Cost-Saving, Renewable Dispersant Technology




Raw material cost increases, regulatory compliance and end-user market demand for more environmentally friendly products are some of the challenges faced by paint formulators. The Archer Daniels Midland Company (ADM) is addressing these needs by introducing cost-effective pigment dispersants derived primarily from renewable resources. This article provides an overview of the new dispersant technology, its chemistry and mode of action, presentation of results obtained from testing in model formulations, and a discussion on advantages over current industry practice. The test results support claims that these dispersants can be used to formulate paints with lower cost and excellent stability, together with improved color acceptance, contrast ratios, scrub resistance and enhanced hard water tolerance with less susceptibility to formulation failure from raw material quality variation.
Technology
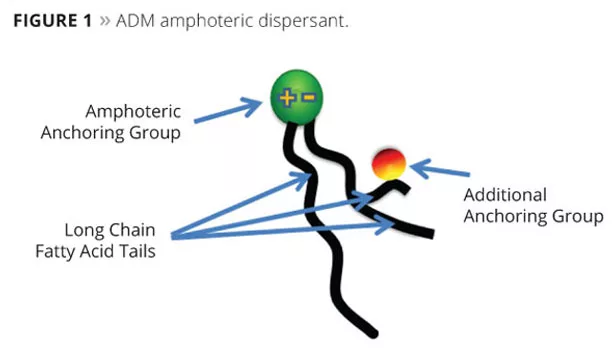
The new renewable dispersant technology is based on amphoteric, non-polymeric chemistry with a schematic molecular structure as shown in Figure 1. The amphoteric structure provides significant advantages over the anionic, nonionic or cationic dispersants traditionally used in paints. Along with the strong pigment anchoring amphoteric head group there are also enhanced fatty acid tails that carry an additional pigment anchoring group. The amphoteric head provides an important key to improved wetting, anchoring and stabilization of pigments. Additional electrostatic and steric stability and compatibility contributions come from the rest of the structure. The unique molecular structure excels at stabilization over a wide variety of pigment chemistries, but is particularly effective with inorganic pigments such as titanium dioxide. The new dispersant has the added benefit of improved surface tension reduction to accelerate pigment wetting and de-aeration, reducing grind energy input and dispersion time without the use of an additional surfactant.
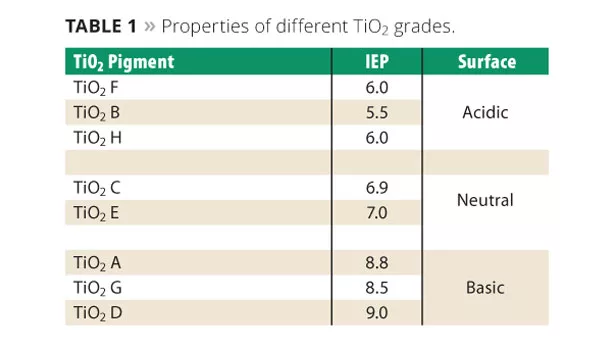
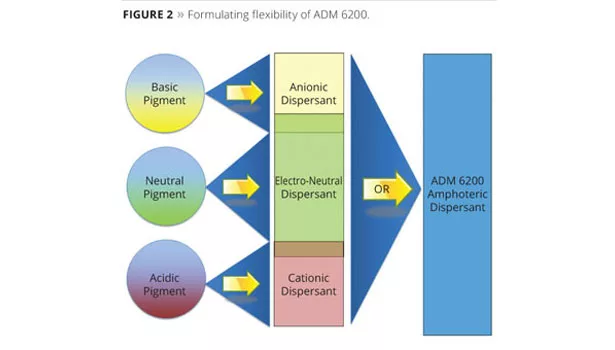
The stabilization of aqueous titanium dioxide (TiO2) dispersions has been widely studied,1,2 and the mechanism is consistent with the Derjaguin, Landau, Verwey and Overbeek (DLVO) theory.3 Pigments with an ionic surface such as TiO2 are best stabilized in the presence of other ions in solution (electrostatic stabilization through the repulsive forces formed).4 However, the ionic character and isoelectric point (IEP) of TiO2 can vary significantly depending upon the surface treatment, as seen with the different grades measured in Table 1. The specific grade of titanium dioxide may therefore require an anionic, cationic or electro-neutral dispersant for proper stabilization. One member of the new dispersant family, designated ADM 6200, provides not only electrostatic stabilization through its amphoteric character, but also adds additional stabilization through its multiple long chain fatty acid tails and added affinic groups. Since it is amphoteric, it can adapt to the change in the ionic character of the titanium dioxide, providing optimum stabilization. Thus, it is effective independent of whether the surface treatment requires an anionic, cationic or electro-neutral dispersant. This provides the formulator the flexibility to use one dispersant system with various TiO2 grades. This flexibility can lead to raw material cost savings. This concept is illustrated in Figure 2.5
Dispersant Mechanism
It has been well documented that the pigment dispersion process can be divided into three stages: 1) wetting 2) dispersing, and 3) stabilization.1,4,6 ADM dispersant technology was evaluated and quantified in all three stages; the stabilization stage and its effect on white tint bases are emphasized in this article. A brief overview is listed below.
Wetting and Dispersing
Effective pigment wetting and dispersing is critical to help obtain maximum processing efficiencies and color development.6 These two steps are intertwined and occur simultaneously. Wetting involves displacing the micro air bubbles around the pigment agglomerates and replacing them with resin, solvent, water or dispersant.4 Once the pigment particles are wetted, energy is added at the dispersion stage, breaking the pigment agglomerates down to their primary particles. The rate of wetting and dispersion is influenced by the surface tension and viscosity of the liquid phase, i.e., the lower the surface tension, the more effective the wetting and dispersion, as expressed by the Washburn equation.7 Faster wetting and more-efficient de-agglomeration lead to better dispersion color development.
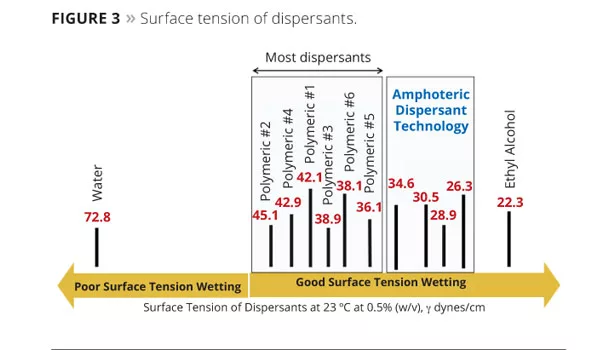
In order to further demonstrate the effectiveness of pigment wetting, the surface tension reduction of water provided by our new amphoteric dispersant technology was compared to a range of commonly used dispersants. Surface tensions of 0.5% (w/v) dispersant solutions were measured at 23 °C using a Kruss Processor – Tensiometer Model K100MK2. The results show that the polymeric dispersants produced a surface tension of approximately 36-46
dynes/cm (Figure 3). The ADM amphoteric dispersant produced a significantly lower surface tension (approximately 26-34 dynes/cm), providing the mechanism for improved pigment particle wetting and deflocculation.
Stabilization
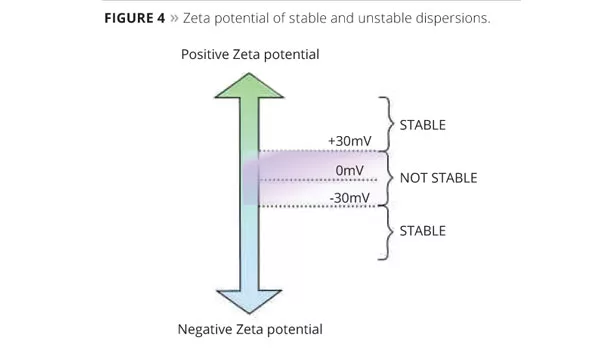
As previously discussed, TiO2 carries an ionic charge and is most effectively dispersed and stabilized electrostatically. Optimal pigment electrostatic stabilization is obtained when the dispersant pigment-affinic groups can interact with the surface charges of the pigment particle. The degree of electrostatic stabilization of the pigment particles can be measured through their Zeta potential at a specific pH, and can help predict dispersion stability. Typically a measured Zeta potential of greater than 30 mV or less than -30 mV produces a stable dispersion (Figure 4).8,9,13
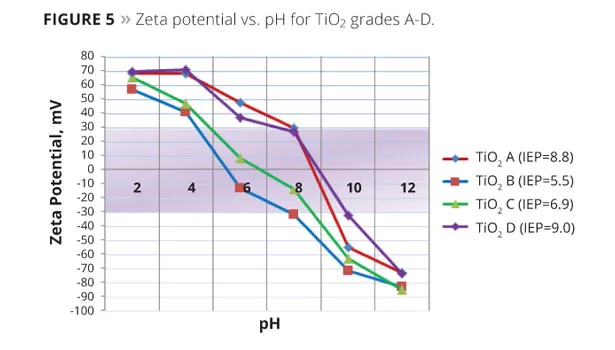
The Zeta potential of four grades of TiO2 found in Table 1 was measured over a wide pH range, as illustrated in Figure 5. As seen, the stability of the TiO2 dispersions varies greatly, dependent upon pH.
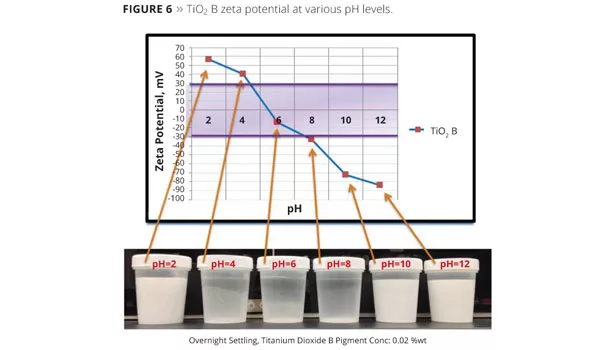
For this study, one grade of TiO2 (B) was chosen from Figure 5. The Zeta potential was measured over a wide pH range using a Zetasizer Nano ZS from Malvern Instruments. All measurements were made in 0.01M KCl both with and without added dispersants. The pH levels of the test dispersions were adjusted with hydrochloric acid and sodium hydroxide solutions. In the absence of dispersants, TiO2 B, with an isoelectric point of 5.5, produced a stable dispersion at a pH less than 4 and greater than 8 (Figure 5). The dispersion was not stable at a pH of 4-8, and the TiO2 quickly fell out of suspension. Figure 6 graphs the Zeta potential and shows the actual dispersion samples at various pH levels. The samples illustrate the stability (milky dispersion) and unstability (translucent) at a specific pH.
When the ADM 6200 amphoteric dispersant was added to the TiO2 (pigment concentration: 0.01% wt., dispersant concentration: 0.01%) the dispersion became stable over a very broad pH range (3-12), as illustrated in Figure 7.
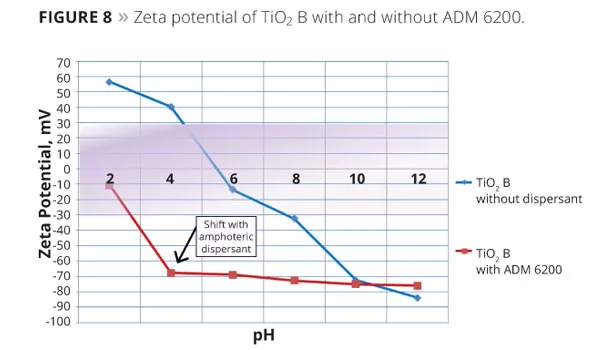
Figure 8 shows the plotted Zeta potential of TiO2 sample B without dispersant (blue line) and with ADM 6200 (red line) over the pH range. Results indicate that the amphoteric ADM 6200 provides TiO2 stabilization over a very broad pH range. Amphoteric chemistry allows ADM 6200 to adapt to the ionic environment of the pigment, regardless of pH.
The Zeta potential of TiO2 B in the presence of an anionic (NH4+ hydrophilic polyacrylate copolymer), nonionic (styrylphenol ethoxylate), cationic (alkylammonium salt of a block copolymer with acidic group), and amphoteric dispersants is shown in Figure 9.10 In the presence of either the cationic or the nonionic dispersant, there is little change in the charge effect on the pigment surface at the pH range studied,10 indicating no increase in electrostatic stability. However, at higher pH the stabilization is independent of the nature of dispersant as the system tends to become more anionic.
With the anionic dispersant, there is a change in charge effect. The IEP for TiO2 B shifts completely, showing some instability at low pH (as the net charge tends to be zero) because of poor ionization characteristics. The anionic dispersants provide good stability of the TiO2, which is why they are commonly used in the formulation of architectural coatings.11
In the case of ADM 6200, amphoteric chemistry allows the dispersant to maintain a high degree of pigment affinity across the pH range. This pigment affinity is so strong at a wide pH range that it remarkably influences the hydrogen bonding around the pigment surface and mimics the action of an anionic dispersant in lowering the Zeta potential of TiO2 B. Such shifts are usually seen with dispersants that can positively influence the charged environment around the pigment through their ability to anchor to the pigment particle with multifunctional groups. Figure 9 indicates that ADM 6200 performs equally as well as commonly used anionic dispersants.
White Tint Base Paint Evaluation
ADM 6200 dispersant was evaluated in architectural white tint bases and its effectiveness compared to the commonly used anionic Na+ and NH4+ copolymer dispersants used in conjunction with nonionic surfactants.
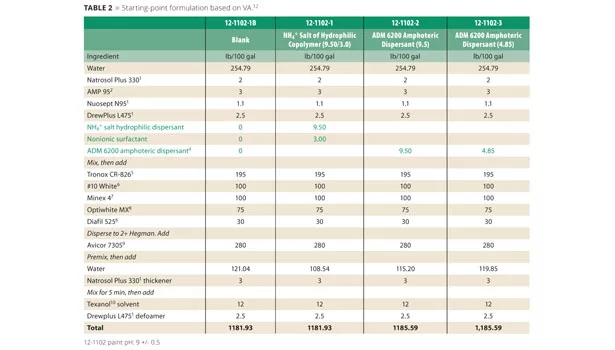
Vinyl acrylic (VA) and vinyl acetate/ethylene (VAE) water-based paint formulas were evaluated using starting-point formulations as published by emulsion manufacturers as controls (with generic raw material substitutions if needed). Complete formulation details are provided in Table 2. Each starting-point formula utilized a common anionic Na+ or NH4+ copolymer dispersant. A blank formulation was also made by removing all dispersant and surfactant present in the control formulation. The blank was used as a baseline vs. the control and formulations containing ADM 6200 amphoteric dispersant. The surfactant was removed from the formulations containing the ADM 6200 due to the good wetting properties.
Vinyl Acrylic Paint
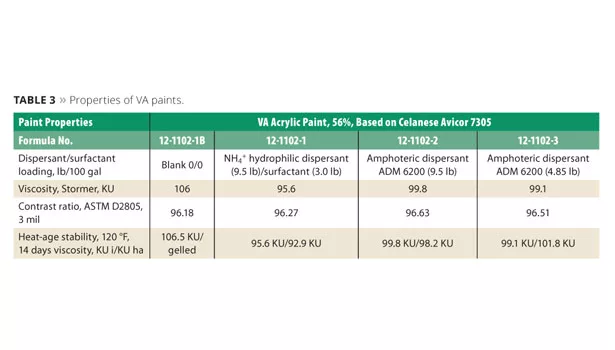
Celanese Avicor 7305 vinyl acrylic was evaluated using the company’s suggested starting-point formula 09-FL56029 incorporating an NH4+salt copolymer dispersant along with a non-ionic surfactant. It was compared to the blank paint and ADM 6200 at two different dispersant levels listed in Table 2. ADM 6200 levels used in these trials represent a 1:1 replacement of the control dispersant on weight and a 1:1 replacement based upon active matter.
Viscosity, heat-age viscosity stability and contrast ratio of the vinyl acrylic paints are listed in Table 3. To measure color acceptance, the paint was tinted by adding 1% of a zero-VOC phthalo blue colorant, Monicolor BH3 from CPS Color. Color development was measured using a BYK handheld Spectro-Guide Sphere Gloss to obtain L*a*b* values.
VA Paint Results
- Blank Paint: Although we were able to make and tint the blank paint without the use of dispersant or surfactant, as expected it was not stable and gelled overnight (Figure 10, Table 3). Therefore, all color development measurements were performed on freshly made samples. It also produced the lowest contrast ratio and color development as measured by b* value (Figure 11).
- Control Paint: The control paint containing the NH4+ anionic dispersant and non-ionic surfactant was very stable. It obtained suitable contrast ratio (Table 3) and color development (Figure 11).
- ADM 6200, 9.5 lb:The formulation was very stable and yielded the highest level of contrast ratio (Table 3) and color development, exceeding the control paint (Figure 11).
- ADM 6200, 4.85 lb: The lowest level of amphoteric dispersant yielded a similar degree of contrast ratio and color development (Figure 11) to that of the control. Figure 10 compares the viscosity stability to that of the blank paint, which was not stable. The lack of surfactant did not adversely affect the stability, contrast ratio or color acceptance compared to the control containing the NH4+ salt of the hydrophilic copolymer dispersant. No compromise in performance was seen, even without the use of surfactant, while lowering the overall cost per gallon.
VAE Paint
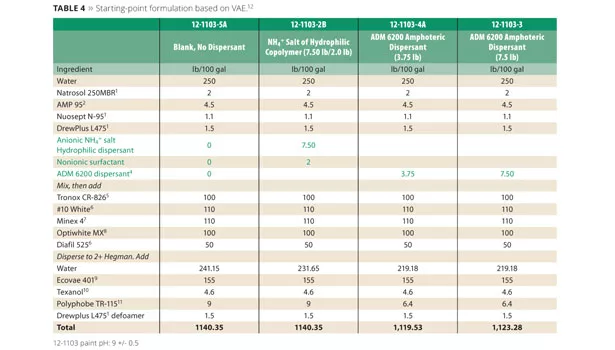
Celanese Ecovae 401 VAE emulsion was evaluated using the company’s suggested starting-point formula 09-FL70009 (Table 4). The starting-point formulation also utilized an NH4+ salt copolymer dispersant along with a non-ionic surfactant. The control was again compared to a blank formulation and two levels of the ADM 6200 without the addition of surfactant.
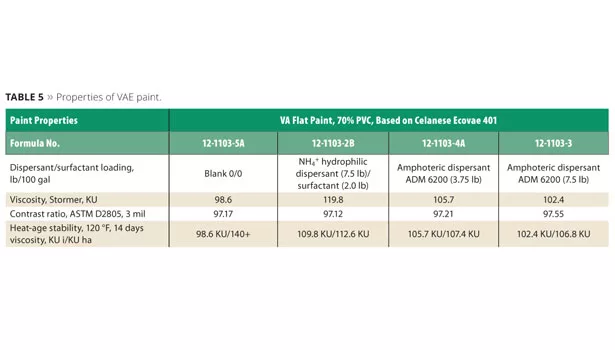
Viscosity and contrast ratio were recorded in Table 5. Excellent heat-age stability of the control formulation and amphoteric dispersant formulations are also noted in Table 5.
To measure color acceptance, the paint was tinted by adding 0.2% of a zero-VOC black colorant, Monicolor CH3 from CPS Color.
VAE Results
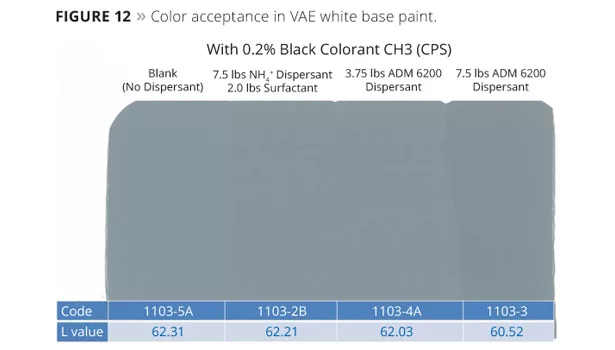
- Blank Paint: Although we were able to make and tint the paint without the use of dispersant or surfactant, the initial viscosity was considerably higher than the control and ultimately not stable. However we were able to tint and measure the contrast ratio and colorant development immediately after making the batch and compare it to that of the control formulation (Figure 12).
- Control Paint: As expected, the control paint containing the NH4+ anionic dispersant and non-ionic surfactant was very stable. It produced an acceptable contrast ratio (Table 5) and color development (Figure 12).
- ADM 6200, 7.5 lb: The formulation without surfactant was very stable (Table 5) and yielded the highest level of contrast ratio and color development, exceeding the control paint (Figure 12).
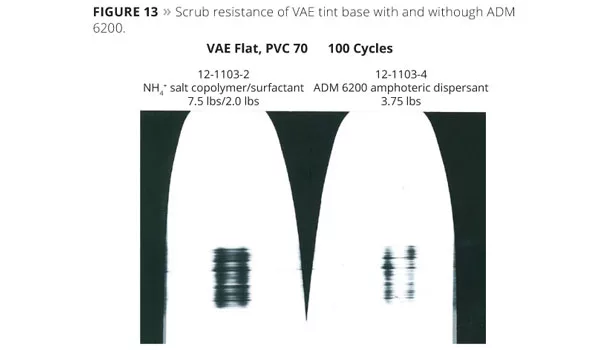
- ADM 6200, 3.75 lb: The lowest level of amphoteric dispersant yielded a similar degree of contrast ratio and color development (Figure 12) to the control paint. The lack of surfactant did not adversely affect the stability, contrast ratio or color acceptance compared to the control containing the NH4+ salt of the hydrophilic copolymer dispersant and non-ionic surfactant. Increasing the dispersant level improved the colorant acceptance without dramatically changing viscosity or contrast ratio. Improved scrub resistance was seen with the ADM 6200-formulated paint, illustrated in Figure 13. It is speculated that the increase in scrub resistance is due to the elimination of surfactant and reduced levels of dispersant, both of which can adversely affect scrub and water resistance.
Conclusions
As expected, the anionic NH4+ salt polymeric dispersant, in conjunction with the nonionic surfactant, provided very good stability and color acceptance in the starting-point formulations. However, the new bio-based ADM 6200 amphoteric dispersant technology proved not only as effective in replacing traditional anionic dispersants in architectural white paints, but offered significant advantages of color acceptance, scrub resistance and stability that also lead to overall cost savings.
The amphoteric dispersant was effective over multiple emulsion technologies including acrylics (not presented here), and offered improved contrast ratio and color acceptance. The efficiency and surface tension advantage, as well as the designed electrosteric stabilization mechanism provided by the amphoteric affinic groups, allowed for the elimination or minimization of added surfactants, further leading to a lower cost/gallon and the improvement of mechanical properties such as scrub resistance.
Overall, the bio-based ADM 6200 amphoteric chemistry provided improved performance properties over traditional NH4+ copolymer dispersants and should be considered when formulating architectural white paint and tint bases. The amphoteric character also offers additional flexibility in raw material sourcing due to its ability to stabilize a variety of anionic, cationic or electro-neutral titanium dioxide pigments efficiently, and it accepts a wide range of both polar and non-polar colorant dispersions, yielding good color development.
The net result of using this novel, versatile dispersant was cost, performance, ease of formulation and bio-based advantages in the architectural white bases.n
Acknowledgments
The author gratefully acknowledges the contributions, testing, editing and development of the data and results of Mario Casco and Dory Tabuena-Salyers, whose contributions made this article possible.
This paper was presented at the 2012 Coatings Trends and Technologies Conference.
References:
1 Stabilization of Titanium Dioxide in Non-Aqueous and Aqueous Coatings, Paint & Coatings Industry, June 1, 2004.
2 Mahli, D.M.; Wegner, J.M.; Glass, J.E.; Phillips, D.G. Waterborne Latex Coatings of Color: II. Surfactant Influences on Color Development and Viscosity, JCT Research, Volume 2, No. 8, October 2005.
3 Croll, S. DLVO theory applied to TiO2 pigments and other materials in latex paints, Department of Polymers and Coatings, North Dakota State University, Progress in Organic Coatings 44 (2002) 131-146.
4 Dispersion Process, http://www.inkline.gr/inkjet/newtech/tech/dispersion/, (accessed 10/30/2012).
5 Adapted from the ATEM Seoul 2012 presentation: Principles and Practice of Pigment Wetting, Dispersion and Stabilization in Organic Coatings.
6 Scholz, W., Paints and Coatings – How Surfactants Can Advance New Developments, CD Proceedings 6th World Surfactant Congress CESIO, Berlin Germany, June 21-23, 2004 (paper # 187).
7 Pal, L.; Fleming, P.D. III. “The Study of Ink Pigment Dispersion Parameters,” The Hilltop Review: Vol. 2 lss.1, Article 9, (2006). Available at: http://scholarworks.wmich.edu/hilltopreview/vol2/iss1/9
8 Mandzy, N.; Grulke, E.; Druffel, T. Breakage of TiO2 agglomerates in electrostatically stabilized aqueous dispersions, Powder Technology 160 (2005) 121-126.
9 Zeta Potential: An Introduction in 30 Minutes, Zetasizer Nano series Amjad, Z., “Factors to Consider in Selecting a Dispersant for Treating Industrial Water Systems,” The Analyst (Association of Water Technologies, Summer-2001 (Vol. VII, No. 3). UPW-Sep-99.
10 Farrokhpay, S.; Morris, G.E.; Fornasiero, D.; Self, P. Influence of Polymer Functional Group Architecture on Titania Pigment Dispersion, Colloids and Surfaces A: PhysicoChem. Eng. Aspects 253 (2005) 183-191.
11 Clayton, J. “Pigment/dispersant interactions in water based coatings”, Pigment & Resin Technology, Vol 27 Iss: 4 pp. 231-239, (1998).
12 Raw Material Supplier Reference Guide:
1. Ashland Chemical Company
2. Dow Chemical
3. Air Products
4. Archer Daniels Midland Company
5. Tronox LLC
6. Imerys
7. Unimin
8. Burgess Pigment Company
9. Celanese
10. Eastman Chemical
11. Arkema
13 Zeta Potential, http://www.silver-colloids.com/Tutorials/Intro/pcs12.html, (accessed 01/28/2013).
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





