Stabilization of Titanium Dioxide in Non-Aqueous and Aqueous Coatings
Titanium Dioxide for Coatings
Titanium dioxide, as used for coating applications, is predominantly the rutile-type, high-purity titanium dioxide, manufactured by the chloride or sulphate process. The primary particle size of TiO2 for "hiding"-type applications is usually 0.25-0.3 µm. To reduce the photoactivity of the pigment, the rutile TiO2 crystal is modified with other metal oxides. In addition, very thin films of inorganic oxides (alumina, silica, zirconia) are deposited onto the pigment surface to influence the adsorption of additives.
Organic surface treatments (trimethylolpropane, neopentyl glycol, various surfactants, silicones) are often utilized to improve the dispersibility of TiO2 in organic or aqueous media. These organic treatments alone, in most cases, do not provide sufficient stabilization for proper deflocculation of the pigment and, therefore, paint producers must use additional additives in order to achieve the maximum level of pigment stabilization.
Additives for Wetting, Dispersion and Stabilization of TiO2
There are several types of additives used to disperse titanium dioxide. Since titanium dioxide is relatively easy to wet and disperse, the main job of the additive is to improve the stabilization of the dispersed pigment against flocculation. Anionic, cationic, electroneutral or amphoteric materials can be used for solventborne and solvent-free systems. In aqueous applications, anionic, cationic and electroneutral products can be used, sometimes in combination with non-ionic-type surfactants. There is obviously a relationship between the best additive type and the pigment's surface treatment, which can also be influenced by the resin and solvent selection. The target of this study is to provide more precise additive recommendations for specific titanium dioxide pigment types in each system.
Additives to Stabilize TiO2 Dosage Level
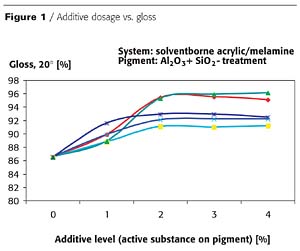
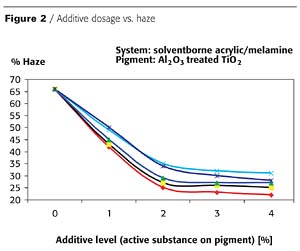
Increasing the levels of various additives types (differing in ionic character, molecular weight and chemistry) provides increasing degrees of pigment stabilization, usually characterized by gloss and haze measurements. Figures 1 and 2 characterize these properties of baking-type solventborne coatings.This data, and very similar data from a large number of other experiments, indicates that the optimal additive level is typically around 2% active substance, calculated on the TiO2 pigment. The proper amount of additive depends on the following factors:
- 1. The coating's specific application requirements - i.e., less for architectural or coil coating applications and more for automotive topcoats.
2. The type and chemistry of the binder system - i.e., aqueous systems typically require the highest levels.
3. The additive's chemistry - i.e., lower- to medium-molecular-weight electroneutral additives are usually more effective than their higher-molecular-weight anionic or cationic counterparts.
Selection of the Optimal Additive Chemistry
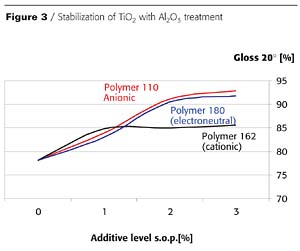
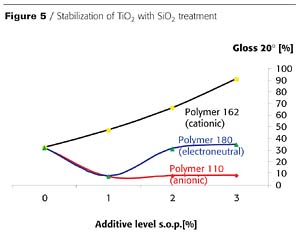
There is a relationship between the modification (inorganic surface treatment) of the titanium dioxide pigment and the chemistry of the most suitable additive. For pigments with basic surface treatment, usually anionic or electroneutral additives provide the best stabilization (Figure 3). For titanium dioxide grades with neutral surface treatment, electroneutral additives are recommended, and for silica-treated grades, cationic additives are recommended. (Figure 4 and Figure 5).Titanium dioxides with high levels of dense silica treatment typically show the greatest differentiation in gloss and haze as a function of the type of additive used for stabilization.
Additional studies have shown that results in aqueous and solvent-free systems follow the same rules as those described for solventborne coatings, but the results show even greater differences.
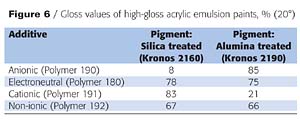
Figure 6 shows the gloss differences of an aqueous high-gloss acrylic emulsion paint, as a function of additive chemistry. Non-ionic polymers - although having good pigment wetting properties - do not provide basic or acidic treated titanium dioxide with a high degree of pigment stabilization. The titanium dioxide dispersions for this study were prepared as aqueous slurries, always with the optimal additive level for each product.
Additive Selection and TiO2 Isoelectric Point
Some titanium dioxide suppliers provide detailed information about the inorganic surface treatment.1,2 However, many commercial suppliers only indicate the nature (alumina, silica, zirconia, etc.) of the treatment, without providing details on the exact amount of the inorganic oxide modification. This lack of information can cause difficulties in the additive pre-selection process.In such cases, it is helpful to determine the isoelectric point of the TiO2 pigment (the pH value where the zeta-potential of the pigment is zero) or obtain the data from the supplier. This information can be used to clearly identify the most suitable additive chemistries for each pigment.
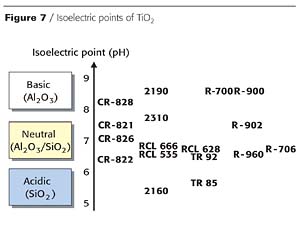
Figure 7 shows the isoelectric points of some common, commercially available titanium dioxides.
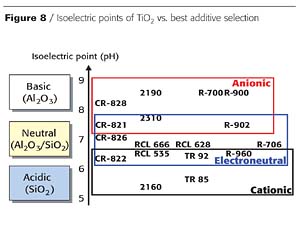
Figure 8 identifies the best additive options, as a function of additive chemistry and isoelectric point. In this portion of the study, 13 different polymeric wetting and dispersing additives (four anionic, three electroneutral and six cationic) were evaluated.
There is some overlap between the areas of anionic/electroneutral and electroneutral/cationic additive chemistries, but in most cases the isoelectric point clearly indicates the most suitable additive chemistry for each pigment.
Side Effects of Using Wetting and Dispersing Additives
Negative Side EffectsNegative side effects are rare, since the optimal dosage level of wetting and dispersing additives, as calculated on TiO2, is relatively low (2%). The most common problem is the effect of the additive's chemistry on the storage stability of some sensitive binder systems.
Cationic additives can shorten the storage stability of epoxy-functional systems due to their ability to polymerize the epoxy groups. Anionic additives can shorten storage stability due to their acid catalytic activity in some highly reactive urea-formaldehyde or melamine-formaldehyde resin-based formulations.
Another negative side effect is the reduction of the coating's water or humidity resistance, especially with very polar additive structures at higher dose levels. Non-ionic wetting agents, or permanently water-soluble anionic, cationic or electroneutral species can cause these effects.
Colorant acceptance of white base paints can be gradually improved when using certain widely compatible wetting and dispersing additive structures, especially in aqueous systems. Very positive effects have been found with cationic/non-ionic type acrylic wetting and dispersing additives in various types of aqueous latex architectural paints.3,4
Recently, some very positive side effects have been observed in high-gloss automotive topcoat formulations, where phosphate functional anionic and electroneutral wetting and dispersing additives significantly improve the chalk resistance of the coating film (Figure 9). These durability tests were carried out in Okinawa Island, Japan, which has similar environmental weather conditions to the established standard Florida test sites.
According to these results, the control formulation lost almost all of its gloss after two years, while phosphate-based wetting and dispersing additives provided much improved gloss retention. Slightly higher levels of additive do not improve initial gloss values, but may increase gloss retention by more than 10 % after two years.
The very great differences between the chalk resistance of the control and additive-containing formulations can be explained not only by better deflocculation of the pigment, resulting in more complete UV absorption by the pigment itself, but also by the fact that some organic phosphates can act as free radical scavengers. This can significantly reduce the degradation of the organic binder by those free radicals (mainly OH_ and HO2_ radicals) formed in UV-induced photochemical reactions at the surface of the TiO2 pigment.5
The greatest improvements were made in polyester-melamine-type binders. In more durable acrylic-melamine resin-based coating formulations, improvements are less pronounced, but still remarkable.

Summary
The inorganic treatment of commercially available titanium dioxide pigments determines to a large extent which type of wetting and dispersing additive will work best for that individual pigment type. For basic surface treatments, anionic and electroneutral additives provide the best deflocculation. For neutral surface treatments, electroneutral additives are best, while cationic additives work best with acidic SiO2-treated pigments. The isoelectric point of each pigment can be used to select the best additive. Organic phosphate-based polymeric wetting and dispersing additives provide not only a high level of pigment stabilization, but may also significantly improve the chalking resistance and durability of high-gloss topcoats.Acknowledgements
The author gratefully acknowledges some technical data and valuable comments from Ms. Anne Linzmaier, Mr. Bernd Dawid and Mr Akihiro Wakahara from BYK-Chemie, additional acknowledgments to Jotun Paints, Du Pont de Nemours, Inc., Kronos Titan, Inc., Sachtleben Chemie GmbH, Ishihara Sangyo Co., and Kansai Paint.
References
1 Du Pont de Nemours, Inc.: Technical data sheets of Ti-Pure Titanium Dioxides for coatings.
2 Kronos Titan, Inc: Technical data sheets of Kronos Titanium Dioxides for coatings.
3 Proceedings of the ChinaCoat Conference 2003, Shanghai; Janos Hajas, Frank Kother: Improvement of Colorant Acceptance in Architectural Paints.
4 Proceedings Eurocoat Conference 2002 Barcelona: Jaume Figueras, Janos Hajas: How to Improve Colorant Acceptance.
5 Company brochures Du Pont de Nemours, Inc., Kronos Titan Inc., Kerr-McGee, Inc. about theory of chalking.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





