Novel Branched Surfactants
Improve Tinting Systems for Architectural Coatings

Color strength and color acceptance are two main attributes, when an architectural coating is formulated; compatibility between the tint or colorant and the paint is critical to achieve great performance when the coating is applied by roller, brush or spray, and for its final properties as a film. To develop, prepare and match a color requires technology and many hard hours of work. To achieve this, many ingredients are interacting, for instance, slurries, lattices, coalescing agents, surfactants, dispersants, etc. In this article, our attention is focused on surfactants, more specifically nonionic branched surfactants.
Branched surfactants possess unique properties. Polyethylene and propylene oxide groups are easy to modify, creating different HLBs¹ and offering an enormous portfolio. Also, being alkylphenol ethoxylate (APE)-free they are a replacement alternative and comply with environmental demands and biodegradability. In the past, different tests and technical studies have been done related to color properties.²⁻⁴ Mahli, et.al⁵ shows that increasing the ethoxylation in nonionic surfactants improves color development for carbon black, yellow and red colorants or tints at a certain concentration range.
In addition to the ethoxylation and location of these hydrophilic groups, another unique property for branched surfactants, for a single straight-chain, is to increase the critical packing parameter (CPP), permitting more adsorption at solid surfaces⁶ and therefore allowing better stability. This stability can be earned on hydrophobic surfaces using polyethylene oxide chains of at least 20 units long. Polyethylene oxide chains are hydrated in water, forming a steric barrier.⁷ However, branched surfactants do not always develop more color strength. Nagose, et.al.⁸ shows that a primary alcohol (ethylene oxide-propylene oxide blend) improves color strength for pigment yellow PY 110 compared to other chemistries, including branched surfactants.
Nevertheless, branched surfactant technology is more likely to have unimers rather than form micelles. This unique feature plays an important role. Having unimers at the interface rather than a micelle structure will help to increase the adsorption, improving color properties. Also, these unimers are responsible to reduce the surface tension in dynamic conditions. Dynamic surface tension reduction is an important characteristic of wetting time, emulsification and others.⁹
Adsorption and Micellization
When surfactants are in solution and the concentration is gradually increased, unimers migrate to the interface. When the surface is completely covered, they aggregate at a certain concentration, forming micelles. This concentration is called critical micelle concentration (CMC). Micelles have a lifetime – the lifetime of one micelle responds to a dynamic process that can be measured in microseconds, presenting a relaxation time.¹⁰ This relaxation time is important to bring dynamic surface tension reduction in different processes.
Unimers could prefer to be aggregated, minimizing energy and forming part of a micelle or to be in solution as unimer. The preference is in part related to its chemical structure, knowing if the surfactant is linear or branched can bring good insights about their preference. Of course, more factors play an important role that need to be evaluated. However, collecting experimental information and calculating the CMC and PC₂₀ or C₂₀, which is the concentration required to decrease the equilibrium surface tension 20 dynes/cm, can bring a central understanding where the unimer selects to be free in solution or forming part of a micelle.
Experiment
White acrylic interior paints with 50% PVC and 50% solids by weight were prepared by a batch process using high-disperser equipment and allowed to rest overnight. The formulation included a 100%-acrylic, APE-free latex and associative rheology modifiers comparable to HEURs. We evaluated five 100% branched surfactants at different HLBs ranging from 8 to 18. The hydrophobic branched group, C12/C15, was kept constant in all our tests. APE-E9, nonylphenol ethoxylate 9 moles, was our standard to compare and used as a reference. The amount tested was 0.25% by weight and was held constant in all trials.
Our evaluation was done in two different categories: color properties and general properties, including rheology. Regarding color properties, six APE-free tints were evaluated – each tint added at 2% by weight in each paint and shaken using a Red-Devil shaker for 10 min. The paints were cast at 3 wet mills and allowed to dry between 1 min, 30 sec to 2 min before rubbing-up the non-sealed part on a Leneta type 1A. The color strength and color acceptance were measured using a colorimeter X-Rite model Ci6X at D65 light source and an angle of 10 degrees.
Color Evaluation and Results
Color strength is an imperative property to be quantified to test surfactants in paints. They significantly influence the color strength; the target is to offer similar values compared to APE-E9, approximately 100% ± 2. Figure 1 shows a family of branched surfactants comparing color strength at different ethoxylation levels and different HLB values. Some of the branched surfactants are above 100%, meaning better color strength is achieved. It is important to emphasize the performance of low-ethoxylation products. Branched surfactants 330 and 350 increased color strength of 106% and 110% for PBk 7 and organic yellow 808-2040 respectively. Generally speaking, branched surfactants present a clear advantage for PBk 7/808-9907 tint – all of them presenting good values.
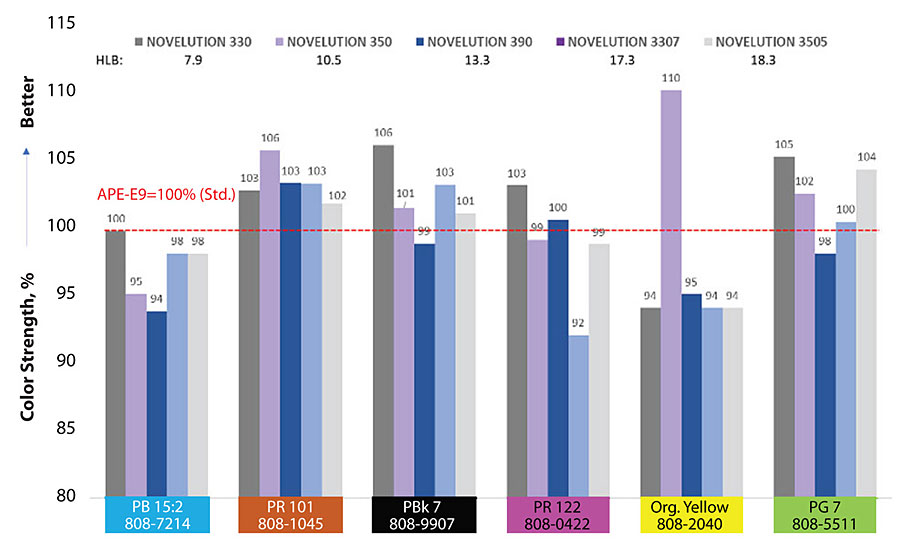
Figure 2 shows the results for color acceptance. In general terms, branched surfactants show a good performance. All of them accept very good Lamp Black PBk7/808-9907 tint. Branched surfactants 390 and 3505 perform very well in five of the six colorants. Possibly, due to their high ethoxylation chains of 3505 they may work as a co-dispersant, providing steric stabilization and improving compatibility.
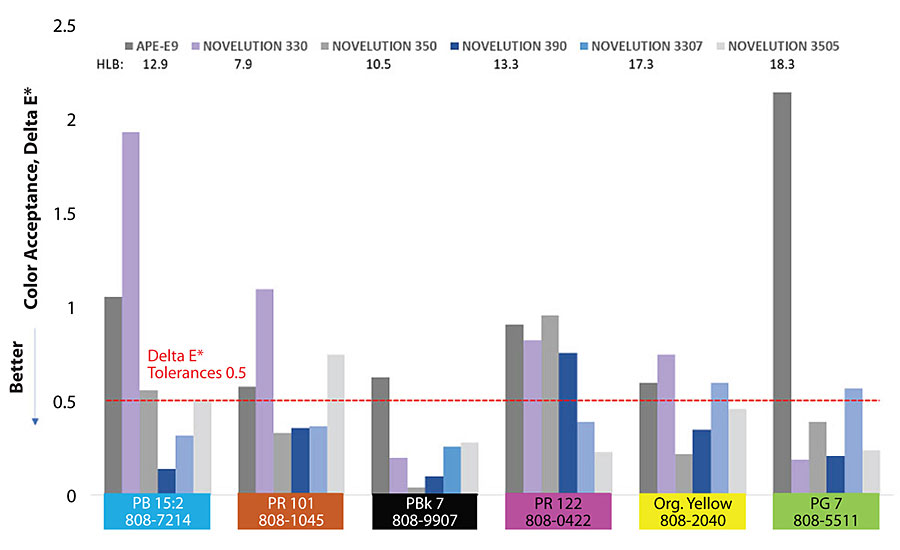
However, the real challenge is to have both attributes – color strength of 100±2% and a Delta E* equal or below 0.5. The surfactant system must comply with color strength and color acceptance together. One single color property will not provide the full criteria to make the right choice. Branched surfactant 3505 shows a good performance for phthalocyanine tints. For PB 15:2 and PG7 the values are below Delta E* 0.5, and color strength 98 and 104 respectively. Branched surfactant 350, due to its novel wetting characteristics, increases its color strength for inorganic PR 101 and PY organic yellow, keeping Delta E* values below 0.5
General Properties and Unimers
In addition to color tests we evaluated different properties. Table 1 shows the results of all white paints. Viscosity in Krebs-Stormer units (KUs) and ICI viscosity (Poise) are the rheological tests. The first one measures the consistency of the paint at two different periods – initial (I) and overnight (ON). A few variations between both means better stability and could predict short-term shelf life behavior. Most of the paints show good values, excluding branched surfactant 330, whose viscosity increased by 6 KUs. These values could be important or be acceptable depending on the system and quality control parameters, but it does not mean we should disregard this surfactant at all. Our stability tests for two weeks at 60 °C support good stability, showing some syneresis and soft sediments. The soft sediments can be easily re-incorporated when mixing the paint. On the other hand, ICI viscosity measures the film build and is related to a mass transfer process depositing paint. In contrast, the same branched surfactant 330 shows the highest ICI value since it’s the most hydrophobic of all of them – an increment of 30% in viscosity is observed. Also important are optical properties such as contrast ratio, whiteness index, yellowness index and gloss at 60 degrees. No significant changes were observed – all paints showed similar results. This is an important fact in the eventual case for a potential replacement of APE-E9 in paint formulas – branched surfactants do not significantly change main properties.
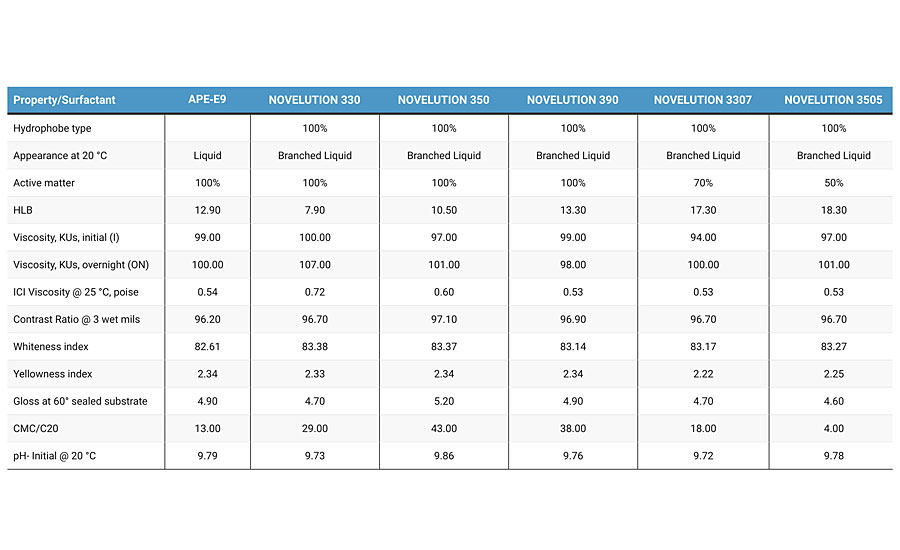
Comparing APE-E9 and branched surfactant 390, both possess similar ethylene oxide content and similar HLB, the ratio of CMC/C20 is 13 and 38 respectively, and branched surfactant 390 is almost three times greater than APE-E9. This means that unimers will bring effective adsorption onto pigments, increasing the stability and other features in the paint. Free unimers present competitive adsorption for absorption into the pigment surface and/or latex and/or associative rheology modifiers, etc. The rest of the ratios for branched surfactants show values greater than APE-E9. Probably a fair comparison is to calculate the same ratio for similar ethylene oxide groups and compare APEs against branched surfactants. However, our previous experience has shown similar results to APE-E9 where branched surfactants show higher values of CMC/C20 compared to APEs.
Conclusions
Branched surfactants show promising results as an effective replacement for APEs in white architectural paints and tinting systems. In this study, we worked at a fixed amount of 0.25% by weight, but more tests and evaluations at different percentages are expected to bring different synergists. Besides color properties, branched surfactants maintain good optical and rheological properties. Due to its novel wetting characteristics, branched surfactant 350 increases color strength and low Delta E* for inorganic PR 101 and PY organic yellow. Branched surfactant 390 can be an effective replacement for APE-E9, improving color acceptance and keeping a similar color strength with the benefit of being APE-free and biodegradable. Branched surfactant 3505 shows a good performance for phthalocyanine tints improving compatibility. We show the important role unimers play and the preference for increasing adsorption at a liquid-solid interface or forming part of a micelle. Low ethoxylate branched surfactants present good wetting properties, and more color strength has been developed when using them. On the other hand, high ethoxylation branched surfactants present good color acceptance, probably by working more as co-dispersants and increasing compatibility.
For more information, contact Jeanine.Snyder@us.sasol.com.
References
¹ Griffin, W.C. Classification of Surface-Active Agents by “HLB”, Journal of the Society of Cosmetic Chemists, 1949, 5, 311-326.
² Reuter, E.; Silber, S.; Psiorz, C. The Use of New Block Copolymeric Dispersing Agents for Waterborne Paints - Theoretical and Practical Aspects. Prog. Org. Coat., 37, 161-167 (1999).
³ Meier, I.K.; Peck, K.M.; Reader, C.J.; Louis C. Color My World! White Paper: New Tools for Improving Colorant Acceptance in Waterborne Architectural Coatings.
⁴ Sis, H.; Birince, M. Wetting and Rheological Characteristics of Hydrophobic Organic Pigments in Water in the Presence of Non-Ionic Surfactants. Colloids Surf. A Physicochem. Eng. 455 (2014) 58-66.
⁵ Mahli, D.M.; Wegner, J.M.; Glass, J.E. J.Coat. Technol. Res. 2(8), 635 (2005).
⁶ Holmberg, K.; Jonsson, B.; Kronberg, B.; Lindman, B. Surfactants and Polymers in Aqueous Solutions, 2nd edition; p. 359.
⁷ Mittal, K.L.; Shah, D.O. Adsorption and Aggregation of Surfactants in Solution. Vol. 109 (2003) pp 265.
⁸ Nagose, S.; Rose E.; Joshi, E. Study on Wetting and Dispersion of the Pigment Yellow 110. Prog. Org. Coat 133, 55-60 (2019).
⁹ Schwartz, J. The Importance of Low Dynamic Surface Tension in Waterborne Coatings, Journal of Coatings Technology, 1992, 64, 65-74.
¹⁰ Aniansson, E.A.G.; Wall, S.N. J. Phys Chem 78:1024, 1974.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




