New Resin Hybrid Technology for the Coatings Formulator
Silicon is the premier building block of inorganic materials. On earth, it makes up more than 25% of the planet's crust and is the second most common element after oxygen. The strong chemical affinity between silicon and oxygen means that they only occur naturally in the particularly stable form of Si-O-compounds as well as SiO2 as sand and quartz, because silicon forms very stable single bonds with oxygen, which is electronegative.
Polysiloxane Chemistry
Silicones are able to form a rigid three-dimensional SiO2 structure. Silicones, in chemical terms "polyorganosilioxanes," are polymeric compounds in which silicon atoms bond with oxygen as chains or networks. The remaining valences of silicon link with organic groups; the structure is similar to organically modified quartz. The organic groups open up this rigid quartz framework to a three-dimensional organic silicone linear structure as in polydimethylsiloxane chains (-[O-Si(Me)2]n - O-Si(Me)2 -). The approach of adding silicone-organic carbon-containing groups combines the durability of quartz with the many qualities of modern plastics.Analogous to silicone fluid, three-dimensional, brittle silicone resins are created by the polycondensation of trifunctional methyltrichlorsilane [Si(Cl)Me3]. Co-hydrolysis with dimethyldichlorsilane [Si(Cl2)Me2] and tetrachlorosilane [Si(Cl2)], respectively, produces softer or harder resin grades. Polysiloxane resins are formed through hydrolysis, co-condensation and polymerization reactions.
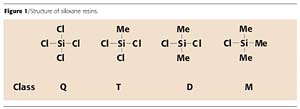
Siloxanes with a higher dimensional structure are typically produced by hydrolytic condensation reaction by being crosslinked with organochlorosilanes (Figure 1: type Q, D, T and M). Not only the number, but also the nature of the reactive and unreactive constituents in the monomers can vary, which gives the resins their special properties.
They can withstand continuous temperatures of 200 - 250 °C, and for short periods, up to 600 °C. In addition, they have ideal dielectric properties and very good mechanical qualities. The chemical nature of the (Si-O) units makes them extremely resistant to atmospheric or chemical breakdown and they are not affected by sunlight and ultraviolet attack.
The behavior of a mono- (M), di- (D), tri- (T) or tetra- (Q) functional building block in the synthesis of siloxane resins is described as follows. The mono-functional silanes are used as chain stoppers, the di-functional units (D) are used to build linear chains, and introducing tri- or tetra-functional units into the chain enables crosslinked structures.
The Value of Siloxane-Epoxy Hybrids to the Formulator
The versatility of siloxane chemistry has resulted in the formulation of an organic-inorganic siloxane hybrid binder with the siloxane as the matrix and epoxy and alkoxy functionality in the side chains. The novel chemistry of siloxane hybrids achieved by the chemical reaction of an aliphatic epoxy with a polysiloxane results in a combination product whose unique chemical and physical characteristics allow its use as a durable resin for the protective coatings industry.
A siloxane-epoxy hybrid resin combines the advantages of epoxy resin with the strength of the polysiloxane. This produces a two-component ambient curing thermoset system. The low-viscosity siloxane polymer in this resin can be formulated with low VOC and very high solids.
Application Testing
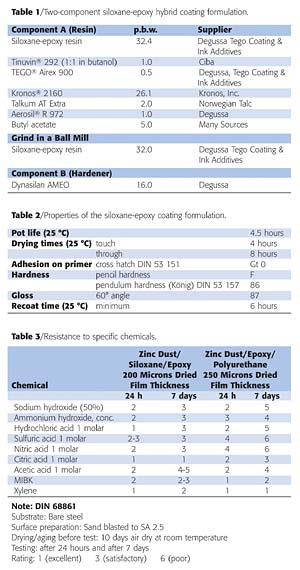
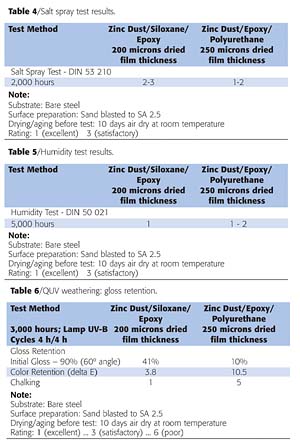
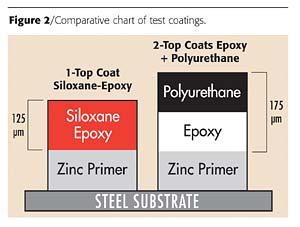
Because a major focus of the coatings industry is to reduce material and labor costs while obtaining improved corrosion protection, we tested a two-coat system consisting of 75 microns of a zinc-epoxy primer that was top coated with 125 microns of a siloxane-epoxy hybrid coating (Tables 1 and 2). This formulation was compared with a three-coat zinc-epoxy primer that consisted of 75 microns of a zinc-epoxy primer top coated with 125 microns of an epoxy, and 50 microns of polyurethane.Both formulations were evaluated to determine resistance to specific chemicals, and in accelerated corrosion testing such as salt spray and in a humidity chamber (Tables 3-5 and Figure 2).
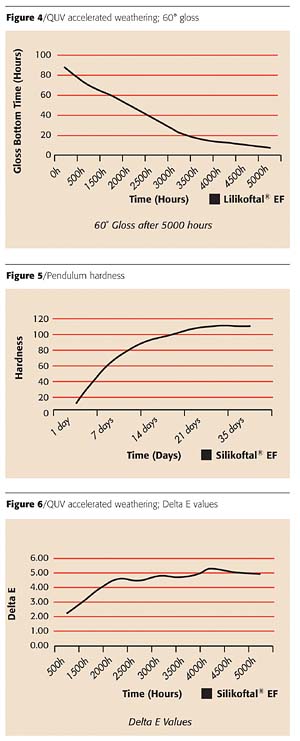
In addition to the comparative tests performed to determine resistance to corrosion, salt spray and humidity, further tests were conducted to determine the performance of the epoxy hybrid for adhesion, hardness, affect on gloss and the change in color shade (Figures 3-6; Tables 6,7). QUV accelerated weathering tests were conducted on this coating formulation to determine gloss and color retention. These tests (Figures 4 and 6), in combination with the specified acceptance criteria, are currently considered as severe performance test requirements for protective coating systems in the coatings industry.

Suggested Corrosion and Marine Applications
Combating corrosion can be an expensive and daunting challenge. High-solids siloxane-epoxy resin coating systems can be used in various applications including structural steel, storage tank exteriors, offshore platforms, marine structures, bridges, industrial plants, concrete walls and floors and the exteriors of railroad cars.This new epoxy siloxane coating system enables the zinc primer to be protected by a single topcoat based on the innovative siloxane-epoxy hybrid resin that combines the advantages of organic polymers and silicones in a single polymer.
This results in reduced application time, less overspray and less complex maintenance for corrosion protection. Furthermore, the reduced number of coats and overspray results in about 70% less solvent emission into the atmosphere.
In addition, the siloxane-epoxy coating provides excellent dirt-repellent and anti-graffiti properties. After the removal of the graffiti, there was no observable change in the gloss. For marine applications, like boat tops and underwater construction, we observed easy removal of fouling layers on the treated surfaces.
Conclusion
The chemically combined organic-inorganic hybrid resin presents an interesting opportunity to formulators as the prototype of a new class of materials in the design of high-performance industrial and specialty coatings with specific benefits:- High-volume solids (90%) and low VOC;
- High-build application characteristics by standard application equipment;
- Excellent color and gloss retention;
- Excellent corrosion resistance in two-coat systems;
- Easy removal of fouling layers of such special surfaces;
- Cost-effective two-coat alternatives to multiple-coat organic-based coating systems;
- Compliance with the most severe requirements of health, safety and environmental regulations
References
1. W. Noll, Chemie und Technologie der Silicone, Verlag Chemie, Weinheim (1968).2. D. Stoye, W. Freitag, Lackharze Chemie und Eigenschaften, Hanser Verlag München (1996).
3. H. Warson, C. A. Finch, Applications of Synthetic Resin Latices, 3 Volume Set.
4. P.K.T Oldring, N. Tuck, Resins for Surface Coatings, Wiley (2001).
5. S. Sawant and A. Wagh, Corrosion Prevention and Control (1991), p. 75 - 77.
6. C.H. Holl, conference CORROSION/86, Paper Nr. 31, Houston (Texas), NACE 1986.
7. C. Giudice and B. Del Amo, Corrosion Prevention and Control (1996), p. 43 - 47.
8. K. Keijman, PCE, 7, 1996, p. 26-32.
9. G. Reusmann, Farbe & Lack 107, 2001, 11, p. 78 - 84.
10. Silicone, Chemie und Technologie, Vulkan-Verlag Essen 1989.
11. Tego Chemie Service GmbH: TEGO Journal, 1999, p. 109-115.
12. Patents: JP 11106530, Deutsche Patentschrift 1520015, SILIKOFTAL‚ EW.
13. Dow: US-Patent 5,952,439 and European Patent EP 0590954 B1.
14. US-Patents 5,618,860 and 5,804,161.
15. European Coating Journal 5, 1999, p. 64-67.
16. European Coating Journal 4, 2001, p. 152-159.
17. Patent-examples: EP 3385550, EP 283009, EP 153500 and WO 9844018.
18. Silicone-Epoxy Hybrid-Binder from Tego Chemie Service GmbH: SILIKOFTAL‚ ED, SILIKOPON ED, SILIKOPON EF.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






