Preparation and Properties of UV-Curable Polyurethane Acrylate Resins for Metal Surfaces
UV-curable coatings are a boon to the paint industry, as they not only reduce VOCs but also speed up production processes and provide protective, durable and attractive finishes. UV-curable polyurethane acrylates have been synthesized from polyester polyol (1,6-hexanediol, adipic acid and ethylene glycol), toluene diisocyanate and three types of reactive diluents i.e., ethoxylated phenol monoacrylate (EOPA), 1,6-hexanediol -diacrylate (HDDA) and trimethylolpropane triacrylate (TMPTA). In this paper several formulations of UV-curable polyurethane acrylate resins were used to study the effect of reactive diluents on performance properties. It was observed from the results that UV-cured polyurethane acrylate resin films on metal surfaces exhibit excellent gloss (60°), mechanical, and chemical- and stain-resistant properties.
UV-curable coatings are a boon to the paint industry, as they not only reduce VOCs but also speed up production processes and provide protective, durable and attractive finishes. UV-curable polyurethane acrylates have been synthesized from polyester polyol (1,6-hexanediol, adipic acid and ethylene glycol), toluene diisocyanate and three types of reactive diluents i.e., ethoxylated phenol monoacrylate (EOPA), 1,6-hexanediol -diacrylate (HDDA) and trimethylolpropane triacrylate (TMPTA). In this paper several formulations of UV-curable polyurethane acrylate resins were used to study the effect of reactive diluents on performance properties. It was observed from the results that UV-cured polyurethane acrylate resin films on metal surfaces exhibit excellent gloss (60°), mechanical, and chemical- and stain-resistant properties.
Photo-curable formulations usually consist of multifunctional monomers, reactive diluents, oligomers and photoinitiators, which generate reactive species on UV exposure.5,6 Typically it is a segment of polyurethane oligomer tipped with acrylic functionality such as 2-hydroxyethylacrylate (HEA) or 2-hydroxyethylmethacrylate (HEMA).
The reactive diluents are acrylic monomers that are added to modify properties and to reduce viscosity of the precursor liquid.
The acrylated urethanes ideally combine the abrasion resistance, toughness, tear strength and good low-temperature properties of PU with the superior optical properties and weatherability of polyacrylates.7-9 The reactive diluent phase is more compatible with the hard segments of polyurethane rather than the soft segments, due to similar polarity and hydrogen bonding formation between the urethane NH and acrylate carbonyls.10
This paper considers the preparation and properties of UV-curable polyurethane acrylates. The NCO-terminated polyurethane prepolymers were synthesized from toluene diisocynate (TDI) and polyester polyol followed by tipping with 2-hydoxyethylmethacrylate (HEMA). In addition, three types of reactive diluents having mono-, di- and tri- functionality were used in different concentrations. A series of coating compositions having PU acrylate and reactive diluents were prepared, and their mechanical, chemical, optical and stain-resistant properties studied.

TDI (an 80/20 mixture of 2,6 and 2,4 isomers) was obtained from BASF and Narmada Chematur. The purity was 98% and was used as such. Laboratory reagent (LR)-grade adipic acid, 1,6-hexanediol, ethylene glycol, 2-hydoxyethylmethacrylate (HEMA) and hydroquinone of M/s Thomas Baker, Mumbai, were used. Reactive diluents were procured from M/s Cognis, Thailand, and photoinitiators were procured from M/s Ciba chemicals.
 Polyester Polyol Synthesis
Polyester Polyol Synthesis
Initially ethylene glycol, adipic acid and 1,6-hexanediol were placed in a three-neck, round-bottom flask in the molar ratio of 1.6 : 3.4 : 4.0 respectively. The esterification reaction was catalyzed by adding dibutyl tin oxide (0.1%) as a catalyst. The temperature was maintained at 140 – 150 °C. An inert atmosphere was maintained by blanketing the reaction mixture with nitrogen. Acid and hydroxyl values (Figures 1 and 2) of the resin were checked at fixed time intervals.
Urethane Acrylate Synthesis
The reaction flask was charged with 2.2 moles of TDI under dry nitrogen gas at 50 °C; a blend of 2.4 moles of HEMA with hydroquinone (50 ppm) was added continuously over a period of one half hour while maintaining a maximum temperature of 60 °C. After the addition was complete, the reaction mass was kept at 60 °C for half an hour to assure the complete conversion of the hydroxyl group of HEMA.
 Polyurethane Acrylate Synthesis
Polyurethane Acrylate Synthesis
The temperature of the reaction flask containing urethane methacrylate precursor was maintained at 50 °C, and a mixture of hydroxyl functional polyester polyol and hydroquinone (50 ppm) was added over a half-hour period. An inert gas atmosphere was maintained in the reaction flask by blanketing dry nitrogen gas throughout the reaction period. The reaction completion was determined by monitoring the hydroxyl value of the resin at definite intervals (Figure 3).

 Coating Sample Preparation
Coating Sample Preparation
Coating samples were prepared by using resin, hydroxylcyclohexyl phenyl ketone (HPCK, 25% by wt.) and reactive diluents in different ratios. The sample designations are given in Table 2.
 Sample Characterization
Sample Characterization
Coating sample viscosities were determined by a Brookfield Viscometer, and refractive index was determined by a Mettler Refractometer (Toledo RE 40D). Data is shown in Table 3.
Curing
The UV-cured PU-acrylate films were prepared by applying the various coating formulations on mild steel panels using a bar applicator. The dry film thickness was maintained at ~12 microns with the help of a film applicator for the evaluation of various mechanical and chemical resistance properties. The panels were then exposed to a UV lamp chamber (GT Ultra Cure, 120 w/cm) maintained as follows.

Figure 4 shows the IR spectrum of the PU-acrylate resin. A weak band 1180 cm-1 is attributed to C-N stretching. A band at 1651 cm-1 confirms the formation of the urethane group. A band at 1534 cm-1 is attributed to N-H deformation. A strong band at 1735 is from the acrylate ester carbonyl group present in PU-acrylate resin. A strong band at 2963 cm-1 is believed to be due to C-H stretching and a strong band at 3345 cm-1 is thought to be arising from –OH stretching, due to residual hydroxyl group present in the resin. Moreover, the absence of any band at 2270 cm-1 in the spectrum confirms that unreacted NCO groups are not present.
 Molecular Weight
Molecular Weight
Figure 5 shows the molecular weight distribution of the PU-acrylate resin. It is clear from the figure that the synthesized resin is a low-molecular-weight resin.
Characterization of Coating Films
Various coating formulation panels were cured in the UV chamber and tested for different mechanical and chemical resistance properties, as per the following test methods: gloss at 60° (ASTM D 523-99); scratch hardness (ASTM D 5178); pencil hardness (ASTM D 3363-00); chemical resistance (EN: 438-2:1991) and stain resistance (ISO-4211).
 Scratch Hardness
Scratch Hardness
Scratch hardness is one of the important mechanical properties required for a coating to fulfill its protective role. The damage caused by a scratch on a cured surface may lead to a change in the gloss properties of the coating or it may be severe enough to cause deformation and finally induce cracking in the coating. Scratch hardness was determined using an automatic scratch hardness tester having a hardened steel hemispherical point of 1 mm diameter as a scratching needle. Results are shown in Table 4.
It is clear from the data that films having a higher concentration of TMPTA, i.e., sample A, have high scratch hardness due to maximum extent of crosslinking of the monomer.
 Cross-Hatch Hardness
Cross-Hatch Hardness
Cross-hatch was measured by using a cross-cut adhesion tester. The tester consists of a die made up of 9 parallel blades, 1/16 inches apart and 1 mm in length. The die is pressed into the panel in two directions at right angles to each other. A strip of self adhesive was staked over the pattern, left in contact for 10 seconds and stripped rapidly by pulling the tape back on itself at an angle of ~120°. The test was rated good if 5% of the squares were removed. All the coating samples had good cross-hatch hardness properties (Table 5).
 Pencil Hardness
Pencil Hardness
In the test, a strip is drawn under a pencil until a hardness grade is reached that will scratch the surface. The coating film is then assigned a hardness value such as H, 2H, 3H, etc., signifying the hardness grade. Coating films of TMPTA showed maximum hardness due to higher functionality that led to a maximum degree of crosslinking (Table 6).
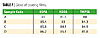 Gloss
Gloss
Gloss was measured using a Triglossometer (Sheen). After watching the films from a 60° angle, it was observed that all the coating films had good gloss. Coating films of EOPA showed maximum gloss (Table 7).
 Flexibility
Flexibility
Flexibility testing was carried out on a ¼-inch Mandrel bend tester. Films of all coatings compositions were flexible enough to pass through the Mandrel (Table 8).
 Chemical Resistance
Chemical Resistance
The coatings samples were tested for their chemical resistance properties. The results of the tests are shown in Table 9.
Distilled Water Resistance
Coating films exhibit good resistance when exposed to distilled water. Samples, which have a high percentage of TMPTA, showed better resistance (Table 9).
 Acid Resistance
Acid Resistance
Films of EOPA and HDDA were affected by acid solution due to poor crosslinking of monomers. Samples of TMPTA showed excellent resistance against acid solution (Table 9).
 Alkali Resistance
Alkali Resistance
To examine alkali resistance, coating films were exposed to 0.1 N NaOH solution. Films of TMPTA showed better resistance due to high functionality of the monomer that leads to maximum crosslinking (Table 9).
 Stain Resistance
Stain Resistance
In accordance with ISO-4211, panels were coated and allowed to cure in the UV chamber. The coated panels were kept at room temperature for 24 hrs. Droplets of tea, coffee and hot water were pipetted onto the films and covered with a glass cup for 24 hours to prevent evaporation. The films were carefully cleaned, and the film’s surface was analyzed (Figures 6 – 8).
For more information, e-mail abhishekhbti27@gmail.com or dahbti@rediffmail.com.
UV-curable coatings are a boon to the paint industry, as they not only reduce VOCs but also speed up production processes and provide protective, durable and attractive finishes. UV-curable polyurethane acrylates have been synthesized from polyester polyol (1,6-hexanediol, adipic acid and ethylene glycol), toluene diisocyanate and three types of reactive diluents i.e., ethoxylated phenol monoacrylate (EOPA), 1,6-hexanediol -diacrylate (HDDA) and trimethylolpropane triacrylate (TMPTA). In this paper several formulations of UV-curable polyurethane acrylate resins were used to study the effect of reactive diluents on performance properties. It was observed from the results that UV-cured polyurethane acrylate resin films on metal surfaces exhibit excellent gloss (60°), mechanical, and chemical- and stain-resistant properties.
Introduction
Light-induced polymerization of multifunctional monomers is one of the most efficient methods to rapidly synthesize highly crosslinked polymer networks. A liquid resin can be transformed within seconds into a solid polymer totally insoluble in organic solvents and very resistant to heat, chemical and mechanical treatments.1,2 This technique has been applied commercially in printing, thin film coatings and adhesives.3,4Photo-curable formulations usually consist of multifunctional monomers, reactive diluents, oligomers and photoinitiators, which generate reactive species on UV exposure.5,6 Typically it is a segment of polyurethane oligomer tipped with acrylic functionality such as 2-hydroxyethylacrylate (HEA) or 2-hydroxyethylmethacrylate (HEMA).
The reactive diluents are acrylic monomers that are added to modify properties and to reduce viscosity of the precursor liquid.
The acrylated urethanes ideally combine the abrasion resistance, toughness, tear strength and good low-temperature properties of PU with the superior optical properties and weatherability of polyacrylates.7-9 The reactive diluent phase is more compatible with the hard segments of polyurethane rather than the soft segments, due to similar polarity and hydrogen bonding formation between the urethane NH and acrylate carbonyls.10
This paper considers the preparation and properties of UV-curable polyurethane acrylates. The NCO-terminated polyurethane prepolymers were synthesized from toluene diisocynate (TDI) and polyester polyol followed by tipping with 2-hydoxyethylmethacrylate (HEMA). In addition, three types of reactive diluents having mono-, di- and tri- functionality were used in different concentrations. A series of coating compositions having PU acrylate and reactive diluents were prepared, and their mechanical, chemical, optical and stain-resistant properties studied.

Experimental
ReagentsTDI (an 80/20 mixture of 2,6 and 2,4 isomers) was obtained from BASF and Narmada Chematur. The purity was 98% and was used as such. Laboratory reagent (LR)-grade adipic acid, 1,6-hexanediol, ethylene glycol, 2-hydoxyethylmethacrylate (HEMA) and hydroquinone of M/s Thomas Baker, Mumbai, were used. Reactive diluents were procured from M/s Cognis, Thailand, and photoinitiators were procured from M/s Ciba chemicals.

Initially ethylene glycol, adipic acid and 1,6-hexanediol were placed in a three-neck, round-bottom flask in the molar ratio of 1.6 : 3.4 : 4.0 respectively. The esterification reaction was catalyzed by adding dibutyl tin oxide (0.1%) as a catalyst. The temperature was maintained at 140 – 150 °C. An inert atmosphere was maintained by blanketing the reaction mixture with nitrogen. Acid and hydroxyl values (Figures 1 and 2) of the resin were checked at fixed time intervals.
Urethane Acrylate Synthesis
The reaction flask was charged with 2.2 moles of TDI under dry nitrogen gas at 50 °C; a blend of 2.4 moles of HEMA with hydroquinone (50 ppm) was added continuously over a period of one half hour while maintaining a maximum temperature of 60 °C. After the addition was complete, the reaction mass was kept at 60 °C for half an hour to assure the complete conversion of the hydroxyl group of HEMA.

The temperature of the reaction flask containing urethane methacrylate precursor was maintained at 50 °C, and a mixture of hydroxyl functional polyester polyol and hydroquinone (50 ppm) was added over a half-hour period. An inert gas atmosphere was maintained in the reaction flask by blanketing dry nitrogen gas throughout the reaction period. The reaction completion was determined by monitoring the hydroxyl value of the resin at definite intervals (Figure 3).

Evaluation and Characterization
An FTIR spectrum of polyurethane acrylate resin was recorded using a NaCl cell on a Perkin-Elmer 1750 FT-IR spectrophotometer. Molecular weight of the resin was obtained by GPC (model HPLC 600, RI detector on a styragel column) using polystyrene as a standard and THF as an eluent. The hydroxyl value of the resin was determined by titration (Table 1).
Coating samples were prepared by using resin, hydroxylcyclohexyl phenyl ketone (HPCK, 25% by wt.) and reactive diluents in different ratios. The sample designations are given in Table 2.

Coating sample viscosities were determined by a Brookfield Viscometer, and refractive index was determined by a Mettler Refractometer (Toledo RE 40D). Data is shown in Table 3.
Curing
The UV-cured PU-acrylate films were prepared by applying the various coating formulations on mild steel panels using a bar applicator. The dry film thickness was maintained at ~12 microns with the help of a film applicator for the evaluation of various mechanical and chemical resistance properties. The panels were then exposed to a UV lamp chamber (GT Ultra Cure, 120 w/cm) maintained as follows.
- UV dryer type : GT Ultra Cure 250
- Curing width: 250 mm
- Bulb type: UV (medium pressure)-GT 250 pure
- Transparent: 120 watt/cm
- Silica Quartz
- Air cooling: 100 mm
- Operating voltage: 440 volts 3 phase A.C.
- Speed: 50 cycles
- Power consumption: 120 w/cm for UV

Results and Discussion
Infrared AnalysisFigure 4 shows the IR spectrum of the PU-acrylate resin. A weak band 1180 cm-1 is attributed to C-N stretching. A band at 1651 cm-1 confirms the formation of the urethane group. A band at 1534 cm-1 is attributed to N-H deformation. A strong band at 1735 is from the acrylate ester carbonyl group present in PU-acrylate resin. A strong band at 2963 cm-1 is believed to be due to C-H stretching and a strong band at 3345 cm-1 is thought to be arising from –OH stretching, due to residual hydroxyl group present in the resin. Moreover, the absence of any band at 2270 cm-1 in the spectrum confirms that unreacted NCO groups are not present.

Figure 5 shows the molecular weight distribution of the PU-acrylate resin. It is clear from the figure that the synthesized resin is a low-molecular-weight resin.
Characterization of Coating Films
Various coating formulation panels were cured in the UV chamber and tested for different mechanical and chemical resistance properties, as per the following test methods: gloss at 60° (ASTM D 523-99); scratch hardness (ASTM D 5178); pencil hardness (ASTM D 3363-00); chemical resistance (EN: 438-2:1991) and stain resistance (ISO-4211).

Scratch hardness is one of the important mechanical properties required for a coating to fulfill its protective role. The damage caused by a scratch on a cured surface may lead to a change in the gloss properties of the coating or it may be severe enough to cause deformation and finally induce cracking in the coating. Scratch hardness was determined using an automatic scratch hardness tester having a hardened steel hemispherical point of 1 mm diameter as a scratching needle. Results are shown in Table 4.
It is clear from the data that films having a higher concentration of TMPTA, i.e., sample A, have high scratch hardness due to maximum extent of crosslinking of the monomer.

Cross-hatch was measured by using a cross-cut adhesion tester. The tester consists of a die made up of 9 parallel blades, 1/16 inches apart and 1 mm in length. The die is pressed into the panel in two directions at right angles to each other. A strip of self adhesive was staked over the pattern, left in contact for 10 seconds and stripped rapidly by pulling the tape back on itself at an angle of ~120°. The test was rated good if 5% of the squares were removed. All the coating samples had good cross-hatch hardness properties (Table 5).

In the test, a strip is drawn under a pencil until a hardness grade is reached that will scratch the surface. The coating film is then assigned a hardness value such as H, 2H, 3H, etc., signifying the hardness grade. Coating films of TMPTA showed maximum hardness due to higher functionality that led to a maximum degree of crosslinking (Table 6).

Gloss was measured using a Triglossometer (Sheen). After watching the films from a 60° angle, it was observed that all the coating films had good gloss. Coating films of EOPA showed maximum gloss (Table 7).

Flexibility testing was carried out on a ¼-inch Mandrel bend tester. Films of all coatings compositions were flexible enough to pass through the Mandrel (Table 8).

The coatings samples were tested for their chemical resistance properties. The results of the tests are shown in Table 9.
Distilled Water Resistance
Coating films exhibit good resistance when exposed to distilled water. Samples, which have a high percentage of TMPTA, showed better resistance (Table 9).

Films of EOPA and HDDA were affected by acid solution due to poor crosslinking of monomers. Samples of TMPTA showed excellent resistance against acid solution (Table 9).

To examine alkali resistance, coating films were exposed to 0.1 N NaOH solution. Films of TMPTA showed better resistance due to high functionality of the monomer that leads to maximum crosslinking (Table 9).

In accordance with ISO-4211, panels were coated and allowed to cure in the UV chamber. The coated panels were kept at room temperature for 24 hrs. Droplets of tea, coffee and hot water were pipetted onto the films and covered with a glass cup for 24 hours to prevent evaporation. The films were carefully cleaned, and the film’s surface was analyzed (Figures 6 – 8).
Conclusion
UV radiation was effectively used to cure PU-acrylate resins in the presence of reactive diluents to obtain coatings for metal surfaces having excellent mechanical and chemical properties. UV-cured metal surfaces also exhibit excellent stain resistance properties. Coating properties were greatly affected when the reactive diluents were incorporated in the resin. At higher concentration of reactive diluents, gloss and scratch hardness increases. TMPTA showed better mechanical and chemical properties than EOPA and HDDA due to higher functionality, which leads to a maximum degree of crosslinking, providing excellent mechanical and chemical properties. Therefore these coatings can be used as protective coatings for metals and can also reduce VOC and hazardous air pollutants.For more information, e-mail abhishekhbti27@gmail.com or dahbti@rediffmail.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






