There Can Be a Large Difference Between the Wet and Dry Tg of Polymers

The “Did You Know….?” series is a bi-monthly note from Emulsion Polymers Consulting and Education (EPCEd) that is intended to present simple questions about topics that are important to those working in the emulsion polymers area. Short and concise answers to those questions are presented to educate readers and to elicit comments and further discussion. Some readers will already know the answers and be familiar with the topic while others, especially those newer to the field, will benefit from the answers and discussion. Experienced practitioners may also find new insights in the discussion. Paint & Coatings Industry magazine has partnered with EPCEd to share the “Did You Know” notes with our readers throughout the year.
Did you know that there can be a large difference between the Tg of polymers when the sample is in the dry or the wet (with water) state? Those working with emulsion polymers encounter this difference as a natural part of their everyday experience, as water is a very strong plasticizer. While very non-polar polymers (like polystyrene) will be little affected by water, the Tg of more polar polymers (due to carbonyl, hydroxyl, and/or amine groups in the monomers) can be significantly decreased in the presence of water. The use of functional monomers such as vinyl acids increases the effect of the water. An example of the effect of water on the polymer Tg is shown in Figure 1.
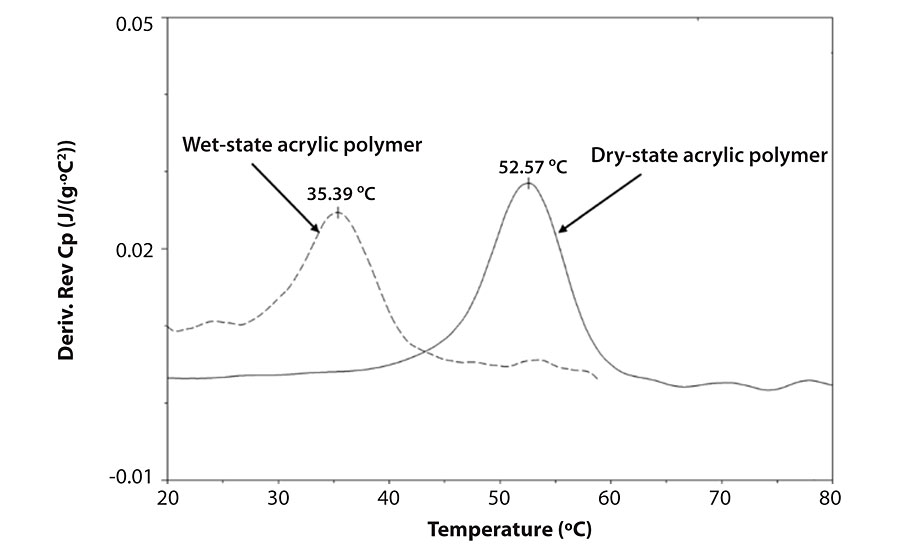
In the data set in the Figure we see a 17 °C decrease in the Tg due to the presence of water dissolved in the acrylic copolymer. Such effects are important in latex film formation, water absorption into dry films, and also in the emulsion polymerization reactions used to create the latex.
This subject is treated in detail in several of our STEPn workshops, especially STEP 2 “Characterization of Latex Particles”. It is also presented in the scientific literature – Langmuir, 26, 9408-9415 (2010).
We welcome comments and further discussion of this topic. Please contact us via our website www.epced.com.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





