New Curing Agents for Dimensionally Stable, Aqueous Epoxy Resin Coatings


Shrinkage Causes
Two major types of shrinkage are distinguished for polymer materials: shrinkage in the course of the process and the so-called after-shrinkage. The combination of both types is considered as total shrinkage.One of the best-known causes for shrinkage of synthetics is the temperature-conditioned contraction or density increase of polymer materials after hot processing. It is considerably lower for amorphous synthetic materials compared with crystalline, respectively semi-crystalline synthetics. The temperature-conditioned density increase is, in crystalline synthetic materials due to the more regular molecule orientation, substantially larger than the density modification in the solidification of amorphous synthetics to vitreous solids. This type of shrinkage, known from the processing of synthetic materials, is virtually irrelevant for coating materials.
After-shrinkage may, on the whole, be a consequence of chemical reactions, substance release, after-crystallization or relaxation. Two of the aforementioned causes for after-shrinkage, the chemical reaction and substance release, are of particular significance1,2 for coatings such as unsaturated polyester, epoxy or acrylate resin systems.
The chemical reaction may be relevant in some ways both for solvent-free and solventborne coating systems. As is known, a distinct volume contraction can be observed in the polymerization of low-molecular-weight monomers to high-molecular-weight polymers. For instance, ethylene polymerizes to polyethylene at a 66% volume decrease and styrene to polystyrene at a 14% volume decrease. The cause for the contraction is the building up of chemical bonds between the molecules formerly individually present. Figuratively speaking, the molecules "move closer"3 during polymerization. The chemical reaction in coatings may be a significant reason for the shrinkage at a correspondingly high percentage of polymerization or wetting groups per volume unit.
The shrinkage by substance release, in particular by solvent evaporation, is of influential importance for all solventborne systems. F. Ruf4 explains the processes initiated. Because of the film formation process, a reduction of the coating thickness occurs that entails a volume shrinkage (Figure 1), in particular by evaporating the solvent.
Shrinkage is a time-dependent variable, as the filming process does not proceed simultaneously in the entire layer. This means that the process of volume decrease does not take place in all three directions in space uniformly. In the direction of the film thickness there is an unimpeded shrinkage. Besides the volume contraction, a tension build-up may follow, which is produced by a time- and direction-dependent density change of the resin during the drying and curing process (Figure 2).
Significance of Shrinkage for the Application
As explained, the coating volume is reduced by chemical reaction, evaporation of solvents such as water, or regular molecule orientation. Unsaturated polyester, epoxy and polyurethane compounds show a considerable shrinkage potential (0.4 - 4.0 percent) in the course of the curing process. Particularly affected in these applications are all solvent- and water-based systems. In accordance with general observations, mainly traditional aqueous epoxy systems are susceptible to shrinkage. This particularly applies to thick-film applications at a layer thickness of more than 500 mm (Figure 3). The changes in internal stress may achieve an order of magnitude that produces crack formation or even peeling of the coating from the substrate.Temperatures of above 25 °C may severely affect the dimensional stability of coatings. The faster curing process of the coating initiated by an increase in temperature frequently entails a more stress-oriented formulation component, which may again result in crack formation. Only a skillful selection of raw materials allows one to compensate for this impact.
Fillers and pigments may produce both positive and negative results. Spherical products have the advantageous property of reducing shrinkage. Fibrous fillers/additives impede the internal alignment of the molecules, thus decreasing shrinkage accordingly. Lamellar fillers, however, enhance in general the shrinkage behavior.

Dimensionally Stable, Shape-Retentive Aqueous Epoxy Coatings
Modern coatings are aqueous-based in order to facilitate handling, reduce health hazards, decrease danger of fire and explosion, and diminish VOCs. Moreover, Sustainability and Responsible Care are of fundamental significance for the selection of raw materials for coating materials.5 This is reflected among other facts by the enhanced employment of renewable raw materials or aqueous systems.The development of dimensionally stable coatings is therefore an important objective for modern, water-dilutable epoxy resin coatings.
In contrast to solventborne systems, the specific feature of water-dilutable products is the evaporation behavior of water. Compared with, to a great extent, variably adjustable evaporation rates in solventborne systems there is only one single flash-off time. Correspondingly, the scope for developing a formulation for a dimensionally stable, shape-retentive epoxy resin coating is on the whole limited to the fillers and binding agents used.
In a variety of applications of aqueous epoxy resin coatings, the dimensional stability and consistency can be significantly increased by adding special pigments and fillers. An increase in the percentage of fillers induces a decreasing portion of resin used and thus also the moiety responsible for the shrinkage in the formulation. Usual dosages of fillers can be as large as 70 percent. A well-balanced adjustment of the binder with the fillers is however a prerequisite to achieve a stress-free coating with low-grade shrinkage. By controlled drying and curing conditions, coating shrinkage can be further minimized; these are nevertheless, particularly in construction, often not controllable.
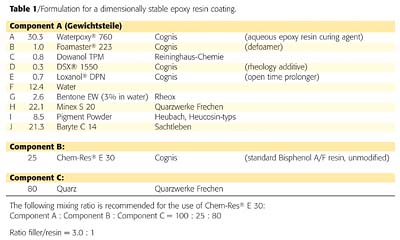
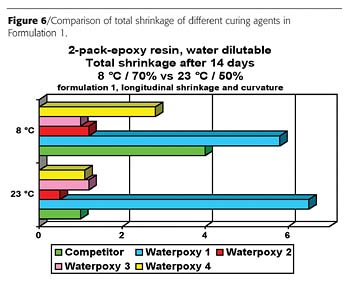
The result is corroborated in two additional formulations (Figures 7 and 8). Also the course of the curing process, based on the shore-D hardness, demonstrates that the shrinkage-free desiccation rapidly produces ready-to-use films.
Measurements have been made on rectangular and circular specimens. The dimensions were as follows: 1 x 1 x 10 cm; 0.5 x 10 x 20 cm; and 0.5 x 10 cm diameter. The overall change of the specimen, length, width and height alterations, was subject to the evaluation.
Summary
Test results revealed that modern water-dilutable epoxy systems allow thick-film and dimensionally stable, low-shrink coatings. It has been shown that the formulation largely influences the results. However, the formulator is easily able to achieve the expected results for coatings by means of a new modified curing agent.It has to be emphasized that practice-oriented values are attainable even at unfavourable temperatures of 8 °C and increased atmospheric moisture for aqueous systems. This way, the demands by the user for practical, thick-film protective coatings are fulfilled. The same applies to legal and ecological demands for the solvent and VOC content of the formulation.
References
1 Hellrich/Harsch/Haenele, Werkstoffführer für Kunststoffe, C. Hanser Verlag München Wien, 8. Auflage, S. 447-452, 2001.
2 Svoboda, M.; Kuchynka, D.; Knapek,B.; Farbe + Lack, 77, 11, (1971).
3 Elias, H.-G.; Makromoleküle Bd 1, Hüthig & Wepf Verlag Basel, S 212-214 (1990).
4 Ruf, J.; Organischer Metallschutz, Vincentz Verlag Hannover, S 460-471, 1993.
5 Henkel KGaA, Hrsg., Nachhaltigkeitsbericht 2002, Düsseldorf (2003); Cognis Deutschland GmbH&Co.KG, Hrsg., Der Cognis Nachhaltigkeitsbericht 2002, Düsseldorf (2003).
6 Roloff, T.; Nagorny, U.; Epoxidharzgebundene Systeme - Wirkungsweisen, Eigenschaften, Anwendungsgebiete, 7. Seminar, Beschichtungen und Bauchemie, Kassel, Oktober 2003.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!







