more dependent on the monomer composition and ratio, additives such as multifunctional neutralizing agents can influence properties, including package stability, freeze/thaw and scrub resistance.
Formula Flexibility with Neutralizers
Multifunctional neutralizing agents can provide formulators the flexibility to optimize performance characteristics. The neutralizing agents can be inorganic or organic. Inorganic neutralizing agents are used in paint formulations only for pH adjustments. Organic alkanolamines are used as neutralizing agents, but they also bring multifunctional characteristics through the stages of a coating’s life. The multifunctional benefits of alkanolamines can be listed as co-dispersancy, neutralization, rheological performance improvement and VOC reduction through formula optimization. Additional in-can performance attributes include benefits like flash rust corrosion inhibition, improved shelf life and enhanced freeze/thaw resistance.
The role of alkanolamines as neutralizing agents is very important for today’s coatings to meet the quality challenges of zero-VOC (<5 g/L) systems. The alkanolamine, 2-amino-2-ethyl-1,3-propanediol (AEPD™ VOX 1000), in addition to the inorganic neutralizing agents ammonia, sodium hydroxide (NaOH), potassium hydroxide (KOH), sodium carbonate (Na2CO3) and potassium carbonate (K2CO3), were studied. The results showed the effects on key performance properties through the stages of a coating’s life. This information gives the formulator the flexibility required to overcome quality and performance challenges in zero-VOC coatings.
Study
Formulation
A semigloss formula without coalescents was used for this study. To represent the new formulation trends, an acrylic latex (Rhoplex™ VSR-2015) was selected as a primary binder for the semigloss formulation. Batches were made with AEPD VOX 1000 neutralizer, ammonia, NaOH, KOH, Na2CO3 and K2CO3. To show the co-dispersancy effects of the multifunctional neutralizer over inorganic dispersants, a combination of Tiona 596 TiO2 slurry and Polygloss 90 extender pigment was used as a pigment system.
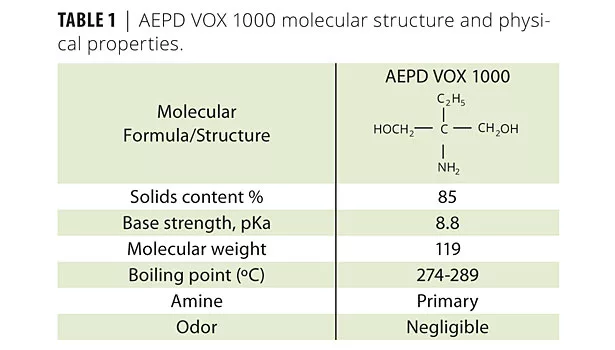
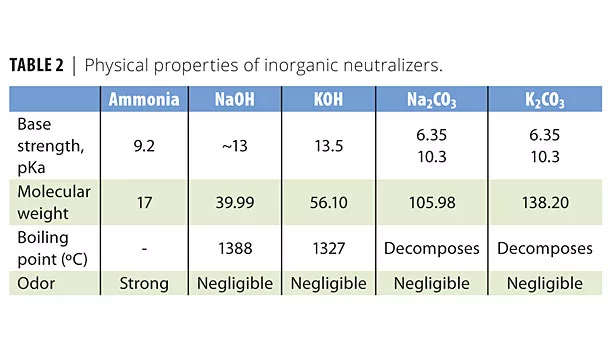
Table 1 shows that AEPD VOX 1000 is a primary amine with two hydroxyl functionalities. The factors that influence the efficiency of an amine are the molecular weight, the base strength and its solubility parameter. In considering amine efficiency, base strength (pKa) should be considered. Hydroxyl functionalities in alkanolamines provide the co-dispersion effects and improve other paint characteristics such as freeze/thaw and scrub resistance. Table 2 shows the physical properties of inorganic neutralizers.
Process
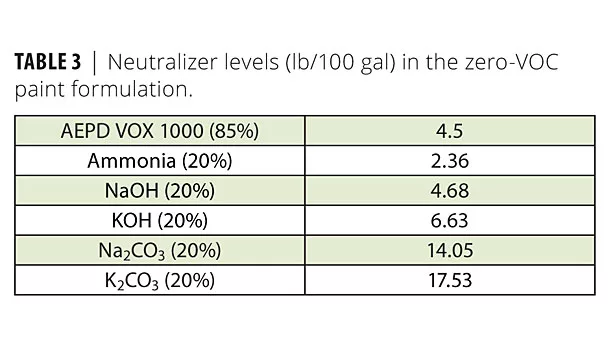
The semigloss paints were prepared with each neutralizer to meet the pH specification 8.5 to 9.0. The neutralizers (Table 3) were added initially in the grind phase before the extender pigment, Polygloss 90. However, Na2CO3 and K2CO3 were added in the let-down stage since the addition of these neutralizers gelled in the grind phase.
Formula Properties
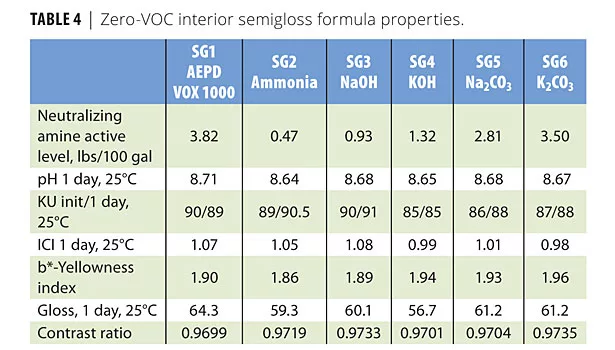
At 8.6-8.7 pH, potassium carbonate was the least efficient among the inorganic neutralizers. The initial formula properties were very similar for low- and high-shear viscosities except the low-KU viscosity of potassium hydroxide. The pigment efficiency of AEPD VOX 1000 was the highest compared to all inorganic neutralizers. These properties are listed in Table 4.
The gloss findings demonstrate that AEPD VOX 1000 neutralizer functions as a co-dispersant. This effect can also be measured through the extender pigment (Polygloss 90) adsorption studies.
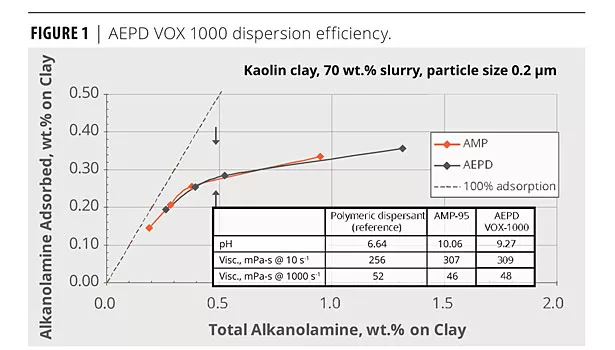
Figure 1 shows the weight percent of multifunctional neutralizer adsorbed onto pigment surface compared to another alkanolamine, AMP-95™ multifunctional neutralizer, which is commonly used as a co-dispersant in latex paint formulations. The polymeric dispersant that was used as a reference was an ammonium salt of a polycarboxylic acid. The co-dispersion effects of both alkanolamines on kaolin clay used in this study are similar.
Gloss Improvement
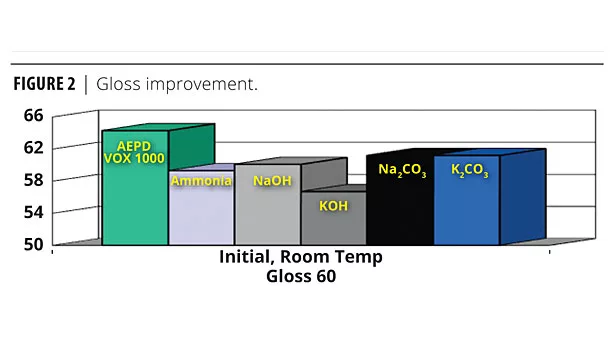
Gloss measurement of semigloss paint systems depends on the latex/pigment levels and particle sizes. Dispersants/surfactants affect the dispersion efficiency, leading to gloss improvement. Alkanolamines function as co-dispersants to enhance gloss.
Listed in order of formula gloss improvement (best to worst) are the neutralizing agents studied: AEPD VOX 1000>K2C03=Na2CO3>NaOH>Ammonia>KOH. Gloss improvement is shown in Figure 2.
Dispersion Stability
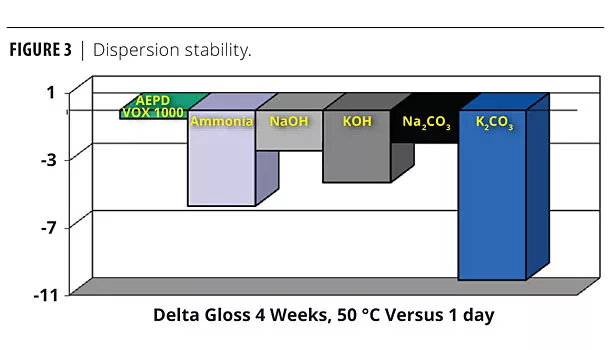
Neutralizing agents affect pigment dispersion stability. Figure 3 shows that the AEPD VOX 1000 system had very stable gloss over aging after four weeks at 50 °C. Listed in order of gloss stability after four weeks (best to worst) are the neutralizing agents studied: AEPD VOX 1000>Na2CO3
>NaOH>KOH>Ammonia>K2CO3.
pH Stability
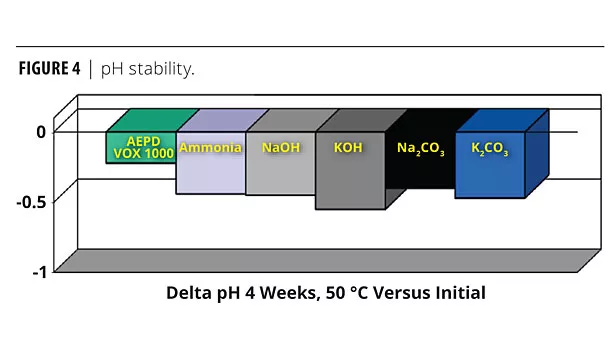
The multifunctional neutralizer shows excellent pH control after four weeks aging at 50 °C. The organic neutralizers provide better pH stability than inorganic neutralizers (Figure 4). Listed in order of pH control after four weeks (best to worst) are the neutralizing agents studied: AEPD VOX 1000>Na2CO3>Ammonia>NaOH>K2CO3>KOH.
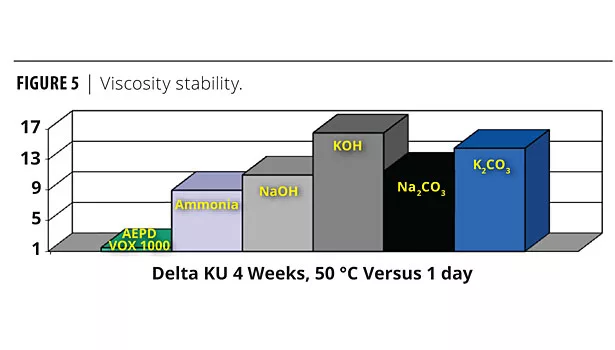
Viscosity Stability
The multifunctional neutralizer showed excellent viscosity stability after four weeks at 50 °C. The systems with inorganic neutralizers showed very poor viscosity stability after four weeks at 50 °C (Figure 5). Listed below in order of viscosity stability after four weeks (best to worst) are the neutralizing agents studied: AEPD VOX 1000>Ammonia>NaOH>Na2CO3>K2CO3>KOH.
Formula Performance
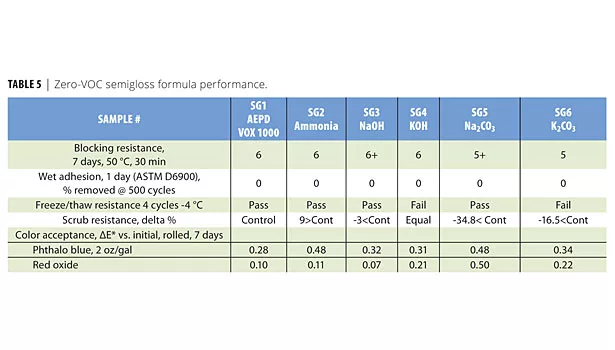
The zero-VOC formulations have shortcomings on some of the paint properties that are listed in Table 5. Performance testing on blocking resistance, freeze/thaw resistance and scrub resistance showed some differentiation with varying degrees among neutralizers.
Freeze/Thaw Resistance
Freeze/thaw testing using ASTM D2243 specifies a temperature of -18 °C (0 °F). Since zero-VOC formulations with no ethylene or propylene glycols fail this test at -18 °C, all formulations were tested at a borderline temperature of -4 °C, which shows the differentiation between formulas. Among all neutralizers, potassium hydroxide and potassium carbonate failed the test.
Scrub Resistance
The scrub resistance of the paint formula containing AEPD VOX 1000 is increased by >34.8% compared to sodium carbonate, and >16.5% compared to potassium carbonate (Figures 6 and 7).
Conclusions
This study showed that alkanolamines can improve the pigment dispersion efficiency and dispersion stabilities in paint formulations. The alkanolamine AEPD VOX 1000 multifunctional neutralizer exhibited better pigment dispersion efficiency, dispersion, pH and viscosity stabilities than all inorganic neutralizers. Among inorganic neutralizers, sodium and potassium carbonates exhibited poor water resistance, with sodium carbonate exhibiting the lowest scrub resistance. Overall, KOH exhibited the lowest pigment dispersion efficiency, pH and viscosity stability. Blocking resistance was slightly worse with sodium carbonate and potassium carbonate. The freeze/thaw resistance with potassium hydroxide and potassium carbonate was poor relative to all other inorganic neutralizers and AEPD VOX 1000.
There were also process differences observed among inorganic and organic neutralizers. The addition of potassium and sodium carbonates gelled in the grind phase. The addition of AEPD VOX 1000 in the grind phase can reduce the process time due to the co-dispersion effect, which can be observed with a reduction in grind phase viscosity.
Although the formulations can be optimized for some properties listed above, there are a wide range of properties such as gloss, scrub resistance and freeze/thaw resistance that have direct effects on latex formulations in addition to the process benefits.
The demand from consumers and more stringent industry regulations to produce products with low to zero VOCs and virtually no odor creates challenges for paint formulators to meet quality standards. Based on increasing technical constraints, new formulations are much more dependent on additive functionalities. Alkanolamines can provide performance versatility, including the optimized pigment dispersion, improved pH stability, improved rheology performance and many other improved properties. This study demonstrates that the multifunctional neutralizer offers formulators technical options to overcome some of the challenges of today’s latex formulations with an excellent environmental health and safety profile and virtually no odor. n
Acknowledgements
The author would like to extend her acknowledgements to Dow Performance Additives Research and Development Technologists John Quinn and Carol Nash for their contribution to lab work for this study.
™Trademark of The Dow Chemical Company or an affiliated company
Polyglass 90 is a registered trademark of KaMin LLC
Tiona 596 is a registered trademark of Millennium Chemicals
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




