Effect of a Novel Antimicrobial Latex Polymer Additive
On Scrub Resistance and Washability
The global antimicrobial coatings industry was $1.6 billion in 2012, and increasing demand will drive it to $3.3 billion in 2018.1 Current antimicrobial coatings have significant shortfalls. First, none are truly broad-spectrum and able to kill bacteria, fungi, viruses and spores. This is important in the case of high-risk, high-touch surfaces that can have many different types of microorganisms living on them at different times. In some cases the antimicrobial activity against a single class of microorganisms is inadequate. Further, many of the currently used active ingredients in antimicrobial paints suffer from established microbial resistance. In addition, some active ingredients, including triclosan, quaternary ammonium compounds and silver, can drive antibiotic resistance. Last, the antimicrobial functions of current paints cannot be easily monitored, and once the paints are applied, the antimicrobial duration cannot be further extended if it is lost due to prolonged use, heavy soil, flooding, etc.
For these reasons, a novel latex polymer was developed in our lab by Drs. Yuyu Sun and Zhengbing Cao. This emulsion is intended to be used as a paint additive. The antimicrobial activity comes from N-halamine functional groups anchored to a polymer backbone. (An N-halamine is a compound containing one or more nitrogen-halogen covalent bonds that is formed by the chlorination of imide, amide or amine groups.) There are several advantages to this new additive. First, it has a broad spectrum and is effective against both gram-positive and gram-negative bacteria, viruses and fungi. Second, the antimicrobial function can be tested using potassium iodide. In addition, if the antimicrobial function is lost, it can be recharged using a dilute bleach solution.
Having demonstrated the antimicrobial effectiveness of the additive, the goal of this study was to determine how the additive affects the performance characteristics of the paint.
Experimental
Methods
• ASTM Method D2486-06 “Standard Test Methods for Scrub Resistance of Wall Paints” Test Method B. An n=6 was chosen for these tests so statistical analysis could be performed on the results.
• ASTM Method D4828-94 “Standard Test Methods for Practical Washability of Organic Coatings”.
• Concentrations of 2% and 4% N-halamine latex emulsion were used because these were the lowest concentrations where a total kill could be seen in previously performed antimicrobial testing. All concentrations are a percentage of weight as a dried film.
Materials
The N-halamine latex was synthesized using a method previously described by Cao and Sun. Briefly, the synthesis consisted of mixing 2,2,6,6-tetramethyl-4-piperidyl methacrylate (TMPM) and dichloroisocyanurate sodium (DCCNA) to make N-chloro-2,2,6,6-tetramethyl-4-piperidyl methacrylate (Cl-TMPM). Cl-TMPM was then polymerized (poly(Cl-TMPM)) in the presence of water and surfactants to create the emulsion.
Two paints were selected for testing. The first was the same used by Cao and Sun in the initial publication, Color Place latex semigloss house white paint. The second was HEALTH-KOTE® low-odor interior eggshell latex white.
For the practical washability testing, charcoal and permanent marker were chosen as soilants. Charcoal was chosen to mimic the everyday grime that can be found on surfaces, while permanent marker was chosen to represent an ink stain.
Results
Concentrations of 2% and 4% poly(Cl-TMPM) emulsion were used because these were the lowest concentrations where a total kill could be seen in previously performed antimicrobial tests.
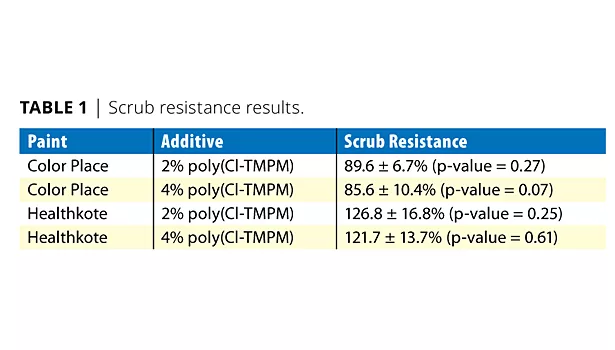
A scrub resistance of 89.6 ± 6.7% (p-value = 0.27) and 85.6 ± 10.4% (p-value = 0.07) was seen in the Color Place paints that contained 2% and 4% poly(Cl-TMPM) respectively. The Healthkote coatings containing 2% and 4% poly(Cl-TMPM) had scrub resistances of 126.8 ±16.8% (p-value = 0.25) for the former and 121.7±13.7% (p-value = 0.61) for the latter (Table 1).
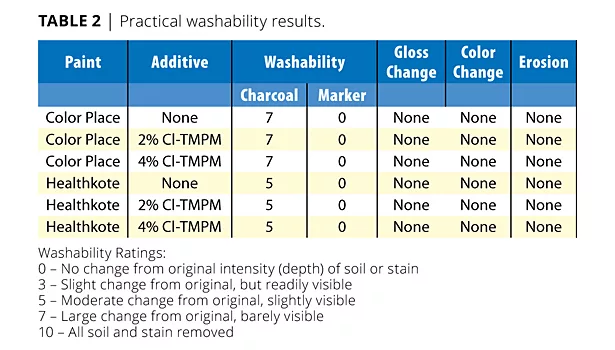
The visibility of the stains after washing were graded according to the scale specified in ASTM Method D4828-94. Charcoal was the first soilant tested. The Color Place paint had a washability rating of 7 for all samples and the control, while Healthkote had a washability rating of 5 for all samples and the control. There was no change from the original intensity of the stain for either paints with or without additive when the permanent marker was used as a soilant. There was also no change in the gloss or color as well as no erosion for all samples (Table 2).
Discussion
The p-values for all of the tests were greater than the predetermined confidence interval of 95%. This would indicate that the null hypothesis should be accepted and that the poly(Cl-TMPM) emulsion additive does not have an effect on scrub resistance.
The same statement can be said about practical washability. There was no difference in washability between the control paints without additive and paints with the additive. This would indicate that the additive had no effect on the washability of the paints.
At first this may seem like a failure since we did not improve scrub resistance or practical washability, but it is our intention that the additive have no effect on the performance characteristics of the paint. From this perspective the tests were a success, and this novel antimicrobial additive is one step closer to becoming an effective tool in the arsenal of products designed to combat microbial growth. n
References
1. Transparency Market Research. Antimicrobial Coatings Market – Global Industry Analysis, Size, Share, Trends, Analysis, Growth and Forecast, 2012-2018. http://www.transparencymarketresearch.com/anti-microbial-coating-market.html. Accessed February 27th, 2013.
2. Wu. X.L.; Ding, J.N.; Gao, J.; Liu, X.X.; Qiu, G.Z. Isolation and identification of metal-resistant iron-oxidinzing bacteria. Minerals and Metallurgical Processing 2007, 24: 57-60.
3. Evans, M.; Davies, J.K.; Sundqvist, G.; Figdor, D. Mechanisms involved in the resistance of Enterococcus faecalis to calcium hydroxide. International Endodontic Journal 2002, 35: 221-228.
4. Park, S.M.; Kim, H.S.; Yu, T. Effect of titanium ion and resistance encoding plasmid of pseudomonas aeruginosa ATCC 10145. Journal of Microbiology 2006, 44: 255-262.
5. Velasco, C.; Rodriguez, J.M.; Conejo, M.C.; Pascual, A. Resistance to imipenem and zinc in clinical isolates of pseudomonas aeruginosa. International Journal of Antimicrobial Agents 2006, 28: 377-378.
6. Gupta, A.; Phung, L.T.; Taylor, D.E.; Silver, S. Diversity of silver resistance genes in IncH incompatibility group plasmids. Microbiology 2001, 147: 3393-3402
7. Silver, S. Bacterial silver resistance: molecular biology and uses and misuses of silver compounds. FEMS Microbiology Review 2003, 27: 341-353.
8. Yazdankhah, S.P.; Scheie, A.A.; Hoiby, E.A.; Lunestad, B.T.; Heir, E.; Fotland, T.O.; Naterstad, K.; Kruse, H. Triclosan and antimicrobial resistance in bacteria: an overview. Microbial Drug Resistance 2006, 12: 83-90.
9. Sundheim, G.; Langsrud, S.; Heir, E.l Holck, A.L. Bacterial resistance to disinfectants containing quaternary ammonium compounds. International Biodeterioration & Biodegradation 1998, 41: 235-239.
10. Cao, Z.; Sun, Y. Polymeric N-halamine latex emulsions for use in antimicrobial paints. ACS Applied Materials Interfaces 2009, 1: 494-504.
11. Worley, S.D.; Williams, D.E. Halamine water disinfectants. CRC Crit Rev Environ Control 1988, 18: 133-175.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






