UV-Light-Sensitive Urethane Acrylate Oligomers

Urethane Acrylate Coatings
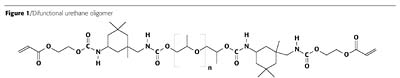
In the present paper we discuss UV-curable urethane acrylate coatings. The urethane acrylate oligomer is the main part of the coatings composition, which determines the properties of both the liquid and the cured coating. As an example, we present a structure of a rather common difunctional urethane oligomer (Figure 1).This oligomer is prepared by the reaction of isophorone diisocyanate (D) with polyol polypropylene glycol (P) and a subsequent endcapping of non-reacted terminal isocyanato groups with 2-hydroxyethyl acrylate (A) (Figure 2).
The two reactions mentioned above lead not only to the unique product ADPDA, with a chemical structure presented in Figure 1, but also to other products like ADPDPDA, ADPDPDPDA, etc., and low-molecular-weight (MW) product ADA.2 Urethane oligomer synthesis is performed in such a way that NCO equivalents are equal to equivalents of all the reactive OH groups. That way, independent of product distribution, there are no loose NCO or OH groups after completion of the reaction.
An isocyanate-terminated oligomer is commonly endcapped with 2-hydroxyethyl (meth)acrylate to obtain urethane acrylate oligomers.
Reactive diluent(s), which are usually mono- or difunctional acrylates of low viscosity, are added to the urethane acrylate oligomer in order to adjust the viscosity so that the coatings are pourable at room or elevated temperatures. Photoinitiators (PIs) and other necessary additives such as adhesion promoters (APs), pigments, etc., are dispersed in the final coating formulation. Coatings cured with electron beam do not require photoinitiators.

LS® Oligomers
We have noticed that many commercially available photoinitiators3,4 have primary, secondary or tertiary aliphatic hydroxyl groups, and they can react with isocyanates (Figure 3).Common photoinitiators include hydroxyalkyl substituted benzophenone, 2-hydroxyethyl-N-maleimide, coinitiators such as di-2-hydroxyethylmethylamine, etc. Two structures of commercially available silane APs with reactive amino and isocyano groups are also presented in Figure 3. Photoinitiators and adhesion promoters are added to coatings formulations at relatively low levels of a few percent. To prepare these light-sensitive oligomers, one may not simply add PI and AP to a formulation - they need to be grafted at a required level. As always, the total equivalents of NCO must equal the total equivalents of OH or (OH + NH2). Such oligomers, with grafted PI, we have named Light Sensitive (LS®) oligomers. By "light" we mean UV-light (UVA, UVB). Some colored grafted PIs (benzoin, isopropylthioxanthen-9-one, etc.) can produce free radicals upon absorption of visible light. It is possible to graft a photoreactive dye to an oligomer, and the oligomer will be visible-light sensitive. The latter means that a formulation will polymerize under irradiation with visible light.
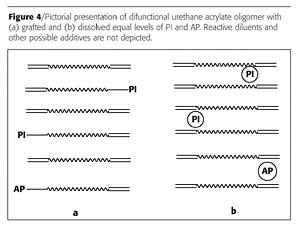
Figure 4 is a pictorial presentation of a difunctional urethane acrylate oligomer with (a) grafted and (b) dissolved PI and AP. It is evident that grafting occurs at the expense of acrylate groups. Usually an LS® oligomer has 2-10% less acrylate groups than its conventional analogue. Often grafting has a small effect on the physical properties of the liquid and cured coatings. For example, there is no difference between viscosities, Mw, molecular weight distribution (MWD), elongations-to-break and tensile properties. Test evaluation data of Bomar oligomers BR-144 and XP-144 LS® were within experimental error for these parameters in both the liquid and cured coatings. The XP-144 LS® oligomer is very similar to BR-144 with PI grafted to the oligomer. LS® oligomers are photoreactive; formulations based on LS® oligomers can be diluted up to two times with reactive diluent, and they are efficiently cured without the addition of extra PI. A typical PI level is approximately 4% in LS® oligomers.
The photoinitiator 1-[4-(2-hydroxyethoxyphenyl)]-2-hydroxy-2-methylpropan-1-one, or Irgacure 2959 from Ciba Additives (Figure 3), is of special interest. It has two reactive hydroxyl groups and can serve as a chain extender and as a PI in urethane acrylate coatings.5,6 The photocleavage of oligomer with Irgacure 2959 in its structure leads to two oligomeric radicals. Esacure One, a photoinitiator available from Lamberti has two tertiary hydroxyls, and can be grafted by two termini. Esacure One was grafted to difunctional aliphatic methacrylate BR-543M and trifunctional aliphatic urethane acrylate BR-144. Both oligomers with grafted PI cured efficiently. The same holds true for BR-144 with Irgacure 2959, which was chemically tethered to the oligomer by two OH groups.
Advantages of Grafting vs. Dissolution
First, in most cases UV cure does not result in a complete consumption of PI. Residual PI leaches from the cured coating, which is detrimental both to the coating and to the environment. It was experimentally demonstrated that in a number of LS® oligomers, the PI leaches into THF more than 20 times less than from a conventional analogue (experimental conditions were as close as possible).Secondly, many customers do not want to spend time on PI selection and subsequent dissolution. The latter may be tedious because many PIs are powders at room and elevated temperatures. Such customers prefer to have a LS® oligomer in order to cure the oligomer or a coating based on the LS® oligomer with their common processor, for example a Fusion processor with D- or H- bulb.
Third, in many cases the cure rate of LS® oligomers is often higher than the cure rate of similar oligomers with a dissolved photoinitiator at the same level. [Reactive diluent may be added at the same concentration to LS® and to conventional oligomer if all other cure conditions are the same.]
The cure of LS® oligomers with grafted benzoin was faster in all studied cases.7 Cure was monitored by disappearance of acrylate groups with RT IR. We speculate that formation of two C-centered radicals under photodissociation of initiator bound to the oligomer results at the first moment in the advanced stage of polymerization compared to a common photopolymerization. At the first moment after absorption of light and the primary photo-physical processes, an oligomeric free radical is produced accompanied by a low-MW radical. On the other hand, a standard photolysis of initiator dissolved in oligomer results at the first moment in the formation of two low-MW radicals. In this instance, only those low-MW radicals that have escaped diffusion-controlled self-termination in the solvent bulk react with oligomer to form an oligomeric free radical.

Oligomers endcapped only by photoinitiator and by adhesion promoter can be prepared separately and after that added to the main "usual" oligomer endcapped by acrylate groups.6 That is another way to prepare an LS® oligomer.
Chemical binding of PI to a polymer or oligomer can slightly alter its absorption spectrum and its reactivity (i.e., a quantum yield of photodissociation).3,4
There is a difference between LS® oligomers and polymeric PIs. LS® oligomers are randomly endcapped with a low level of PI moieties. Polymeric PI is polymer made out of PI moieties or it has a backbone with chemically tethered, repeated PI moieties. It is difficult to state upfront what will be the effect of the addition of polymeric PI or LS® oligomer on the cure rate compared to the cure of the same or very similar oligomer with a dissolved PI at the same level. There are observations that polymeric PI leads to a faster cure.9 On the other hand, one can expect that the macroradical, which is a PI fragment formed under photolysis and bound to a polymer chain, has lower mobility and thus is less accessible for acrylic double bonds.3,4 The same may hold true for LS® oligomers. The kinetics of the cure of coatings is an extremely complicated process, and kinetic predictions are not reliable. 10,11
There is a promising avenue of development of new coatings that does not need an addition or a grafting PI at all. These new oligomers, or reactive diluents, are photoreactive under UV irradiation, and a coating is cured "by itself" under UV-irradiation.12-14 Analysis of such coatings is beyond the scope of the present paper.

Conclusions
Oligomers with grafted photoinitiators (LS®) and adhesion promoters demonstrate, in many cases, superior properties compared to similar oligomers with dissolved photoinitiators and dissolved adhesion promoters. LS® oligomers usually undergo faster cure, they do not have leachable additives (residual-free photoinitiators) and they have strong adhesion to glass.7
Acknowledgment
The author is grateful to his colleagues at Bomar Specialties, J. DeSousa, Dr. R. Finch, J. Leon, K. Swiderski, and D. Zopf, for fruitful discussions.
References
1 Fouassier, J.-P. Photoinitiation, Photopolymerization, and Photocuring. Fundamentals and Applications; Hanser: Munich, 1995.
2 Swiderski, K.W.; Khudyakov, I.V., Ind. Eng. Chem. Res., 2004, 43, 6281.
3 Dietliker, K. A Compilation of Photoinitiators. Commercially Available for UV Today. SITA Technology, Leith, 2002.
4 Crivello, J.V., Dietliker, K. Photoinitiators for Free Radical Cationic & Anionic Photopolymerisation. Ed. Bradley. G. Vol. III, Wiley, Chichester, 1998, ch. II, p. 204.
5 Finch, R.W., unpublished results, 1999.
6 Purvis M.B.; Khudyakov; I. V.; Overton; B. J.; Gantt, T. W. US Patent 6,596,786.
7 Gantt, T.W.; Khudyakov, I.V.; Purvis, M.B.; Overton, B.J. Crawley, H.R. EP 1417244.
8 Khudyakov, I.V.; Overton, B.J.; Purvis, M.B. US Patent 6,171,698.
9 Fuchs, A.; Bolle, T.; Ilg, S.; Hüsler, R. In RadTech Europe. Conf. Proceedings, 2003, v. 1, pp 507-511.
10 Khudyakov, I. V.; Purvis, M.B.; Turro, N. J. In Photoinitiated Polymerization; Belfield, K.D.; Crivello, J.K., Eds; ACS Symposium Series 847; American Chemical Society, Washington, DC, 2003, pp 113-126.
11 Khudyakov, I.V.; Purvis, M.B., Overton; B. J. In Proceedings of RadTech 2002; Indianapolis, IN, pp 269-283.
12 Khudyakov, I.V.; Fox W.S; Purvis, M.B. Ind. Eng. Chem. Res. 2001, 40, 3092.
13 Hoyle, C.E.; Lee, T.Y.; Jönsson, S.; Guymon, C.A. In Proceedings of e/5 2004; Charlotte, NC.
14 Narayan-Sarathy, S.; Gould, M.; Zaranec, A.; Hahn, L. In Proceedings of e/5 2004; Charlotte, NC.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





