Amine Blush Testing: Elusive Mystery or Good Old-Fashioned Organic Chemistry?



There is a method now available to determine the presence of an amine blush that is relatively quick, can be used in the lab or the field, and utilizes a simple colorimetric test. This article will discuss this method along with several other methods to detect amine blush that are currently on the market, along with the benefits and drawbacks of these methods.
Industry terms are often used incorrectly, so it is important to first define them to give us a common foundation going forward.
Amines: “A class of organic compounds [bases] of nitrogen that may be considered as derived from ammonia (NH3) by replacing one or more of the hydrogen atoms with alkyl [or aryl] groups.”1
Amine Blush: Amines “…react with atmospheric carbon dioxide and moisture to form a carbamate that can produce a severe surface blush.”2 This is differentiated from the amine rising to the surface without reacting with the water and carbon dioxide, which is more commonly referred to as an amine bloom.
[Amine] Bloom:“A thin coating of an ingredient [an amine in our case]…that migrates to the surface…”1 The term amine bloom is used for the amines at the surface that have not reacted to become a carbamate. These amines at the surface may further react to become blush or, in the absence of moisture or carbon dioxide, may remain as the amines, which may also adversely impact the performance and/or the application of subsequent coatings.
Carbamate: “A salt or ester of carbamic acid. When amines blush, a carbamate is formed when carbon dioxide and moisture react with an amine.”2
Chemistry Involved
Amine blush is a chemical reaction that can occur when environmental conditions are favorable for the amine curing agent on the surface of the coating to react with carbon dioxide and moisture in the atmosphere to form a carbamate (Figure 1).2
Issues Caused by Amine Blush
When an amine-cured epoxy exhibits the blush phenomenon, several problems can occur. The blush may cause low hardness, poor paintability, poor intercoat adhesion, water spotting, poor solvent resistance, and/or poor gloss retention and white solids formation.3 It is important to note that the amount of amine blush may be low enough that it is not readily visible to the unaided eye, but yet may still have adverse affects on the coating. In contrast, when an amine bloom has occurred and is present on the surface, one typically sees a waxy or greasy amber or yellow residue on the surface, leading to the same or similar problems as outlined above.
Risk Factors
The choice of amine during development of the coating is an important factor in determining the risk of forming amine blush. Low-molecular-weight primary amines will have a higher tendency to blush than high-molecular-weight secondary or tertiary amines. Therefore, the properties of the desired coating film must be weighed against the risk of forming the amine blush and the remedial work to remove any blush formed.
Beyond formulation considerations, environmental conditions such as lower temperatures and high humidity need to be considered and assessed, as even the most stringent formulation becomes susceptible for amines to blush in these types of environments. An often overlooked factor that increases the potential for an amine blush to form is the operational industrial equipment used (i.e., heaters) that produces carbon dioxide.
Test Methods
As stated, there are various ways to approach testing for amine blush. On one hand, testing may be performed for the specific carbamate created through submitting samples to a qualified lab and utilizing highly sophisticated and expensive equipment. On the other hand, testing can be less specific, such as a quick field test for the group of compounds called carbamates. While both methods will yield results, the quick field test may be more desirable, as it would allow the contractor to effectively remove the amine blush before over coating, thus eliminating delays or a more costly repair if the affected surface was overcoated prior to the lab results being obtained.
pH Testing
Testing with a pH detection kit should be limited to detecting the presence of amine bloom (where the amine has simply risen to the surface of the coating), but should not be used to address whether amine blush is present. An amine bloom increases the risk that the amine curing agent under the right conditions may blush, but still needs the carbon dioxide and moisture for the amine to blush. Another flaw to using a pH test is that it cannot confirm that a carbamate is present. Additionally, it can give a false positive if the high pH is from some other alkaline compound such as residue from a cleaning process (i.e., sodium hydroxide). Another drawback of the pH test is false negatives. The carbamate formed during the reaction with an amine is typically a neutral salt. It therefore stands to reason (and has been borne out in testing) that if most of the amine is converted to the carbamate, then the pH may no longer be alkaline. The resulting pH test would measure neutral and therefore be considered negative, even though the carbamate is actually present.
Gas Chromatography Mass Spectrometry
One of the more specific types of amine blush testing is Gas Chromatography Mass Spectrometry (GC/MS) or Gas Chromatography Tandem Mass Spectrometry (GC/MS/MS). This is the use of gas chromatography for separation with mass spectroscopy as the detector. A sample of the area where blush is believed to be present is taken and sent to a laboratory with the appropriate equipment. Samples of the amine curing agent or agents should also be sent along with the sample believed to have blushed or it could take additional time to elucidate the structure of the carbamate. The amine curing agent is used to create the standard for the carbamate formed to check the mass spectrum against. After separation into the individual components, the sample enters the mass spectrometer and fragments in a specific pattern, which allows for the identification of the specific curing agent that blushed.
GC/MS has proven quite practical in situations when specific information and identification of amine blush is required, and has been found most useful when the specific carbamate causing the blush needs to be known or where there is a need to distinguish the source of the blush between different coatings. GC/MS has worked well when determining if an outside source of carbamate, such as pesticides, may be the source of the carbamate. The EPA and agricultural industry have developed many methods for the detection or quantification of residual carbamate pesticides in and on such things as foodstuffs, ground water run-off, plants, soil and more.
While the GC/MS technique is easily adapted for use in the coatings industry, several drawbacks to using this test method exist, including the inability of this method to be performed easily in the field, the amount of time this method takes, and the sample has to be shipped to a qualified lab. The lab then needs to create a standard from the curing agent and analyze the data, all of which is expensive due to the costs associated with the lab equipment and the technical expertise needed to interpret the results.
Fourier Transform Infrared Spectroscopy
The Fourier Transform Infrared Spectroscopy (FTIR) technique focuses electromagnetic radiation onto the sample and analyzes the changes in radiation, such as absorption or emission, after it interacts with the sample. During the analysis of a sample containing an amine blush, the spectra obtained separately identifies the asymmetrical stretching band for the carboxylate ion (COO-) from the amine curing agent4 (bond from the RNHCOO- formed in blushing ), seen in the FTIR spectra at 1640 cm-1 to 1550 cm-1 wavenumber region.
While primarily a lab test, the introduction and declining cost of handheld attenuated total reflectance (ATR) FTIR devices has garnered some field use applicability for this test method. However, because of the many and various nuances of properly conducting the test and obtaining results, ATR testing still requires a trained spectroscopist to properly interpret the results. It is the author’s experience that the biggest drawback to using the FTIR test method is that other factors also affect this peak wavenumber region. For example, if the amine curing agent is polyamide (R2NHCO) rather than polyamine (R2NH), a strong peak is already present in the 1650 cm-1 to 1640 cm-1 region. This occurrence would make it quite difficult to determine whether there was a change in the intensity of the peak caused from amine blush. The change in intensity from amine blush may be so small that it is obfuscated under the peak already present from the amide concentration.
Continuing on with this example but considering another variable, if there is an abundance of the amide group present due to the epoxy being mixed off ratio, the spectra would show an increase in intensity in the 1650 cm-1 to 1640 cm-1 region and may have a similar appearance to the spectra from the sample with amine blush; again resulting in suspect conclusions. It should also be noted that if degradation of the sample being analyzed has occurred, the effect on the spectra is also an increase in intensity of a peak near the 1660 cm-1 to 1640 cm-1 range. This fact also illustrates why it is very difficult to rely on the FTIR test method as a positive test for detecting amine blush on the polyamide epoxies; at best FTIR testing can be used to suggest to the user that further testing to determine the cause of the increase in intensity in the 1660 cm-1 to 1640 cm-1 range needs to be conducted.
The Elcometer 139 Amine Blush
Screen Testing Method5
Another method developed and being utilized for detecting amine blush is based on the presence of the carbamate compounds and not on physical phenomena such as pH. This method has field and lab applications, is quick and easy to use, and does not require expensive pieces of laboratory equipment. The medical field and Homeland Security have several different quick methods to test for carbamates in case of emergency, such as pesticide poisoning or a terror threat involving chemical agents. In these instances, there is not an abundance of time to allow for the use of the GC/MS and FTIR equipment; lost time equals lost lives. Instead, these agencies use testing methods that involve a biological response or inhibition of the enzyme cholinesterase to the carbamate class of compounds. The inhibition of cholinesterase takes place as a result of the phosphorylation by organophosphates or carbamylation by carbamates of the serine residue in the site of the enzyme.
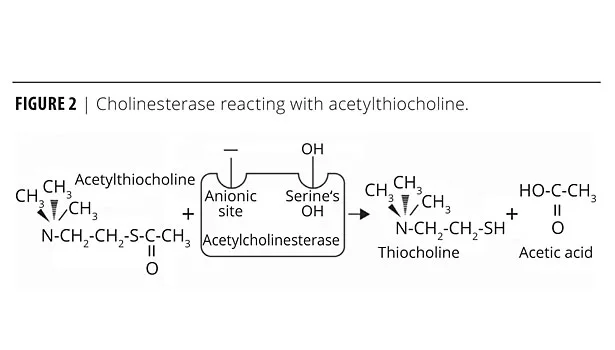
The Elcometer 139 Amine Blush Screen Test Kit(s) testing method was developed for the coatings industry by adapting these test methods based on the biological response of the enzyme to carbamates. Cholinesterase normally reacts with acetylthiocholine (ATC), resulting in the cleavage of acetylthiocholine and yielding acetic acid and thiocholine (Figure 2).
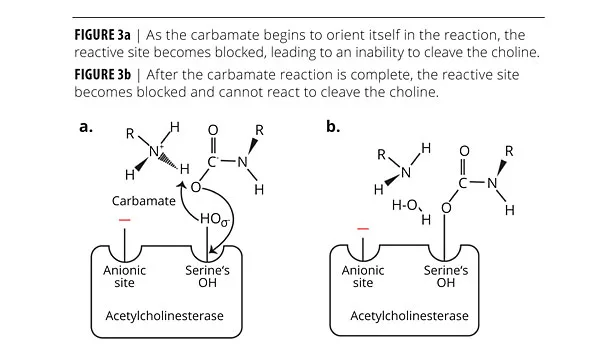
This reaction may easily be observed using Ellman’s reagent. Thiocholine reacts with the chromogen (Ellman’s reagent, also called coloring agent) and turns yellow. If the cholinesterase is exposed to a sample containing a carbamate compound, the carbamate aligns itself with the cholinesterase due to the different polarities at specific active sites within the cholinesterase attracting the oppositely charged sites of the carbamate salt (Figure 3a). The spatial arrangement assists in the reaction of the serine OH from the cholinesterase active site with the carbamate blocking the cholinesterase activity (Figure 3b), thereby preventing the enzyme from reacting with acetylcholine. This phenomenon is referred to as cholinesterase inhibition.
When ATC is added to the sample test solution, the amount of cholinesterase available to cleave the ATC is reduced by the carbamate compound. This results in less thiocholine being available to react with the chromogen, thus yielding a less intense yellow color. As the amount of carbamate compound present in a sample increases, the amount of cholinesterase available decreases and the yellow color decreases.
Field sampling the amine blush using the testing method used in the Elcometer test kit is as simple as getting the amine blush into a solution through either wiping the suspect surface with a swab moistened with IPA or by placing a chip into the test tube supplied. A color control standard is prepared in the same way as the test sample but with no carbamate compound introduced into it, and this control is used for color comparison. The sample solution, prepared with the carbamate compound present or suspected, is tested and the color is compared to the control standard. If all of the available cholinesterase is depleted (inhibited), a colorless solution is observed in the test sample vial, while the control sample vial results in a yellow solution. An alternate method of conducting this test (generally in a lab environment) uses a field-portable spectrophotometer as a quantitative means to evaluate color differences.
Several benefits of this testing kit are that it requires only a few chemicals, it can be performed in a test tube or small vial, it can be conducted onsite in a short period of time by most inspectors or applicators, and the cost of conducting this test is relatively inexpensive compared to the other methods discussed. The testing method of the Elcometer 139 Amine Blush Screen Test Kit(s) is specific to, and qualitatively identifies the presence of, carbamate compounds, thus resulting in the elimination of the prospect of obtaining false positive or false negative results associated with the flawed process of measuring for pH values.
In actuality, one of the few false positives that this test kit could potentially be subject to would be sources of carbamate compounds deposited through means other than the amine blush. Carbamate-based pesticides will result in a carbamate compound being detected, and if the user was not aware of the potential presence of these carbamates, a false positive for amine blush presence would be indicated. It is therefore suggested that the user know their surroundings and identify potential variables or sources of contamination before assigning the origin of the carbamate. However, chemistry is chemistry and contamination is contamination, and even if the source of the carbamate was not the amine, good painting practice would dictate that the carbamate be remediated or the coating may experience the same recoating or performance issues as if it had come from the amine.
As alluded to earlier, a challenge for the coatings industry for this type of test method as compared to the medical, agricultural and Homeland Security personnel is that these entities also detect for organophosphates, while the coatings industry does not. This poses a challenge for the coatings industry, as the potential exists for a false positive result being returned due to organophosphates that may be present on coatings from other outside sources such as agricultural overspray when treating crops. The Elcometer 139 Amine Blush Screen Test Kit(s) addresses this issue by not oxidizing the organophosphates, which results in this kit not detecting organophosphates and thereby lowering the risk of a false positive result being reported.
Summary
The phenomenon known as amine blush has been studied for decades. In 1977, JP Bell et al... described the phenomenon in the article Amine-Cured Epoxy Resins: Adhesion Loss Due to Reaction with Air, and indicated that his article, “…reports the observation of microscopic crystal formation at the interface of the resin with air, resulting in a variably lower resin curing rate, extent of cure, and up to 10 times lower adhesive bond strength. These crystals were identified as an amine bicarbonate salt, resulting from the reaction of the amine at the surface of the curing mixture with air. The bicarbonate formation seems to be general for the types of hardeners used in room-temperature curing.”6 In 1998, Clive H. Hare described the phenomenon as amines reacting “…with atmospheric carbon dioxide and moisture to form a carbamate that can produce severe surface blush.”2 Once identified and realized as being problematic, scientists began working on methods to determine whether amine blush was present on coating surfaces.
During the early 2000s several test methods were developed based on pH. And while these test methods were quick and could be performed in the field, they relied solely on the measurement of hydrogen ion concentration to attempt to indicate an amine blush. But as we have discussed, these pH tests do not identify blush but only the possible presence of an amine that could lead to amine blush. The FTIR method has also historically been utilized to detect amine blush but, as pointed out in this article, has limitations due to the fact that the target peak being analyzed may be affected by the presence of an amide group or degradation, thus making the technician’s ability to distinguish the effect of the amine blush on the peak intensity very difficult. Lastly, the GC/MS method has recently been utilized more as a way of determining the presence of amine blush, but as noted requires sophisticated equipment and technical expertise in analyzing the data, relegating its use to the laboratory environment when one needs to conduct specific testing to pinpoint the exact origin of the carbamate.
Recognizing all of the existing methods’ shortcomings and associated costs with the way amine blush determinations have been investigated in the past, Elcometer worked with a team of forward-thinking coating forensic analytical chemists and a leading immunochemistry product manu-facturing company to develop an innovative approach and test that will accurately detect amine blush compounds. Elcometer 139 Amine Blush Screen Test Kit(s) is the result of these efforts, and is a quickly applied test based on carbamate chemistry and the cholinesterase biological reaction to the carbamate class of compounds.
The healthcare field and Homeland Security have used similar testing for years to determine the presence of carbamates in situations where time was critical. The testing method utilized by the test kit is an accurate and conclusive test for amine blush utilizing methods ranging from swabbing samples in the field to analyzing chips or shavings in the laboratory. Requiring significantly less time (approximately an hour) to test the surface of the epoxy coating for amine blush, one now has the ability to determine conclusively whether an amine blush has occurred and eliminate the risk of an unsatisfactory coating application prior to the application of a subsequent coat of paint.
The Elcometer testing method(s) reveals the presence of amine blush in a method suitable for use in the field or laboratory, and work is currently being done to develop an ASTM standard that utilizes the testing method(s) of the test kit. It is important to note that at this time, the minimum level of carbamate from amine blush needed to create any performance issues with coatings has not been clearly defined. When a positive test for amine blush occurs, it is up to individual owners, contractors and other users of the test to determine when the presence of amine blush needs to be remediated. The test kit is useful in the determination of whether amine blush is present, thereby giving the user some of the information needed to make educated decisions for the best performance of the coating system.
References
1 Lewis, R.J. Hawley’s Condensed Chemical Dictionary 14th Edition, Sr., John Wiley & Sons Inc., New York, 2001, p. 49, 150.
2 Hare, C.H. Protective Coatings: Fundamentals of Chemistry and Composition, Society for Protective Coatings, Pittsburg, PA, 1998, p. 210.
3 Burton, B.L. Amine Curing of Epoxy Resins: Options and Key Formulation Considerations, Paint and Coatings Industry Magazine, June 2006, pp 68-77.
4 Spectrometric Identification of Organic compounds, John Wiley and Sons, New York, 1981, p. 121.
5 Elcometer 139 Amine Blush Screen Testing Method includes the Elcometer 139 Amine Blush Swab Test Kit and the Elcometer 139 Amine Blush Chip Screen Test Kit.
6 Bell, J.P.; Reffner, J.A. and Petrie, S. Amine-cured epoxy resins: Adhesion loss due to reaction with air. Journal of Applied Polymer Science, 1977, 21: 1095–1102. doi: 10.1002/app. 1977.070210423.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






