Isosorbide: A Sustainable, Safe, High-Performance Plant-Based Feedstock for Coatings

Better resistance to UV light, abrasion, scratching and impact. Better adhesion and improved properties for water-based formulation. The demands on today’s coatings, adhesives, sealants and elastomers (CASE) create a long list of desirable properties for a ‘would be’ feedstock. Step forward isosorbide. Perfectly safe and produced from an annually renewable feedstock, isosorbide has a unique combination of properties that offers excellent potential for a range of CASE applications.
What is Isosorbide and How is It Made?
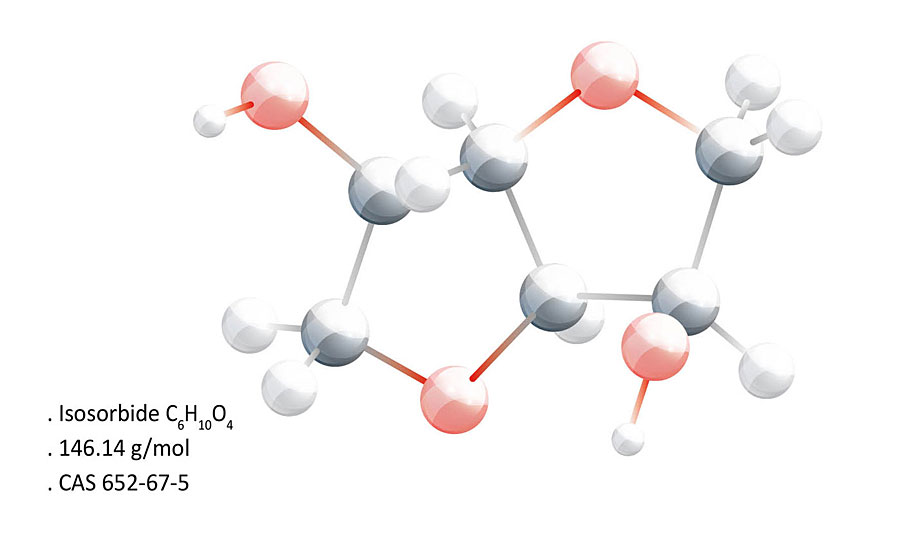
Isosorbide is a plant starch-derived bicyclic diol with rich functionality for a range of applications in the packaging, coatings, adhesives, sealants and elastomers (CASE), and automotive sectors (Figure 1).
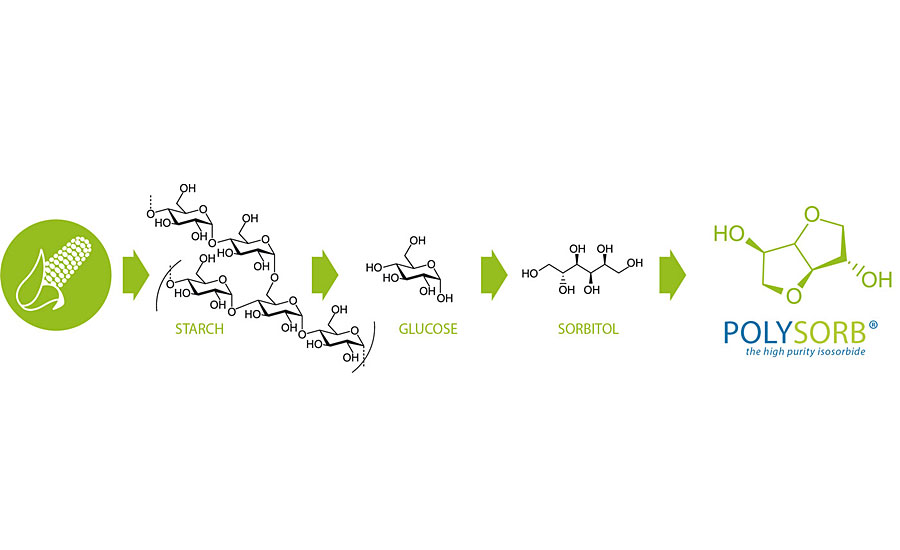
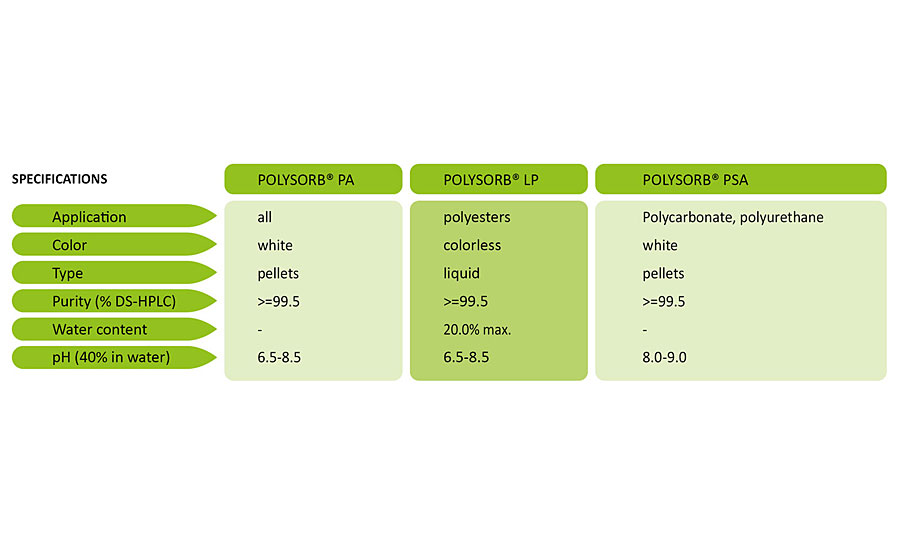
The schematic in Figure 2 illustrates how isosorbide is made. Starch from annually renewable feedstocks is hydrolyzed to produce glucose, which is then converted to sorbitol and on to isosorbide by hydrogenation. Over the last two decades, Roquette has refined and optimized this manufacturing route to produce stable, high-purity isosorbide in industrial quantities. The company’s flagship plant produces isosorbide of three different grades, each tailored to specific industrial applications (Table 1).
As a plant-based, sustainable feedstock with a carbon footprint of just 0.09 kg CO₂/kg of product, isosorbide is an attractive substitute for monomers such as bisphenol A, which has a carbon footprint around 60 times higher.* However, impressive environmental credentials are just the start. Isosorbide is:
- Non-toxic
- Non-carcinogenic
- Non-endocrine disruptor
- REACH compliant
- Suitable for food contact and for the manufacture of cosmetics and pharmaceuticals.
These attributes make the performance benefits of isosorbide of interest and value in a diverse range of applications.
What Are the Properties of Isosorbide and How Can It Be Used?
Isosorbide can be incorporated alongside other monomers to modify the properties of, for example, polyethylene terephthalate (PET), polycarbonates (PC), polyurethanes (PU) and epoxy resins. When used in this way it can enhance:
- Thermal properties, for example, increasing Tg, the glass transition temperature.
- Chemical resistance, to both water and organic solvents (such as acetones and esters).
- Optical performance, characteristics such as transparency and birefringence.
- UV resistance, reduced yellowing upon exposure to the sun.
- Mechanical properties, such as scratch, impact and bending resistance.
- Adhesion, a crucial property for the CASE sector.
- Appearance, for instance imparting high gloss.
Isosorbide can reduce the environmental burden associated with the use of existing materials, while maintaining performance, or enable the realization of new performance profiles, to meet the exacting requirements of different markets.
Let’s take a closer look at how isosorbide modifies the properties of some commonly used polymers and at the industrial value of the resulting materials.
Industrial Applications for Isosorbide
The CASE market is characterized by diversity, with products formulated specifically to meet precisely defined, sometimes unique applications. However, certain trends prevail across the industry, such as the move away from petroleum-derived ingredients and increasing use of water-based formulation, to reduce volatile organic compound (VOC) emissions, particularly for coatings. Isosorbide is a relatively new ingredient for CASE formulation that combines sustainability with technical performance, with specific advantages depending on the application of interest.
Polyurethane (PU) Coatings
PUs are made by reacting alcohols with two or more reactive hydroxyl groups per molecule i.e. diols, triols or polyols. 1,4 butanediol (BDO) is one such alcohol, routinely used as a chain extender, to increase the molecular weight of the PU. Substituting BDO with isosorbide creates coatings with higher heat resistance, better adhesion and improved impact and abrasion resistance.
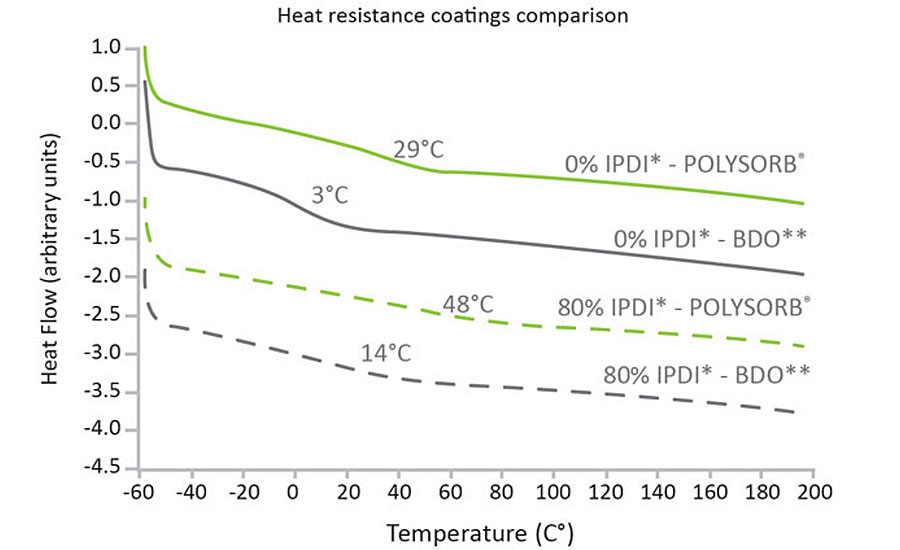

Switching from BDO to isosorbide increases the Tg of PUs made by reaction with isophorone diisocyanate (IPDI) (see graph in Figure 3), thereby improving heat resistance. Impact resistance is also significantly improved (see images in Figure 3), the absence of any red coloration showing that the isosorbide coating effectively prevents copper oxidation as a result of coating failure.
Epoxy Resins
The most common commercial epoxy resins are made by reacting epichlorohydrin and bisphenol A to form diglycidyl ether (BADGE -bisphenol A digylcidyl ether), which is then reacted with an amine to form the resin. Isosorbide can be directly substituted for bisphenol A to make isosorbide-based digylcidyl ether, to reduce toxicity and improve the quality of the finished product.
Isosorbide-based epoxies deliver an improved aspect, with a shiny gloss and better UV resistance than aromatic-based BADGE analogues. Appearance is further enhanced by the absence of the white haze that can appear with BADGE/amine systems in the presence of water; isosorbide-based epoxies are less sensitive to the classical amine carbonation phenomenon that produce this effect. Isosorbide epoxy derivatives can, in fact, be miscible and soluble with water, a unique and valuable feature. Just as with PUs, impact resistance is also improved and, in addition, isosorbide-based epoxy exhibits superior adhesion and deformation properties.


Displacing bisphenol A with isosorbide results in an epoxy resin with improved deformation properties, as evidenced by the images (Figure 4, left) from a conical mandrel bending test using an epoxy coating applied to a normalized Q panel. The isosorbide-based coating shows superior resistance to deformation and better adherence to the panel. The superior adhesive properties of the isosorbide-based epoxy are also demonstrated by comparative cross cutter testing (Figure 4, right).
Looking Ahead
Isosorbide is a plant-based, sustainable monomer with a low carbon footprint and significant potential for a wide range of applications. Substituting isosorbide for existing petrochemical ingredients not only lightens the environmental impact of industrial polymers, it also drives up performance. New types of aerosol packaging, touch screens with enhanced clarity and scratch resistance, better adhesives with improved UV and impact resistance. These are just some of the products that isosorbide is already beginning to deliver as polymer chemists and formulators learn exactly what it can do.
*Internal comparative study based on life cycle analysis methodology, peer-reviewed by an external˛auditor.
For more information, visit www.roquette.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






