Influence of Nanoclay on Elastic and Adhesion Properties in Polyurethane Coatings
Current automotive coatings systems consist of several layers, such as an adhesion-promoting layer, primer, basecoat and clearcoat. The bottom layer, i.e., the adhesion-promoting layer, plays a vital role in terms of providing better adhesion over substrates such as mild steel, aluminum and galvanized iron, and also provides a highly resistant layer to environmental conditions. In general, primers are based on different resin chemistries (alkyd, epoxy and polyurethane) and pigments (anticorrosive pigments and extenders), which depend on the application area, such as decorative or marine applications. The major ingredients in undercoats are extender/filler pigments, which vary in size and shape and are essential for undercoat properties like filling, sanding, flow and durability.
Most undercoat primer systems consist of larger-particle-size extenders like china clay, talc, chalk and barytes. These impart matting, filling and moderate resistance properties to the paint system. But these extenders also provide a less-flexible system with poor hiding properties. However, at the nanoscale level, the size and shape of nanoextenders are different and may provide better coating properties.
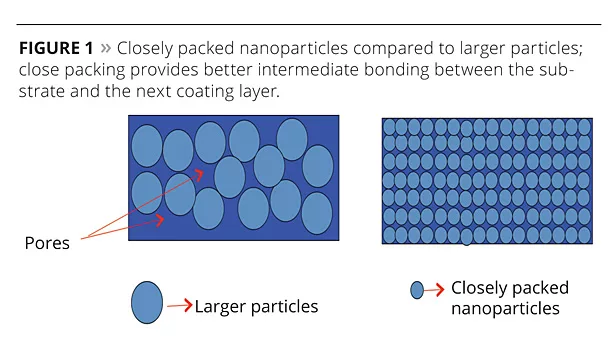
Nanomaterials have a relatively large surface area. As particles decrease in size, a greater number may be found at the surface, creating a more reactive surface than achieved with larger-size particles (Figure 1). Since nanoparticles provide a larger surface area and quantum effects, it is expected that replacing existing larger-size extenders with nanosize extenders could provide better coating properties. This article discusses the results of a study using nanoclay in place of larger-size extenders.
Experimental
Synthesis of Nanoclay-Based Coatings
Nanoclay, which is organically modified nanodispersable layered silicate with an average particle size of 25 nm (Al2O3, 2SiO2, 2H2O), was used for the study. The synthesis of nanoclay coatings was carried out in two steps.
1. Nanoclay Dispersion
Generally, dispersion of nanoparticles is extremely difficult due to their larger surface area. Polyurethane coatings were selected, due to their excellent durability. Acrylic polyol (80% on solids) and polyester polyol (20% on solids) were used as dispersing resins. The following samples were dispersed until a fineness of gauge less than 20 microns was achieved:
· 0% nanoclay;
· 2% nanoclay; and
· 5% nanoclay.
2. Preparation of Nanoclay PU Coatings
The above dispersions were mixed separately with trimer isocyanate, and the OH-NCO ratio was maintained around 1:1.5 for all experimental samples. Then all the samples were sprayed directly over a mild steel substrate and finished with a solventborne topcoat.
Test Methods and Results
The following properties were evaluated after seven days of curing.
Pendulum Hardness
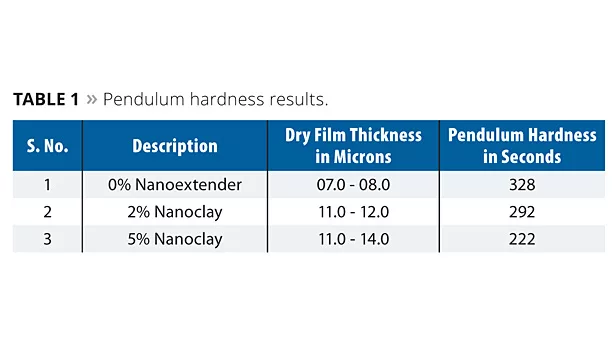
The cured adhesion-promoting layer based on nanoclay applied over a mild steel substrate was tested for pendulum hardness. A BYK Gardner pendulum hardness tester was used to measure the surface hardness of the coatings. The weight of the Persoz pendulum was 500 gm. The measure of Persoz damping was the number of oscillations that were recorded with a pendulum deflection of 12° until a drop to 4°. The results are shown in Table 1 and Figure 2.
The pendulum hardness evaluation results are essential to know the surface roughness/smoothness level. Nanoclay is finer and more closely packed together, which makes it prone to inferior interlayer adhesion properties. However, the results showed that the smoothness level decreases with an increase in nanoclay. It clearly indicates that 2% and 5% additions of nanoclay create roughness, which is essential for adhesion to the next layer.
Dry Adhesion, ASTM 3359 Method B
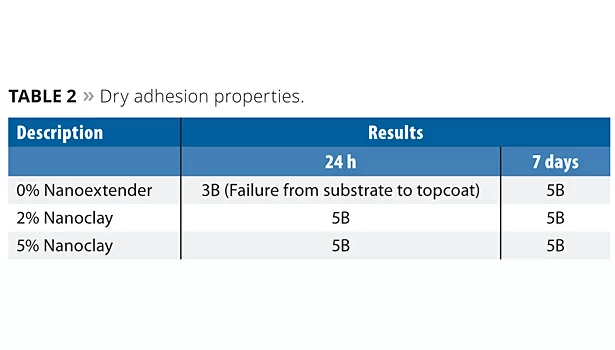
The applied systems were tested for adhesion after 24 h and 7 days. A crosshatch pattern was made through the film to the substrate. A distance of 2 mm was maintained between the lines. Pressure-sensitive tape was applied over the crosshatch area and smoothed until no air inclusion was visible. Then the tape was pulled from the test panel at an angle of 45°. Adhesion was judged on a scale of 0B-5B, where 5B=no failure; 4B=0-5% failure; 3B=5-15% failure; 2B=25-35% failure; 1B=35-65% failure and 0B= above 65% failure. The results are shown in Table 2.
The results show that dry adhesion improved significantly with the use of nanoclay. However, a further increase in the quantity of nanoclay from 2% to 5% resulted in similar levels.
High-Pressure Humidity Test (SAAB STD-3976-98)
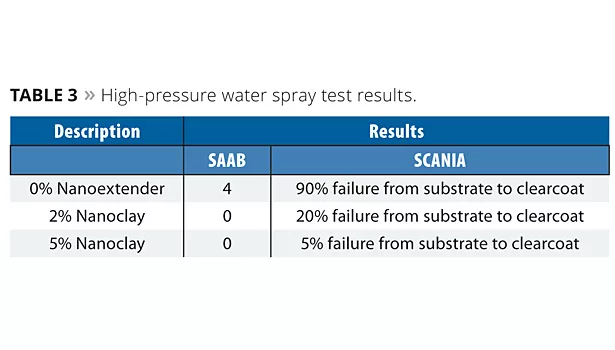
The cured samples were tested for high-pressure water spray as per the SAAB7 and SCANIA methods. The painted substrates were subjected to a water jet with fixed pressure and temperature as per SAAB (45 ºC/80 bar pressure) and SCANIA (55 °C/120 bar pressure) methods. The distance between the nozzle of the spray and the panel was fixed to 10 cm. A cross cut was made to the paint film through to the substrate before exposing to high pressure. The ranking method for SAAB is: 0 = no failure and 5 = complete failure. Results are shown in Table 3.
The results reveal that incorporating nanoclay improves adhesion properties between coating layers. In particular, the 5% concentration level exhibits better adhesion properties than the 0% concentration level.
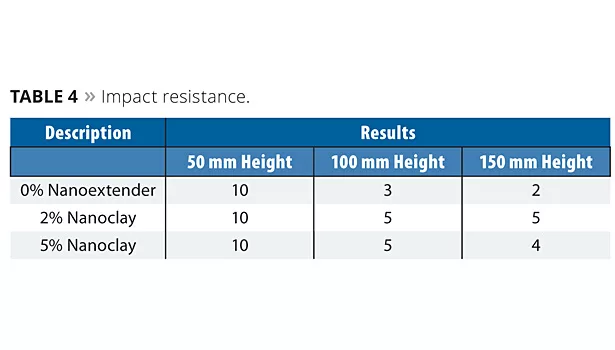
Impact Test (ISO6272)
The cured coating film was tested for indirect impact resistance. A mass with an indenter was guided by a graduated tube that fell from variable heights, from 1 mm to 1000 mm. The falling weight was 1 kg and the indenter diameter was 2 cm. Results are shown in Table 4.
The samples tested at a 50 mm height showed no cracks at the impact area. When the height was increased to 100 mm, minor cracks appeared. However, nanoclay-based samples showed comparatively better results than those with 0% nanoextender. A similar trend was exhibited at 150 mm height.
From the above results, coating flexibility improves slightly with the use of nanoclay. However, a further increase in the quantity of nanoclay from 2% to 5% resulted in similar levels.
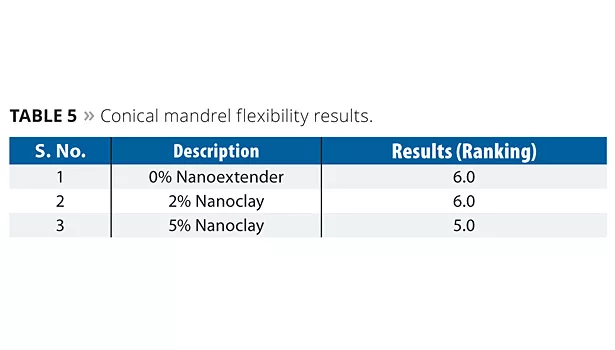
Conical Mandrel Test (ISO-6860)
The cured panels were subjected to bending using a conical mandrel. The mandrel width was 5 mm to 230 mm, the total mandrel length was 20.4 cm, and panels were clamped at the 5 mm point. The painted panel was bent through 180° at a steady rate without jerking for 1 to 2 seconds. The coating panel was immediately examined for cracking or detachment from the substrate using a magnifying lens. The length of cracking was measured in mm; the lower the unit the better the flexibility. Results were converted into a ranking, where 10 = no delamination of film and 1 = complete failure. Results are shown in Table 5.
The results indicate that 2% incorporation of nanoclay did not affect flexibility, whereas slight failure was noticed at a 5% concentration level.
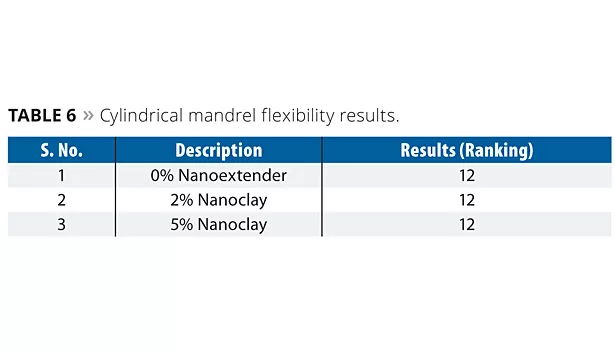
Cylindrical Mandrel Test (ISO-1519)
The samples were subjected to a cylindrical test, which determines coating film flexibility.
The 7-day-aged painted metal substrate was bent through 180° at a steady rate without jerking for 1 to 2 seconds over a cylindrical mandrel with the 25 mm diameter cylinder. The higher the rank (12) means the more resistance towards cracking, peeling and detachment. The results are shown in Table 6.
The results indicate that the flexibility of the coating films was unaltered with the incorporation of nanoclay. All the samples showed no cracking, peeling and delamination.
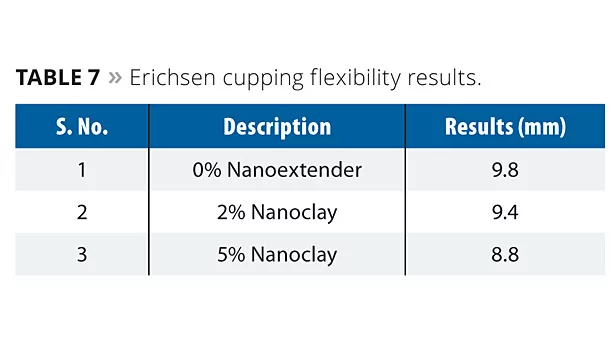
Erichsen Cupping Test (ISO-1520)
For the Erichsen cupping test, the cured films were tested for maximum depth (mm) of indentation at which the coating cracks or detaches from the substrate. The apparatus pressed the cap of the spherical punch into the test panel at a uniform speed (0.2 mm/s). Cracking or peeling of the coating was monitored and recorded (Table 7).
The sample without nanoextender exhibited better maximum depth, with 9.8 mm. The incorporation of nanoclay showed cracks at 9.4 mm for 2% concentration and 8.8 mm for 5% concentration. Thus, nanoclay has a negative influence on flexibility.
Conclusion
The study revealed that incorporating nanoclay improves dry adhesion as well as high-pressure hot water spray test properties significantly. The elastic properties remained unaltered in the conical mandrel and cylindrical mandrel tests. The impact resistance showed slight improvement, whereas the Erichsen cupping test showed a negative influence. Overall, it can be concluded that nanoclay incorporation is an option for coating formulators to enhance dry adhesion and impact resistance. n
Editor’s Note: Part 2 of this article, which evaluates water/humidity and corrosion resistance, will appear in the February issue.
Acknowledgement
The authors would like to acknowledge Dr. Sudhakar Dantiki, Managing Director AkzoNobel Automotive & Aerospace Coatings India Pvt. Ltd. and management who allowed us to use instrumentation.
References
1 Bentley, J.; Turner, G.P.A. Introduction to Paint Chemistry and Principles of Paint Technology, 1997. ISBN 0412723204.
2 Talbert, R. Paint Technology Handbook, 2007. ISBN 1574447033.
3 Woodbridge, P.R. Principles of Paint Formulation, 1991. ISBN 0412029510.
4 Deer, W.A.; Howie; Zussman, J. An introduction of rock-forming minerals, 2nd edition, Harlow, Longman, 1992, ISBN_0-582-30094-0.
5 Ullaman’s encyclopedia of industrial chemistry 6th edition, talc.
6 American Society for Testing and Materials. ASTM D3359 Standard Test Method for Measuring Adhesion by Tape Test; Philadelphia, PA, 1998.
7 SAAB STD3976-98 assessment of adhesion high pressure water test.
8 The mineral kaolinite – mineral galleries.
9 Weismantel, G.E. Paint Handbook.
10 Kapole, S.A.; Kulkarni, R.D.; Sonawane, S.H. Performance Properties of Acrylic and Acrylic Polyol–Polyurethane-Based Hybrid System Via Addition of Nano-CaCO3 and Nanoclay, Article first published online, Feb. 9, 2011.
11Maji, P.K.; Guchhait, P.K.; and Bhowmick, A.K. Effect of Nanoclays on Physico-Mechanical Properties and Adhesion of Polyester-Based Polyurethane Nanocomposites: Structure–Property Correlations, Journal of Materials ScienceVolume 44, Number 21, 5861-5871.
12 García-López, D.; Gobernado-Mitre, I.; Fernández, J.F.; Merino, J.C.; Pastor, J.M. Properties of Polyamide 6/Clay Nanocomposites Processed by Low-Cost Bentonite and Different Organic Modifiers, ; Polymer Bulletin, Volume 62, Number 6, 791-800.
13Sabzi, M.; Mirabedini, S.M.; Zohuriaan-Mehr, J.; and Atai, M. Surface Modification of TiO2 Nano-Particles With Silane Coupling Agent and Investigation of its Effect on the Properties of Polyurethane Composite Coating, Progress in Organic Coatings
Volume 65, Issue 2, June 2009.
14 Parvinzadeh, M.; Moradian, S.; Rashidi, A.; Yazdanshenas, M.E. Effect of the Addition of Modified Nanoclays on the Surface Properties of the Resultant Polyethylene Terephthalate/Clay Nanocomposites. Polymer-Plastics Technology and Engineering2010, Volume 49, Issue 9, 874 – 884.
15Sung, L.P.; Comer, J.; Forster, A.M.;Hu, H.; Floryancic, B.; Brickweg, L.; Fernando, R.H. Impact of Nanoparticles on the Scratch Behavior of a Polyurethane Coating.
16 Wypych, G. William Andrew Publishing, 2001, Handbook on Solvents.
17Wypych, G. William Andrew Publishing, 1999, Handbook of Fillers.
18Mezger, T.G. The Rheology Handbook, February 2011 ISBN: 9783866308640
19Westhues, U.M. Polyurethane Coatings, Adhesive and Sealants, November 2007.
20Muller, B. Understanding Additives, January 1970 ISBN: 9783866308688.
21 Poth, U. Automotive Coatings Formulation, May 2008 ISBN: 9783866309043.
22Schulz, U. Accelerated Testing, November 2008, ISBN: 978386630908.
23Poth, U.; Schwalm, R.; Schwartz, M. Acrylic Resins, August 2011, ISBN: 9783866308572.
24Gysau, D. Fillers for Paints, March 2011, ISBN: 9783866308701.
25Sander, J. Anticorrosive Coatings, July 2010 ISBN: 9783866309111.
26 Mischke, P. Film formation in Modern Paints, December 2009 ISBN: 9783866308619.
27Oyarzún, J.M. Pigment Processing, May 2000, ISBN: 9783878705567.
28 Dietrich, R. Paint Analysis, August 2009, ISBN: 9783866309128.
29Bhushan B. (Ed.). Springer Handbook of Nanotechnology, 2nd revised and extended edition. Springer Science + Business Media, Inc. Berlin, Heidelberg, New York 2007.
30Dosch H.; Van de Voorde M.H. Gennesys. White Paper. Grand European Initiative on Nanoscience and Nanotechnology Using Neutron – and Synchrotron Radiation Sources. Max-Planck-Institut für Metallfor-schung, Stuttgart 2009; www.mpi-stuttgart-mpg.de (Acces-sed 26 April 2010).
31Comyn, J. Adhesion Science, Royal Society of Chemistry Paperbacks, 1997.
32Kinloch, A. J. Adhesion and Adhesives: Science and Technology; Chapman and Hall, 1987.
33 Israelachvili, J.N. Intermolecular and Surface Forces; Academic Press: New York, 1985.
34 Newby, B.Z.; Chaudhury, M.K.; Brown, H.R. Macroscopic Evidence of the Effect of Interfacial Slippage on Adhesion. Science 1995, 269 (5229): 1407–9.
35 Majmuder, A.; Ghatak, A.; Sharma, A. Microfluidic Adhesion Induced by Subsurface Microstructures. Science 2007, 318 (5848): 258–61.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!







