Decorative and Functional Metallic Effect Pigments





The coatings market continues to grow and change at a rapid pace. Not only are coatings formulators looking for metallic effects with brilliant finishes and mirror-like chrome appearance for both liquid and powder coatings, but there is increased demand for improved performance characteristics like corrosion resistance, barrier effects and thermal durability. The ever-growing importance of waterborne technology has also created exciting innovation opportunities for metallic pigment manufacturers.
This article highlights a number of decorative and functional pigments available in the Benda-Lutz®Metallic Effects portfolio by Sun Chemical. Metallic effect pigments consist of flakes or platelets of aluminum, copper, copper-zinc alloys, zinc and other metals (Figure 1). These flake pigments or pastes can be used to provide uniquely attractive aesthetics (high metallic brilliance, strong color flop etc.) in a wide variety of coatings applications. Furthermore, many metallic pigments can be used to impart improved functional properties. Corrosion resistance, barrier resistance and thermal durability are just a few examples of functional characteristics that can be improved with metallic effects.
Aluminum
Metallic silver is traditionally a top-selling color globally for automotive, consumer electronics and other decorative coatings. Silvers and other tinted metallic finishes are most often formulated using aluminum flake pigments. Aluminum pigments have outstanding outdoor durability and weathering characteristics. This makes them ideal for automotive topcoats, which can drive color trends in other markets. The optical properties of aluminum flakes are mainly derived from their geometry and surface roughness. Flake structures that most efficiently reflect incident light with minimal scattering give improved metallic brilliance and flop (light to dark color travel with different viewing angle).
In the majority of processes used to produce aluminum flake pigments, lubricants are required to prevent cold welding.1 These lubricants can impart leafing or nonleafing properties when used in coatings. Due to their high surface tension, leafing grades migrate toward the surface of the paint film, as shown in Figure 2a. This enrichment at the boundary yields a strongly reflective metal surface giving high metallic brilliance and gloss. For liquid coatings, leafing aluminums are ideal for anticorrosive applications (roof, tank, marine and heat-resistant coatings). These grades both reflect radiation (visible, UV and IR) and provide a barrier effect from moisture and other reactive media into the paint film.
Nonleafing aluminum flakes are fully wetted by the binder and uniformly distribute throughout the paint film, as shown in Figure 2b. For this reason, these pigments are highly suitable for tinted or polychromatic paints such as automotive topcoats. Nonleafing pigments are also preferred for applications requiring excellent adhesion or scratch resistance. They provide a significant improvement in surface properties compared to leafing grades. Some common nonleafing flake geometries are shown in Figure 3. These products are arranged from left to right in order of reflecting efficiency. The flakes shown range from irregular platelets (cornflakes) to silver dollar flakes to vacuum metallized pigments (or VMPs).
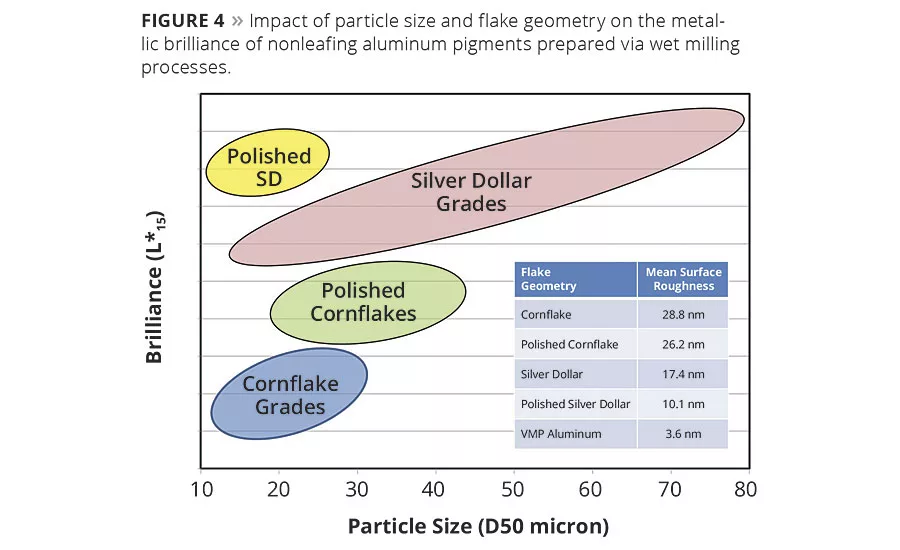
All flake geometries shown in Figure 3, except VMP grades, are produced via the Hall wet-grinding process. Figure 4 displays the impact of particle size and shape on the brilliance of aluminum pigments prepared by wet milling. The irregular flake geometries of cornflake grades (Figure 3a) are characterized by lower surface-to-edge ratio and much higher surface roughness (Figure 4). Light scattering occurs at the edges, resulting in lower metallic brilliance and less travel relative to more sophisticated flake geometries. These pigments find more use in industrial coatings for their exceptional opacity.
Premium silver dollar or lenticular aluminum flake geometries are displayed in Figure 3c. In addition to their uniform flake structure and reduced surface roughness, these pigments are precisely controlled to have a very narrow particle size distribution. As shown in -Figure 4, metallic effect pigments are the most brilliant the coarser the grade, the smoother the flake surface and the narrower the particle size distribution. In general, brilliance and sparkle increase with particle size, and opacity decreases. The manufacturing of fine metallic pigments with high brilliance is a target of continuous research and development. These pigments require special processing to produce highly uniform, monodisperse flakes that give high specular reflection with minimal flake surface area.
Unlike other geometries shown in Figure 3, VMP pigments are produced by physical vapor deposition. These flakes are uniformly deposited and stripped from a moving web substrate, yielding perfectly smooth aluminum flakes with carefully controlled thickness. In terms of optical quality, vacuum metallized aluminum offers a level of metallic brilliance and reflecting power unattainable by traditional milling processes. Therefore, they are most often used to create mirror and chrome-like effects in high-end finishes (wheel coatings, automotive trim and consumer electronics). A full review of this line of pigments is given in a recent article in PCI.2
Many markets, most notably China, have developed new regulations designed to improve air quality and reduce VOC emissions. This has required paint producers to transition from solventborne to waterborne or low-VOC formulations. It is well known that untreated aluminum will react with water, liberating hydrogen gas. The most common methodologies for stabilizing aluminum flakes for waterborne are chemical passivation, encapsulation or sometimes a combination of both. Encapsulation offers a higher level of gassing stability with less impact on the optical and application properties of the base aluminum flake. Functional properties such as gassing resistance, mechanical stability, acid and base resistance,3 adhesion and outdoor durability can be enhanced by encapsulating the flake surface.
Goldbronze
Goldbronzes are flake pigments made from copper or copper-zinc alloys (sometimes referred to as brass). These pigments are popular for decorative ink and coating applications due to their unique gold and metallic bronze effects. The final color of goldbronze pigments is determined by the ratio of copper to zinc used in the alloying process. In most cases, the copper content ranges from around 70% to 100%. Goldbronzes are generally provided in a variety of particle sizes in four standard colors, namely rich gold (~ 70:30 Cu to Zn ratio), rich pale gold (~ 85:15), pale gold (~ 90:10) and copper (100% copper). The color gamut of goldbronzes can be extended using controlled oxidation processes to produce fine layers of metal oxides on the surface of the goldbronze flake. These surface layers produce interference effects that give exciting colors such as orange and fire red.
The lower temperature stability of goldbronze is well established. Standard, untreated goldbronze powders are only stable to 80 °C, which excludes them from applications such as powder coatings. Inorganic encapsulation can be used to produce goldbronzes with significantly higher temperature stability (up to 200 °C). This encapsulation technology also gives the pigments improved chemical resistance for aggressive binder systems.
Zinc
While goldbronze pigments are typically used for decorative coatings, the majority of zinc pigments are used to provide functionality. Due to their position in the electrochemical series of elements, zinc pigments are most often used to provide corrosion protection. In general, the most common anticorrosion coating is zinc-rich primers prepared using zinc dust (comprised of at least 90% zinc in the dried film). Benda-Lutz ZINC is a line of flaked zinc pigments that provide the excellent cathodic protection of zinc in a platelet form, allowing for coatings with much lower film thickness and lower tendency to settle. In addition, this flake geometry provides excellent long-term barrier protection and gives the finished coating a smooth, unique metallic appearance.
Corrosion protection requires conductive contact between the substrate and the coated zinc particles. Zinc flakes provide much more extensive surface contacts, yielding higher electrical conductivity and therefore better corrosion resistance. Since zinc flakes react more rapidly than zinc dust; it is common to use zinc flake in combination with aluminum flake. This reduces the reactivity of the zinc flake and extends the period of cathodic protection.
In addition to the outstanding corrosion properties, the zinc line of pigments also includes a true black zinc flake. Figure 1 provides an image of the dry pigment while Figure 5 provides an image of the product used in a coatings application. Typical zinc flakes are grey or silver in appearance. This unique black zinc flake provides new styling opportunities for applications such as high-heat-resistance coatings (automotive exhaust, brake rotors) and other ancillary automotive parts. Relative to zinc dust, zinc flakes provide better handling, increased formulation flexibility, improved application properties and improved sustainability (lower material consumption per covered area).
References
1 Wissling, P. Metallic Effect Pigments, European Coatings Literature, Vincentz Network, 2006.
2 Mitchell, J.; Hollman, A.M.; Venturini, M.T. Finding Chrome Formulations Puzzling? PCI, October 2013, p. 54.
3 Hollman, A.M. Improved Chemical Resistance, PPCJ, Vol. 200 No 4546, March 2010, p. 12.
For more information, e-mail aaron.hollman@sunchemical.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






