The Effect of HMMM Crosslinker on the Coating Properties of Chemoenzymatically Synthesized Aliphatic Urethane Oil
The polymers known as urethane oils3,4 include materials that incorporate the carbamate functional group as well as other functional groups. In the present article, castor oil5 is used as a hydroxyl group containing polyol and isophorone diisocyanate as polyfunctional diisocyanate. The hydroxyl functionality of castor oil has been widely utilized to synthesize urethane oils. Castor oil, being a non-drying oil, was enzymatically inter-esterified with linseed oil6 (dry oil) to facilitate the drying of oils. The inter-esterification product thus formed was used for the production of urethane oils.
The present paper illustrates the study of the role of HMMM on coating properties of these new urethane oils synthesized using chemoenzymatically modified castor oil. Lipase, (EC 3.1.1.3) a triglyceride hydrolyzing enzyme,7-9 was used in the synthesis of partial esters to avoid difficulties such as color darkening, incomplete reaction, or loss of ingredients caused by using chemical catalysts. These reactions can be operated at mild conditions and are less prone to side reactions, which make them attractive candidates to conduct inter-esterification reactions.10-12
Experimental
Materials
British Standard Specifications (BSS)-grade castor oil [Acid value, 2.0 mg KOH/g, hydroxyl value 162 mg KOH/g and iodine value (Wij method) 82-90] was procured from M/s Jayant Oil Mill (India). Refined linseed oil was obtained from Mahendra Oil Mill. (Mumbai, India). Isophorone diisocyanate was purchased from E.Merck Ltd. The catalysts used for the synthesis were as follows. Lipozyme IM 60 [7.1 BAUN (Batch Acidolysis Units Novo)/g], commercially available lipase from the fungus mucor miehei immobilized on macroporous anion exchange resin, was obtained from Novo-Nordisk, Denmark. Dibutyl tin dilaurate (DBTL) was obtained from the local market and CYCAT 4040 was procured from Cytec, USA.Crosslinker
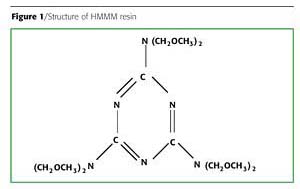
The HMMM used was commercial grade CYMEL 303 (Figure 1) from Cytec, as shown in Table 1.
Reagents
Chloroform, petroleum ether, diethyl ether, acetic acid and toluene were purchased from s. d. fine chem. Ltd., (Mumbai, India).
Methods
Enzymatic Inter-Esterification of Castor Oil and Linseed Oil (Scheme 1)
Castor oil (CO) and linseed oil (LO) in 50:50% w/w ratio were added to a covered reaction kettle equipped with thermometer, stirrer and nitrogen inlet. The mixture was heated in a water bath to 60 ºC and Lipozyme IM 60 catalyst (1.0%w/w based on the reactants) was added. The temperature was maintained between 60 ± 1 ºC, with constant stirring in a nitrogen atmosphere for 16 ± 2 h. The progress of the reaction was monitored by thin layer chromatography. The acid13 and hydroxyl14 values of the resulting interesterification product were determined (Table 2).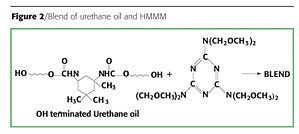
Synthesis of Urethane Oils Based on Inter-Esterification Product
In a reaction kettle, 1.0-gram equivalent of inter-esterification product (IP) was reacted with 0.27-gram equivalent of isophorone diisocyanate. DBTL, 0.01% w/w based on the reaction mixture, was added as a catalyst. The reaction mixture was stirred using toluene (50% w/w based on the reactants) as a solvent. The progress of the reaction was monitored by IR spectroscopy until the -NC peak at 1740 cm-1 disappeared.
The resultant urethane oil (1000 eq. wt.) was then thinned to 50% solids using toluene. A detailed study of coating properties using different percent compositions of HMMM (Figure 2) based on urethane oil solids was conducted. CYCAT 4040 0.5% w/w based on total solids was added as a catalyst.
Characterization
Thin Layer Chromatography
For thin layer chromatographic investigation, 10% (w/v) solutions of IP samples were prepared in chloroform. The developing solvent system was pet-ether, diethyl ether and glacial acetic acid taken in a ratio of 70:30:2 by volume, respectively.IR Spectroscopy
Infrared spectra of the urethane oils were obtained from a Shimadzu FTIR 4200 series spectrophotometer. Being in the form of thick syrup, a thin film of resin was cast over NaCl block.Coating Properties of Urethane Oils
The samples were applied onto mild steel and glass panels using a "Sheen make" bar coater (50m film thickness). Drying times, which include air and baked drying (120 ºC) for the different urethane oil - HMMM compositions were determined.Hardness15: The panels, uniformly coated using the bar coater, were allowed to dry. The relative hardness measurement was achieved by using lead pencils of the same brand ranging in hardness from (softest) 6B 5B 4B........4H 5H 6H (hardest).
Adhesion15: Mild steel panels coated with alkyd resins were examined to measure the adhesion property by using the crosscut adhesion method, which is a simple and effective method for evaluation of adhesion.
Impact Resistance16: Impact resistance was measured using a falling weight-type impact tester (Komal Scientific, Mumbai, India).
Flexibility17: This property is related to elongation and was measured by the conical mandrel test. Tin plates were coated to check for flexibility on conical mandrel (1/4").
Storage Stability: The storage stability of the samples was determined by exposing them to air in cylindrical sample containers for three months. After this period, the sample surface was visually observed and the inferences were drawn.
Chemical Resistance18: Chemical resistance tests were carried out by immersing coated (50m film thickness) glass panels into water, acid and alkali baths. The ends of the glass panels were coated with wax in order to prevent migration under the film from the open ends. The panels were then dipped into water, 3% (w/w) sulphuric acid solution or 1% sodium hydroxide solution and were examined after 24 hours for a change in appearance. The solvent resistance of the cured films was mainly judged from their resistance to acetone, which is evaluated by the rub test.

Results and Discussion
The method used for the synthesis of urethane oils and their cross-linking with HMMM is described in Scheme 1.Enzyme-Catalyzed Inter-Esterification
The inter-esterification reaction between castor oil and linseed oil was monitored by high-performance thin layer chromatography. The reaction time was optimized by terminating the reaction as soon as all triglycerides were used up and before the onset of the free acid formation started. This prevented the acyl migration from sn 1,2 to sn 1,3.19 Also, a gradual increase in the acid value was observed, which has detrimental effects on the coating properties.A hydroxyl-containing intermediate (IP) thus formed was reacted with isophorone diisocyanate to form hydroxyl-terminated urethane oil having 1000 equivalent weight. This urethane oil was further blended with different proportions of HMMM and films were cast on metal and glass panels. The panels were kept in an air-circulated oven initially at 120 ºC for 30 min. and then at 180 ºC for 15 min.
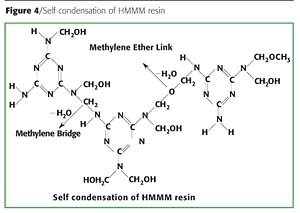
At 120 ºC, the solvent was flashed off and predominant transetherification reaction between hydroxyl-terminated urethane oil and the methoxymethylol group of HMMM took place. This reaction is usually referred to as external crosslinking. At elevated temperatures (180 ºC) an internal crosslinking, known as a self-condensation reaction, occurs between the methoxy groups (OCH3) of the same HMMM molecule, or it takes place through methylene linkages of different HMMM molecules. Other side reactions, which occur to form NCH2N and NCH2OCH2N bridges, play an important roll in the crosslinking process.
Coating Properties
A detailed study was carried out on the coating properties of urethane oils crosslinked with different compositions of HMMM.IR Spectroscopy
IR spectra of urethane oil showed characteristic absorption bands at 1740 cm-1 and 3400 cm-1 corresponding to urethane and amide II (-NH stretching) respectively.
Hardness
It can be seen from Table 3 that as the % composition of HMMM increases, the films become harder. The 100/30 composition of urethane oil and HMMM showed excellent hardness (6H) (Figure 3). This was due to:- Rigidity of the coated films obtained through cyclic isocyanates.
- The crosslinking between HMMM and polyurethane segments, resulting in a complex structure.
- Formation of a heterogeneous phase of excessive hard domains mainly consisting of HMMM self-crosslinking product as the self-condensation (Figure 4) of HMMM dominates the crosslinking process over the reaction between hydroxyl and HMMM.
- The effective insertion of double bonds of LO in CO during the inter-esterification step to form IP.
Adhesion
All the compositions of PU and HMMM showed excellent adhesion (Table 3). As the resin was in solution form and baking was done, the penetration of resin in the irregularities of the surface of the substrate was achieved. It was also because of the basic good wetting properties of urethane oils and HMMM.Flexibility
All the samples tested for flexibility passed the 1/4" bend test and even when the panels were twisted upside down no cracks were observed. Hardness and good flexibility are unique properties of HMMM (Table 3). The good flexibility was also because of the primary film former i.e. urethane oil, which is comprised of two types of crosslinking, the covalent crosslinks and the crosslinks resulting from hydrogen bonding between urethane groups on different molecules. At low levels of stress, the hydrogen bonds remain intact, which at higher stress levels become separated, permitting extension without rupture of the covalent crosslinks. When the stress is removed, the hydrogen bonds re-form.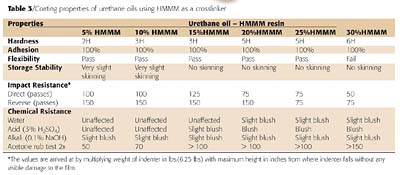
Storage Stability
It can be seen from Table 3 that as the % composition of HMMM increases, the films showed greater storage stability. At low concentrations of HMMM, films showed slight skinning. This may be because of LO, a reactive drying oil with a considerable oxidizing potential to catalyze crosslinking and the formation of skin. The reactivity of linseed oil was due to the high content of linolenic acid (above 50%) having high iodine value. This observation strongly suggests that HMMM can be used as a reactive diluent and an additive for improving the storage stability of the urethane oil.Impact Resistance
In the direct impact test, initially as the composition of HMMM was increased, a slight improvement in the impact resistance was observed. This may be because of the good film flexibility. Good reverse impact resistance was also attributed to the soft elastomeric nature of the castor oil film. At higher % of HMMM, a detrimental effect on coating properties was observed. This was attributed to an increase in the crosslink density of the films formed by self-condensation reactions of HMMM resin forming hard and brittle films. Coating films based on 15% HMMM showed the optimum impact resistance (Table 3).Chemical Resistance
From Table 3, it can be concluded that all the films having different compositions showed very poor resistance as the % composition of HMMM increases. This was due to the loss of crosslinking due to hydrolysis of the acetal linkage. The factor in the hydrolysis of HMMM is the basicity of melamine, which leads to the absorption of acid in the film. Lower levels of HMMM, (5-10%), showed better chemical resistance to attack from alkali, acid and water because of predominant urethane linkages which are more resistant.
The solvent resistance of the cured films was mainly judged from their resistance to acetone. The acetone double rub test revealed that, as the percentage of HMMM in the coating increased the number of double rubs that the coating sustained without any damage, increased. This is attributed to the fact that as the % HMMM increased the crosslinking density of the film was also improved substantially resulting in highly solvent-resistant films. The films with 30% HMMM resin showed the best solvent resistance.
Conclusions
Urethane oils synthesized from enzymatically modified CO were lighter in color than the conventionally synthesized urethane oils. When HMMM was used as a crosslinker, a significant improvement in the hardness and solvent resistance was achieved. All the urethane oils showed excellent adhesion, flexibility, and good impact resistance. It was also observed that HMMM can be used to enhance the storage stability. The only disadvantage of using HMMM is its poor acid/alkali resistance; its use in excess has a detrimental effect on impact resistance. Overall, urethane oils having 15% of HMMM composition, showed optimum film properties.For more information, contact Dr. V.D. Athawale at vilasda@yahooo.com.
References
1 Chu, G.; Jones, F.N. J. Coat. Technol., 891, (1993), 43.
2 CYMEL 303®, Technical bulletin of Cytec Industries Inc., USA.
3 Saunders, K.J. "Organic Polymer Chemistry," First Edition, Chapman and Hall, (1973) 318.
4 Doyle, E. N. The Development and Use of Polyurethanes, 1971.
5 Achaya, K. T. J. Amer. Oil Chem. Soc., 48(11) (1971) 758
6 Formo, M. W. "Paints, Varnishes and Related Products, in Bailey's Industrial Oil and Fat Products", Fourth Edition, Edited by D. Swern, John Wiley and Sons, Inc., New York, 1 (1979) 725.
7 Fogila, T.A.; Petruso, K.; Feairheller, S.H. J. Amer. Oil Chem. Soc. 70 (1993) 281.
8 Marangoni, A.G.; McCurdy, R.D.; Brown, E.D. J. Amer. Oil Chem. Soc. 70 (1993) 737.
9 Kabasakal, O.S.; Guner, F.S.; Arslan, A.; Ergan, A.; Erciyes, A.T.; Yagci, Y. J. Coat. Technol., 68 (860) (1996) 57.
10 Athawale, V.D.; Bhabhe, M.D. J. Coat. Technol. 70 (1998) 43.
11 Klibanov, A.M. CHEMTECH, 16 (1986) 354.
12 Athawale, V.D.; Gaonkar, S.R. Biotechnol. Lett., 15 (1994) 251.
13 ASTM D 1639-90, Standard Test Method for Acid Value of Organic Coating Materials.
14 ASTM D 1957-86, Standard Test Method for Hydroxyl Value of Fatty Oils and Acids.
15 Odell, L.B. "Encyclopedia Of Industrial Chemical Analysis," Enamels, Edited by F. D. Snell and L. S. Ettre, Interscience Publishers a division of John Wiley and Sons, Inc, United States of America , 12 (1971) 170.
16 Roy, T.K; Mannari, V.M.; Raval, D.A. J. Sci. Inds. Res., 55 (1996) 975.
17 ASTM D 522-93a, Elongation of Attached Organic Coatings with Conical Mandrel Apparatus, Annual Book of American Society for Testing and Materials Standards, Philadelphia, Pa., 06.03 (1994) 64.
18 ASTM D 1647-89, Annual Book of American Society for Testing and Materials, 06.01 (1991) 236.
19 Bloomer, S.; Adlercreutz, P.; Mattiason, B. J. Amer. Oil Chem. Soc., 69 (1992) 966.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!







